LEUCOVORIN CALCIUM tablet
Leucovorin Calcium by
Drug Labeling and Warnings
Leucovorin Calcium by is a Prescription medication manufactured, distributed, or labeled by Ingenus Pharmaceuticals, LLC, Novast Laboratories, Ltd., Ingenus Pharmaceuticals NJ, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LEUCOVORIN CALCIUM TABLETS safely and effectively. See full prescribing information for LEUCOVORIN CALCIUM TABLETS.
LEUCOVORIN CALCIUM tablets, for oral use
Initial U.S. Approval: 1952RECENT MAJOR CHANGES
INDICATIONS AND USAGE
Leucovorin calcium tablets are a folate analog indicated:
- To reduce the toxicity of:
- Methotrexate in adult patients with impaired methotrexate elimination, and
- Folic acid antagonists or dihydrofolate reductase (DHFR) inhibitors following an overdose in adult patients. (1.1)
- For the treatment of cerebral folate transport deficiency in adult and pediatric patients who have a confirmed variant in the folate receptor 1 gene (FOLR1-CFTD). (1.2)
Limitations of Use
Leucovorin calcium tablets are not recommended for use in patients with a deficiency of methenyltetrahydrofolate synthetase (MTHFS) because MTHFS is a primary enzyme in the metabolism of leucovorin to 5-methenyltetrahydrofolate. (1.2)
Limitations of Use
Leucovorin calcium tablets are not indicated for the treatment of pernicious anemia or other megaloblastic anemias, due to the lack of vitamin B12, because of the risk of progression of neurologic manifestations despite hematologic remission. (1.3)
DOSAGE AND ADMINISTRATION
- Leucovorin calcium tablets are for oral administration only and can be taken with or without food. Crushing of leucovorin tablets and mixing with food or liquid has been reported in literature. (2.1)
- Administer leucovorin calcium tablets as soon as possible after a folic acid antagonist or dihydrofolate reductase (DHFR) inhibitor overdose and within 24 hours of methotrexate use when there is impaired methotrexate elimination. (2.2)
Recommended Dosage to Reduce Methotrexate Toxicity in Patients with Impaired Methotrexate Elimination
- 10 mg/m2 (up to 25 mg) orally every 6 hours until the serum methotrexate levels are less than 10-8M (0.01 micromolar). If a dosage greater than 25 mg every 6 hours is needed, an injectable formulation of leucovorin should be administered parenterally. (2.2)
Recommended Dosage to Reduce the Toxicity of Folic Acid Antagonists or DHFR Inhibitors in Patients Following an Overdosage
- 5 mg to 15 mg per day. (2.2)
- For patients with impaired methotrexate elimination and following a methotrexate overdose, administer intravenous fluids (3 L/day) and alkalinize the urine to maintain the urine pH at 7.0 or greater. (2.2)
Recommended Dosage to Treat FOLR1-CFTD
DOSAGE FORMS AND STRENGTHS
Tablets: 5 mg of leucovorin, 10 mg of leucovorin, 15 mg of leucovorin and 25 mg of leucovorin (3)
CONTRAINDICATIONS
History of hypersensitivity reaction, depending on indication, to leucovorin (folinic acid), levoleucovorin, folic acid, or any component of leucovorin calcium tablets:
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
Adverse reactions included pruritus, rash, urticaria, dyspnea, hypersensitivity reactions, rigors, and temperature change. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Ingenus Pharmaceuticals, LLC at 1-877-748-1970 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Certain Antiepileptic Drugs: Increase monitoring for seizure activity in leucovorin calcium tablets-treated patients taking certain concomitant antiepileptic drugs. Certain antiepileptic drugs may reduce the effectiveness of leucovorin calcium tablets. (7.1, 7.2)
- Trimethoprim-Sulfamethoxazole: Avoid concomitant use of leucovorin calcium tablets with trimethoprim-sulfamethoxazole. (7.1)
- Fluorouracil: Leucovorin may enhance the toxicity of fluorouracil. Deaths from severe enterocolitis, diarrhea, and dehydration have been reported in elderly patients. (7.1)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2026
- To reduce the toxicity of:
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Reduction of Toxicity of Folic Acid Antagonists or Dihydrofolate Reductase Inhibitors
1.2 Cerebral Folate Transport Deficiency with Folate Receptor 1 Genetic Variant
1.3 Limitations of Use
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
2.2 Recommended Dosage to Reduce the Toxicity of Methotrexate in Patients with Impaired Methotrexate Elimination or to Reduce the Toxicity of Folic Acid Antagonists or Dihydrofolate Reductase Inhibitors Following Overdose
2.3 Recommended Dosage for Cerebral Folate Transport Deficiency with Folate Receptor 1 Genetic Variant
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 Effects of Leucovorin on Other Drugs
7.2 Effect of Other Drugs on Leucovorin
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Cerebral Folate Transport Deficiency with Folate Receptor 1 Genetic Variant (FOLR1-CFTD)
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Reduction of Toxicity of Folic Acid Antagonists or Dihydrofolate Reductase Inhibitors
Leucovorin calcium tablets are indicated to reduce the toxicity of:
- Methotrexate in adult patients with impaired methotrexate elimination, and
- Folic acid antagonists or dihydrofolate reductase (DHFR) inhibitors following an overdose in adult patients.
1.2 Cerebral Folate Transport Deficiency with Folate Receptor 1 Genetic Variant
Leucovorin calcium tablets are indicated for the treatment of cerebral folate transport deficiency in adult and pediatric patients who have a confirmed variant in the folate receptor 1 gene (FOLR1-CFTD).
Limitations of Use
Leucovorin calcium tablets are not recommended for use in patients with a deficiency of methenyltetrahydrofolate synthetase (MTHFS) because MTHFS is a primary enzyme in the metabolism of leucovorin to 5-methenyltetrahydrofolate (5-MTHF) [see Clinical Pharmacology (12.3)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
Each indication has a different method for calculating the dosage (i.e., fixed dosage, body surface area-based dosage, or body weight-based dosage). Ensure that the correct method for calculating the dosage is used [see Dosage and Administration (2.2, 2.3)].
Leucovorin calcium tablets are for oral administration only and can be taken with or without food [see Clinical Pharmacology (12.3)]. Crushing of leucovorin tablets and mixing with food or liquid (e.g., water, breastmilk, infant formula) has been reported in literature. If administering via this method, administer immediately after mixing.
2.2 Recommended Dosage to Reduce the Toxicity of Methotrexate in Patients with Impaired Methotrexate Elimination or to Reduce the Toxicity of Folic Acid Antagonists or Dihydrofolate Reductase Inhibitors Following Overdose
Administer leucovorin calcium tablets as soon as possible after a folic acid antagonist or DHFR inhibitor overdose and within 24 hours of methotrexate administration when there is impaired methotrexate elimination. The effectiveness of leucovorin calcium tablets decreases as the time interval between leucovorin calcium tablets administration and the folic acid antagonist or DHFR inhibitor increases.
For patients with impaired methotrexate elimination, monitor serum methotrexate concentrations and serum creatinine to determine the recommended dosage and duration of leucovorin calcium tablets.
For patients with impaired methotrexate elimination and in patients following a methotrexate overdose, administer intravenous fluids (3 Liters per day) and alkalinize the urine to maintain a urine pH of 7.0 or greater.
The recommended leucovorin calcium tablets dosage to reduce methotrexate toxicity in patients with impaired methotrexate elimination:
- 10 mg/m2 (up to 25 mg) orally every 6 hours until the serum methotrexate levels are less than 10-8M (0.01 micromolar).
- When a dosage greater than 25 mg every 6 hours is needed for this use, leucovorin calcium tablets are not recommended because this dosage and the formulation may be inadequate to treat significant methotrexate toxicity, resulting in possible methotrexate toxicity fatalities. Refer to the prescribing information for leucovorin injection for dosage recommendations.
- If the 24-hour serum creatinine has increased 50% over baseline or if the 24-hour methotrexate level is greater than 5 x 10-6 M or the 48-hour level is greater than 9 x 10-7 M, higher doses of leucovorin are needed; discontinue oral administration of leucovorin calcium tablets and administer leucovorin intravenously or intramuscularly. Refer to the prescribing information for leucovorin injection for the appropriate dosage and duration.
- In patients who experience non-oliguric renal failure, continue leucovorin, hydration and alkalinization of the urine (pH of 7.0 or greater) until the methotrexate level is 0.05 micromolar.
- Extend the duration of leucovorin calcium tablets administration for an additional 24 hours in subsequent courses of methotrexate in patients who experience significant methotrexate toxicities, impaired methotrexate elimination including third-space fluid accumulation and inadequate hydration, or renal impairment.
The recommended leucovorin calcium tablets dosage to reduce the toxicity of folic acid antagonists or DHFR inhibitors (trimethoprim or pyrimethamine) following an overdose:
- 5 mg to 15 mg orally once daily.
2.3 Recommended Dosage for Cerebral Folate Transport Deficiency with Folate Receptor 1 Genetic Variant
The recommended oral dosage of leucovorin calcium tablets for patients with FOLR1-CFTD is based on the patient's weight (see Table 1). Adjust dosage based on clinical response [see Clinical Studies (14.1)].
Table 1. Recommended Leucovorin Calcium Tablets Dosage for Patients with FLOR1-CFTD - * Round doses to the nearest tablet strength or combination of strengths.
- † Bioavailability is reduced with individual leucovorin doses above 25 mg in adults [see Clinical Pharmacology (12.3)].
Patient Weight Initial Total Daily Dosage* Maximum Total Daily Dosage* Frequency of
Administration†Less than 40 kg 1 to 2 mg/kg/day 8.5 mg/kg/day Administer the total daily dosage once daily or in divided doses up to 6 times per day. Single doses of 25 mg or less are preferred; do not administer more than 75 mg as a single dose. 40 kg or more 1 to 2 mg/kg/day 330 mg/day -
3 DOSAGE FORMS AND STRENGTHS
Leucovorin calcium tablets, USP:
- 5 mg: White to off-white, round, biconvex tablets debossed with “ING” above “181” on one side and scoreline on other side and free from physical defects.
- 10 mg: White to off-white, round, biconvex tablets debossed with “ING” above “182” on one side and score line on other side and free from physical defects.
- 15 mg: Peach colored, round, biconvex tablets debossed with “ING” above “183” on one side and score line on other side and free from physical defects.
- 25 mg: Peach colored, round, biconvex tablets debossed with “ING” above “184” on one side and scoreline on other side and free from physical defects.
-
4 CONTRAINDICATIONS
Leucovorin calcium tablets are contraindicated in patients with a history of hypersensitivity reaction depending on indication as described below, to leucovorin (folinic acid), levoleucovorin, folic acid, or any component of leucovorin calcium tablets [see Description (11)]:
- Folic acid antagonist or DHFR inhibitor toxicity: history of severe hypersensitivity reaction
- FOLR1-CFTD: history of any hypersensitivity reaction
Reactions have included anaphylactic reactions [see Warnings and Precautions (5.1)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylactic reactions and urticaria, have been reported following the administration of leucovorin. Leucovorin calcium tablets are contraindicated for the treatment of folic acid antagonist or DHFR inhibitor toxicity in patients with a history of a severe hypersensitivity reaction and for the treatment of FOLR1-CFTD in patients with a history of any hypersensitivity reaction to leucovorin, levoleucovorin, folic acid, or any component of leucovorin calcium tablets [see Contraindications (4)]. Withhold or permanently discontinue leucovorin calcium tablets based on the severity of hypersensitivity.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)].
The following adverse reactions have been identified during postapproval use of leucovorin (d,l-leucovorin) or levoleucovorin (l-leucovorin). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Dermatologic: Pruritus, rash.
- Respiratory: Dyspnea.
- Other Clinical Events: Rigors, temperature change.
Safety information for the treatment of FOLR1-CFTD with oral leucovorin is limited. The available evidence is based on published case reports [see Clinical Studies (14.1)].
-
7 DRUG INTERACTIONS
7.1 Effects of Leucovorin on Other Drugs
Certain Antiepileptic Drugs
Increase monitoring for seizure activity in leucovorin calcium tablets-treated patients taking certain concomitant antiepileptic drugs.
Folic acid in high doses may reduce the effectiveness of certain antiepileptic drugs (e.g., phenobarbital, phenytoin, and primidone) and thereby increase the frequency of seizures in susceptible patients, including pediatric patients. It is not known whether folinic acid, including leucovorin calcium tablets, has the same effects; however, both folic and folinic acids, including leucovorin calcium tablets, share some common metabolic pathways.
Trimethoprim-Sulfamethoxazole
Avoid concomitant use of leucovorin calcium tablets with trimethoprim-sulfamethoxazole.
The effectiveness of trimethoprim-sulfamethoxazole can be decreased if used concomitantly with leucovorin calcium tablets, which was associated with increased rates of treatment failure and mortality in patients with HIV infection who receive trimethoprim-sulfamethoxazole for the acute treatment of Pneumocystis jirovecii pneumonia.
Fluorouracil
Leucovorin may enhance the toxicity of fluorouracil. Deaths from severe enterocolitis, diarrhea, and dehydration have been reported in elderly patients receiving weekly leucovorin and fluorouracil. Concomitant granulocytopenia and fever were present in some but not all of the patients.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data on the intermittent use of leucovorin for the treatment of folic acid antagonist or DHFR inhibitor toxicity during pregnancy have not identified a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. There are no adequate data on the use of leucovorin for the treatment of FOLR1-CFTD in pregnant women. Adequate animal reproductive and developmental studies have not been conducted with leucovorin. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Risks with Concomitant Use of Leucovorin Calcium Tablets and Chemotherapy
Drugs administered in combination with leucovorin calcium tablets may cause fetal harm. Refer to the Prescribing Information for the chemotherapy administered in combination with leucovorin calcium tablets for additional information, as appropriate.
8.2 Lactation
Risk Summary
There are no data on the presence of leucovorin in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for leucovorin calcium tablets and any potential adverse effects on the breastfed infant from leucovorin or from the underlying maternal condition. Refer to the Prescribing Information for chemotherapy administered in combination with leucovorin calcium tablets for breastfeeding recommendations, as appropriate.
8.4 Pediatric Use
Leucovorin calcium tablets are indicated for the treatment of cerebral folate transport deficiency in pediatric patients who have a confirmed variant in the folate receptor 1 gene (FOLR1-CFTD) [see Clinical Studies (14.1)].
The safety and effectiveness of leucovorin calcium tablets have not been established to reduce the toxicity of methotrexate in pediatric patients with impaired methotrexate elimination or in pediatric patients to reduce the toxicity of folic acid antagonists or dihydrofolate reductase (DHFR) inhibitors following an overdose.
Folic acid in large amounts may counteract the antiepileptic effect of phenobarbital, phenytoin, and primidone, and increase the frequency of seizures in susceptible pediatric patients [see Drug Interactions (7.1)].
8.5 Geriatric Use
There is insufficient information in patients 65 years of age and older on the use of leucovorin calcium tablets to reduce the toxicity of methotrexate, other folic acid antagonists, or DHFR inhibitors, and there is no information on the use of leucovorin calcium tablets to treat FOLR1-CFTD in patients 65 years of age and older to determine whether they respond differently from younger patients [see Clinical Studies (14.1)].
- 10 OVERDOSAGE
-
11 DESCRIPTION
Leucovorin is a racemic mixture of the 5-formyl derivative of tetrahydrofolic acid. The biologically active compound of the mixture is the (-)-L-Levoisomer, known as Citrovorum factor, or (-)-folinic acid or levoleucovorin. Leucovorin is a water soluble form of reduced folate in the folate group.
The chemical name of leucovorin, a folate analog, is the calcium salt of N-[4-[[(2-amino-5-formyl-1,4,5,6,7,8-hexahydro-4-oxo-6-pteridinyl)methyl] amino]benzoyl]-L-glutamic acid. The molecular formula is C20H21CaN7O7 and the molecular weight is 511.51 g/mol. The structural formula of leucovorin calcium is:
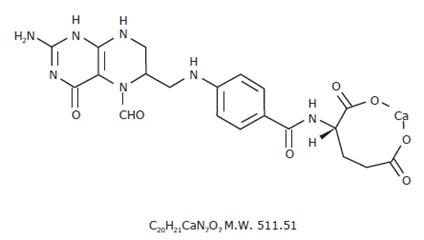
Leucovorin calcium tablets, USP are for oral administration. Each 5 mg tablet contains 5 mg of leucovorin (equivalent to 5.4 mg of leucovorin calcium), each 10 mg tablet contains 10 mg of leucovorin (equivalent to 10.8 mg of leucovorin calcium), each 15 mg tablet contains 15 mg of leucovorin (equivalent to 16.2 mg of leucovorin calcium) and each 25 mg tablet contains 25 mg of leucovorin (equivalent to 27.01 mg of leucovorin calcium). The 5 mg, 10 mg, 15 mg and 25 mg tablets contain the following inactive ingredients: lactose monohydrate, anhydrous lactose, microcrystalline cellulose, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate. Additionally, the 15 mg and 25 mg tablets contains FD&C yellow #6.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Levoleucovorin, a reduced folate and the pharmacologically active isomer of leucovorin (5-formyltetrahydrofolic acid), can mitigate the toxic effects of folate antagonists, including methotrexate and other agents that inhibit dihydrofolate reductase (DHFR). Inhibition of DHFR blocks the formation of tetrahydrofolate, an essential cofactor for DNA synthesis and repair.
Levoleucovorin has been observed to increase levels of 5-MTHF, an active metabolite of folate, in case studies of FOLR1-CFTD [see Clinical Studies (14.1)].
12.2 Pharmacodynamics
Levoleucovorin and its metabolites (5,10-methenyltetrahydrofolate, 5,10-methylenetetrahydrofolate, and 5-MTHF) serve as cofactors in “one carbon” metabolism. These reactions are involved in the generation of nucleic acids and the regulation of gene expression.
12.3 Pharmacokinetics
Leucovorin is a racemic mixture of (l)-or levoleucovorin and (d)-or dextroleucovorin. Following oral administration of leucovorin to healthy adults, dextroleucovorin, levoleucovorin, and 5-MTHF exposures increased in a dose proportional manner with doses up to 25 mg, but in a less than dose proportional manner with doses greater than 25 mg.
Absorption
Following oral administration of leucovorin in adults, the apparent bioavailability of levoleucovorin is 97% for 25 mg, 75% for 50 mg, and 37% for 100 mg, and dextroleucovorin is approximately 19% for 25 mg, 20% for 50 mg, and 7% for 100 mg. After a single oral 15 mg (7.5 mg/m2) dose of leucovorin calcium tablets, time to peak serum folate concentration is 1.7 hours.
Effect of Food: The effect of food on the pharmacokinetics of leucovorin calcium tablets has not been evaluated. As leucovorin is a highly soluble and well absorbed drug, and different immediate-release oral formulations of leucovorin (oral tablet and oral solution) showed relatively higher bioavailability of total folates (>95%), food is not expected to have a clinically significant effect on the pharmacokinetics of leucovorin or 5-MTHF. Crushing of leucovorin and mixing with food or liquid has been reported in literature.
Distribution
Levoleucovorin is minimally bound to human serum albumin. The reported human serum albumin binding of 5-MTHF ranges from 42-49%.
Leucovorin is not observed in cerebrospinal fluid (CSF) and 5-MTHF is reported to accumulate in CSF in children with leukemia.
Elimination
After intravenous administration of leucovorin in adults, the reported mean plasma elimination half-life in the literature was 0.5-1.3 hours for levoleucovorin and 3-7 hours for 5-MTHF.
Metabolism: Following administration of oral leucovorin, levoleucovorin undergoes metabolism in intestinal cells via methenyltetrahydrofolate synthetase (MTHFS) and methylenetetrahydrofolate reductase (MTHFR) to its active metabolite, 5-MTHF. 5-MTHF is the main active metabolite in plasma after oral administration of leucovorin.
Excretion: Leucovorin is mainly excreted by the kidney as unchanged dextroleucovorin, levoleucovorin, or as 5-MTHF, the metabolic product of levoleucovorin.
Specific Populations
Patients with Renal Impairment: The kidney is reported to contribute to the elimination of dextroleucovorin, levoleucovorin and its active metabolite, and plasma concentrations of dextroleucovorin, levoleucovorin, and 5-MTHF may be increased in patients with renal impairment. However, clinical studies on the impact of renal impairment have not been conducted.
Patients with Hepatic Impairment: The liver is reported to contribute to the metabolism of levoleucovorin, and plasma concentrations of levoleucovorin and 5-MTHF may be increased in patients with hepatic impairment. However, clinical studies on the impact of hepatic impairment have not been conducted.
- 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
14.1 Cerebral Folate Transport Deficiency with Folate Receptor 1 Genetic Variant (FOLR1-CFTD)
FOLR1-CFTD is a very rare neurological syndrome. No clinical trials have been conducted to examine the efficacy and safety of leucovorin in patients with FOLR1 variants. Evidence for the efficacy and safety of leucovorin in patients with FOLR1-CFTD was derived from the published literature. Forty-six patients with FOLR1-CFTD who received leucovorin treatment via various administration routes were identified in 26 published case reports and case reviews through 2024. Thirty cases were described in more than one publication.
Of the 46 patients, a total of 27 (59%) were reported as having received leucovorin only via the oral administration route. These 27 patients ranged from approximately 2 months of age to 33 years of age at treatment initiation, and 25 of the patients had dosing information. The starting oral dose ranged from 0.5 to 3 mg/kg/day, and was 2 mg/kg/day in 14 of the 25 patients. Of the 20 patients with dose escalation reported, 17 had a maximum dose ≤6 mg/kg/day (reported range: 1.7 to 8.5 mg/kg/day). Information related to duration of use of leucovorin was limited, and there was no obvious relationship between the starting or maximum oral dose with patient demographics or disease severity. In some cases, it was reported that dose increase was based on clinical review of patient response. A range of clinical improvements in various neurological symptoms following treatment with oral leucovorin was reported for 24 of the 27 patients (e.g., reduction in severity or number of seizures; improvements in motor function, communication, and/or behavior). The remaining 3 patients showed either no change or no progression of symptoms; both the observed clinical improvements and the lack of disease progression are unexpected when compared to the progressive natural history of these patients with FOLR1-CFTD.
CSF 5-MTHF measurements were collected at varying, unspecified time points across patients, with timing broadly categorized as before or after treatment initiation in most cases. In the subset of 27 FOLR1-CFTD patients who received oral leucovorin only, pre-treatment 5-MTHF levels were very low (<10 nmol/L in 17 of 21 patients with observed levels) compared to reported reference ranges from 40 up to 240 nmol/L. A subset of 7 patients had CSF 5-MTHF levels measured both before and after leucovorin initiation. All 7 patients experienced an increase in CSF 5-MTHF levels following treatment initiation, with 5 achieving normalization above 40 nmol/L.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Leucovorin calcium tablets, USP contain:
- 5 mg of leucovorin: White to off-white, round, biconvex tablet; debossed with “ING” above “181” on one side and scoreline on other side and free from any physical defects.
NDC: 50742-181-30: Bottle of 30 Tablets
NDC: 50742-181-01: Bottle of 100 Tablets
- 10 mg of leucovorin: White to off-white, round, biconvex tablet; debossed with “ING” above “182” on one side and scoreline on other side and free from any physical defects.
NDC: 50742-182-12: Bottle of 12 Tablets
NDC: 50742-182-24: Bottle of 24 Tablets
- 15 mg of leucovorin: Peach colored, round, biconvex tablet; debossed with “ING” above “183” on one side and scoreline on other side and free from any physical defects.
NDC: 50742-183-24: Bottle of 24 Tablets
- 25 mg of leucovorin: Peach colored, round, biconvex tablet; debossed with “ING” above “184” on one side and scoreline on other side and free from any physical defects.
NDC: 50742-184-25: Bottle of 25 Tablets
-
17 PATIENT COUNSELING INFORMATION
Administration Instructions
Advise patients or caregivers that leucovorin tablets may be dissolved in an age-appropriate liquid (e.g., water, breastmilk, or infant formula) or crushed and mixed with soft food before administration. If leucovorin is to be administered in this manner, instruct patients or caregivers to administer the dissolved or mixed product immediately after mixing [see Dosage and Administration (2.1)].
Hypersensitivity Reactions
Advise patients to inform their healthcare provider if they develop a hypersensitivity reaction while taking leucovorin calcium tablets [see Warnings and Precautions (5.1)].
Drug Interactions
Advise patients to inform their healthcare providers of all concomitant drugs, including prescription drugs, nonprescription drugs, vitamins, and herbal products [see Drug Interactions (7)].
Manufactured for:
Ingenus Pharmaceuticals, LLC
Orlando, FL 32811
Rx Only
554302
Revised: 03/2026

-
PRINCIPAL DISPLAY PANEL
Package Label - Principal Display Panel – 5 mg, 30's Label
ingenus
NDC: 50742-181-30
Leucovorin Calcium Tablets, USP
5 mg*
Rx Only
30 Tablets

-
PRINCIPAL DISPLAY PANEL
Package Label - Principal Display Panel – 5 mg, 100's Label
ingenus
NDC: 50742-181-01
Leucovorin Calcium Tablets, USP
5 mg*
Rx Only
100 Tablets

-
PRINCIPAL DISPLAY PANEL
Package Label - Principal Display Panel – 10 mg, 12's Label
ingenus
NDC: 50742-182-12
Leucovorin Calcium Tablets, USP
10 mg*
Rx Only
12 Tablets

-
PRINCIPAL DISPLAY PANEL
Package Label - Principal Display Panel – 10 mg, 24's Label
ingenus
NDC: 50742-182-24
Leucovorin Calcium Tablets, USP
10 mg*
Rx Only
24 Tablets

-
PRINCIPAL DISPLAY PANEL
Package Label - Principal Display Panel – 15 mg, 24's Label
ingenus
NDC: 50742-183-24
Leucovorin Calcium Tablets, USP
15 mg*
Rx Only
24 Tablets

-
PRINCIPAL DISPLAY PANEL
Package Label - Principal Display Panel – 25 mg, 25's Label
ingenus
NDC: 50742-184-25
Leucovorin Calcium Tablets, USP
25 mg*
Rx Only
25 Tablets

-
INGREDIENTS AND APPEARANCE
LEUCOVORIN CALCIUM
leucovorin calcium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50742-181 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEUCOVORIN CALCIUM (UNII: RPR1R4C0P4) (LEUCOVORIN - UNII:Q573I9DVLP) LEUCOVORIN 5 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color WHITE (white to off-white) Score 2 pieces Shape ROUND (biconvex) Size 6mm Flavor Imprint Code ING;181 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50742-181-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 07/30/2020 2 NDC: 50742-181-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 07/30/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211132 07/30/2020 LEUCOVORIN CALCIUM
leucovorin calcium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50742-182 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEUCOVORIN CALCIUM (UNII: RPR1R4C0P4) (LEUCOVORIN - UNII:Q573I9DVLP) LEUCOVORIN 10 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color WHITE (white to off-white) Score 2 pieces Shape ROUND (biconvex) Size 8mm Flavor Imprint Code ING;182 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50742-182-12 12 in 1 BOTTLE; Type 0: Not a Combination Product 07/30/2020 2 NDC: 50742-182-24 24 in 1 BOTTLE; Type 0: Not a Combination Product 07/30/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211132 07/30/2020 LEUCOVORIN CALCIUM
leucovorin calcium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50742-183 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEUCOVORIN CALCIUM (UNII: RPR1R4C0P4) (LEUCOVORIN - UNII:Q573I9DVLP) LEUCOVORIN 15 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) Product Characteristics Color YELLOW (peach) Score 2 pieces Shape ROUND (biconvex) Size 6mm Flavor Imprint Code ING;183 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50742-183-24 24 in 1 BOTTLE; Type 0: Not a Combination Product 07/30/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211132 07/30/2020 LEUCOVORIN CALCIUM
leucovorin calcium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50742-184 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEUCOVORIN CALCIUM (UNII: RPR1R4C0P4) (LEUCOVORIN - UNII:Q573I9DVLP) LEUCOVORIN 25 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) Product Characteristics Color YELLOW (peach) Score 2 pieces Shape ROUND (biconvex) Size 8mm Flavor Imprint Code ING;184 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50742-184-25 25 in 1 BOTTLE; Type 0: Not a Combination Product 07/30/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211132 07/30/2020 Labeler - Ingenus Pharmaceuticals, LLC (833250017) Registrant - Ingenus Pharmaceuticals, LLC (833250017)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.