FLAWLESS FINISH FOUNDATION by J. A. Cosmetics U.S. INC / Shanghai J. A. Cosmetics Trading CO., LTD.
FLAWLESS FINISH FOUNDATION by
Drug Labeling and Warnings
FLAWLESS FINISH FOUNDATION by is a Otc medication manufactured, distributed, or labeled by J. A. Cosmetics U.S. INC, Shanghai J. A. Cosmetics Trading CO., LTD.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
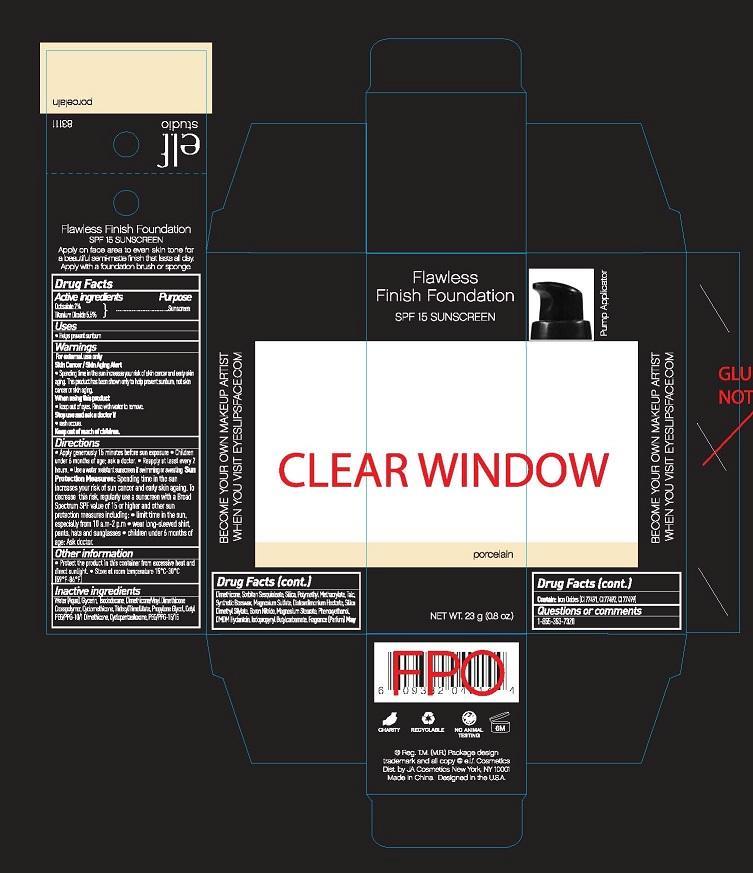
FLAWLESS FINISH FOUNDATION SPF 15 SUNSCREEN- octisalate, titanium dioxide liquid
J. A. Cosmetics U.S. INC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Warnings:
For external use only
SKIN CANCER/SKIN AGING ALERT
- SPENDING TIME IN THE SUN INCREASES YOUR RISK OF SKIN CANCER AND EARLY SKIN AGING. THIS PRODUCT HAS BEEN SHOWN ONLY TO HELP PREVENT SUNBURN, NOT SKIN CANCER OR SKIN AGING.
Directions:
- APPLY GENEROUSLY 15 MINUTES BEFORE SUN EXPOSURE
- CHILDREN UNDER 6 MONTHS OF AGE; ASK A DOCTOR.
- REAPPLY AT LEAST EVERY 2 HOURS.
- USE A WATER RESISTANT SUNSCREEN IF SWIMMING OR SWEATING.
SUN PROTECTION MEASURES: SPENDING TIME IN THE SUN INCREASES YOUR RISK OF SUN CANCER AND EARLY SKIN AGING. TO DECREASE THIS RISK, REGULARLY USE A SUNSCREEN WITH A BROAD SPECTRUM SPF VALUE OF 15 OR HIGHER AND OTHER SUN PROTECTION MEASURES INCLUDING:
- LIMIT TIME IN THE SUN, ESPECIALLY FROM 10 A.M. - 2 P.M.
- WEAR LONG-SLEEVED SHIRT, PANTS, HATS AND SUNGLASSES
- CHILDREN UNDER 6 MONTHS OF AGE: ASK A DOCTOR.
OTHER INFORMATION
- PROTECT THE PRODUCT IN THIS CONTAINER FROM EXCESSIVE HEAT AND DIRECT SUNLIGHT.
- STORE AT ROOM TEMPERATURE 15°C - 30°C (59°F - 86°F)
Inactive Ingredients:
WATER (AQUA), GLYCERIN, ISODODECANE, DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER, CYCLOMETHICONE, TRIDECYLTRIMELLITATE, PROPYLENE GLYCOL, CETYL PEG/PPG-10/1 DIMETHICONE, CYCLOPENTASILOXANE, PEG/PPG-15/15 DIMETHICONE, SORBITAN SESQUIOLEATE, SILICA, POLYMETHYL METHACRYLATE, TALC, SYNTHETIC BEESWAX, MAGNESIUM SULFATE, DISTEARDIMONIUM HECTORITE, SILICA DIMETHYL SILYLATE, BORON NITRIDE, MAGNESIUM STEARATE, PHENOXYETHANOL, DMDM HYDANTOIN, IODOPROPYNYL BUTYLCRBOMATE, FRAGRANCE (PARFUM) MAY CONTAIN: IRON OXIDES (CI 77491, CI 77492, CI 77499)
| FLAWLESS FINISH FOUNDATION
SPF 15 SUNSCREEN
octisalate, titanium dioxide liquid |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - J. A. Cosmetics U.S. INC (186705047) |