Aspirin by CARDINAL HEALTH / Pharbest Pharmaceuticals, Inc. ASPIRIN tablet
Aspirin by
Drug Labeling and Warnings
Aspirin by is a Otc medication manufactured, distributed, or labeled by CARDINAL HEALTH, Pharbest Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
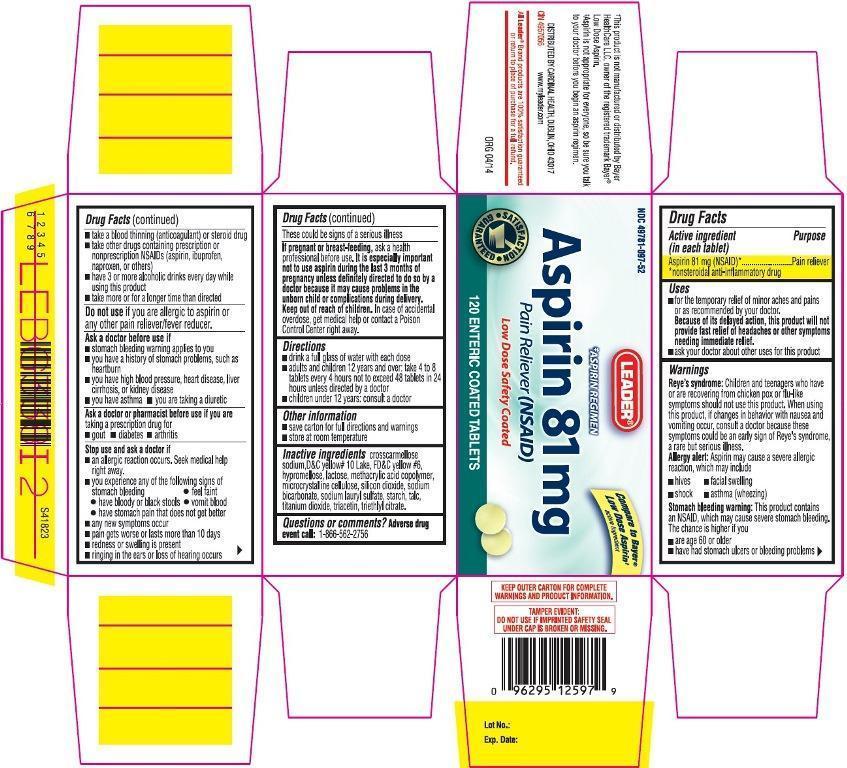
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include- facial swelling
- shock
- hives
- asthma (wheezing)
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Ask a doctor before use if
- stomach bleeding warning applies to you
- you have a history of stomach problems, such as heartburn
- you are taking a diuretic
- you have asthma
- you have high blood pressure, heart disease, liver cirrhosis or kidney disease
Ask a doctor or pharmacist before use if you are
taking a prescription drug for
- gout
- diabetes
- arthritis
Stop use and ask a doctor if
- an allergic reaction occurs. Seek medical help right away.
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- any new symptoms occur
- pain gets worse or lasts more than 10 days
- redness or swelling is present
- ringing in the ears or loss of hearing occurs
These could be signs of a serious illness.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
PRINCIPAL DISPLAY PANEL
NDC: 49781-097-52
LEADER
†Compare to Bayer® low dose aspirin active ingredient
LEADER
#ASPIRIN REGIMEN
Aspirin 81 mg
Pain Reliever (NSAID)
Low Dose Safety Coated
120 ENTERIC COATED TABLETS
-
INGREDIENTS AND APPEARANCE
ASPIRIN
aspirin tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 49781-097 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 81 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) STARCH, CORN (UNII: O8232NY3SJ) HYPROMELLOSES (UNII: 3NXW29V3WO) LACTOSE (UNII: J2B2A4N98G) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM BICARBONATE (UNII: 8MDF5V39QO) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color yellow Score no score Shape ROUND Size 8mm Flavor Imprint Code EA8 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 49781-097-52 1 in 1 CARTON 09/20/2014 1 120 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC: 49781-097-58 300 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part343 09/20/2014 Labeler - CARDINAL HEALTH (097537435) Registrant - Pharbest Pharmaceuticals, Inc. (557054835)
Trademark Results [Aspirin]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ASPIRIN 76481358 2788617 Dead/Cancelled |
Simon Carter Accessories, Ltd. 2003-01-10 |
 ASPIRIN 75209895 not registered Dead/Abandoned |
Bayer Aktiengesellschaft 1996-12-09 |
 ASPIRIN 73234351 1171777 Dead/Cancelled |
McIntyre; William A. 1979-10-09 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.