LEVOXYL- levothyroxine sodium tablet
LEVOXYL by
Drug Labeling and Warnings
LEVOXYL by is a Prescription medication manufactured, distributed, or labeled by Pfizer Laboratories Div Pfizer Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LEVOXYL safely and effectively. See full prescribing information for LEVOXYL.
LEVOXYL® (levothyroxine sodium) tablets, for oral use
Initial U.S. Approval: 2001WARNING: NOT FOR TREATMENT OF OBESITY OR FOR WEIGHT LOSS
See full prescribing information for complete boxed warning.
INDICATIONS AND USAGE
LEVOXYL is L-thyroxine (T4) indicated in pediatric and adult patients for:
- Hypothyroidism: As replacement in primary (thyroidal), secondary (pituitary), and tertiary (hypothalamic) congenital or acquired hypothyroidism. (1)
- Pituitary Thyrotropin (Thyroid-Stimulating Hormone, TSH) suppression: As an adjunct to surgery and radioiodine therapy in the management of well-differentiated thyroid cancer. (1)
Limitations of Use:
- - Not indicated for suppression of benign thyroid nodules and nontoxic diffuse goiter in iodine-sufficient patients. (1)
- - Not indicated for treatment of hypothyroidism during the recovery phase of subacute thyroiditis. (1)
DOSAGE AND ADMINISTRATION
- Administer once daily, on an empty stomach, one-half to one hour before breakfast with a full glass of water. (2.1)
- Administer at least 4 hours before or after drugs that are known to interfere with absorption. (2.1)
- Evaluate the need for dose adjustments when regularly administering within one hour of certain foods that may affect absorption. (2.1)
- Starting dose depends on a variety of factors, including age, body weight, cardiovascular status, concomitant medical conditions (including pregnancy), concomitant medications, co-administered food, and the specific nature of the condition being treated. Peak therapeutic effect may not be attained for 4 to 6 weeks. (2.2)
- See full prescribing information for dosing in specific patient populations. (2.3)
- Adequacy of therapy determined with periodic monitoring of TSH and/or T4 as well as clinical status. (2.4)
DOSAGE FORMS AND STRENGTHS
Tablets: 25 mcg, 50 mcg, 75 mcg, 88 mcg, 100 mcg, 112 mcg, 125 mcg, 137 mcg, 150 mcg, 175 mcg, 200 mcg (3)
CONTRAINDICATIONS
Uncorrected adrenal insufficiency. (4)
WARNINGS AND PRECAUTIONS
- Cardiac adverse reactions in the elderly and in patients with underlying cardiovascular disease: Initiate LEVOXYL at less than the full replacement dose because of the increased risk of cardiac adverse reactions, including atrial fibrillation. (2.3, 5.1, 8.5)
- Myxedema coma: Do not use oral thyroid hormone drug products to treat myxedema coma. (5.2)
- Acute adrenal crisis in patients with concomitant adrenal insufficiency: Treat with replacement glucocorticoids prior to initiation of LEVOXYL treatment. (5.3)
- Prevention of hyperthyroidism or incomplete treatment of hypothyroidism: Proper dose titration and careful monitoring is critical to prevent the persistence of hypothyroidism or the development of hyperthyroidism. (5.4)
- Worsening of diabetic control: Therapy in patients with diabetes mellitus may worsen glycemic control and result in increased antidiabetic agent or insulin requirements. Carefully monitor glycemic control after starting, changing, or discontinuing thyroid hormone therapy. (5.5)
- Decreased bone mineral density associated with thyroid hormone over-replacement: Over-replacement can increase bone resorption and decrease bone mineral density. Give the lowest effective dose. (5.6)
ADVERSE REACTIONS
Common adverse reactions for LEVOXYL are primarily those of hyperthyroidism due to therapeutic overdosage: arrhythmias, myocardial infarction, dyspnea, muscle spasm, headache, nervousness, irritability, insomnia, tremors, muscle weakness, increased appetite, weight loss, diarrhea, heat intolerance, menstrual irregularities, and skin rash. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Pfizer, Inc. at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
See full prescribing information for drugs that affect thyroid hormone pharmacokinetics and metabolism (e.g., absorption, synthesis, secretion, catabolism, protein binding, and target tissue response) and may alter the therapeutic response to LEVOXYL. (7)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 12/2018
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: NOT FOR TREATMENT OF OBESITY OR FOR WEIGHT LOSS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 General Administration Information
2.2 General Principles of Dosing
2.3 Dosing in Specific Populations
2.4 Monitoring TSH and/or Thyroxine (T4) Levels
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Cardiac Adverse Reactions in the Elderly and in Patients with Underlying Cardiovascular Disease
5.2 Myxedema Coma
5.3 Acute Adrenal Crisis in Patients with Concomitant Adrenal Insufficiency
5.4 Prevention of Hyperthyroidism or Incomplete Treatment of Hypothyroidism
5.5 Worsening of Diabetic Control
5.6 Decreased Bone Mineral Density Associated with Thyroid Hormone Over-Replacement
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 Drugs Known to Affect Thyroid Hormone Pharmacokinetics
7.2 Antidiabetic Therapy
7.3 Oral Anticoagulants
7.4 Digitalis Glycosides
7.5 Antidepressant Therapy
7.6 Ketamine
7.7 Sympathomimetics
7.8 Tyrosine-Kinase Inhibitors
7.9 Drug-Food Interactions
7.10 Drug-Laboratory Test Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: NOT FOR TREATMENT OF OBESITY OR FOR WEIGHT LOSS
- Thyroid hormones, including LEVOXYL, either alone or with other therapeutic agents, should not be used for the treatment of obesity or for weight loss.
- In euthyroid patients, doses within the range of daily hormonal requirements are ineffective for weight reduction.
- Larger doses may produce serious or even life-threatening manifestations of toxicity, particularly when given in association with sympathomimetic amines such as those used for their anorectic effects [see Adverse Reactions (6), Drug Interactions (7.7), Overdosage (10)].
-
1 INDICATIONS AND USAGE
Hypothyroidism
LEVOXYL is indicated in pediatric and adult patients as a replacement therapy in primary (thyroidal), secondary (pituitary), and tertiary (hypothalamic) congenital or acquired hypothyroidism.
Pituitary Thyrotropin (Thyroid-Stimulating Hormone, TSH) Suppression
LEVOXYL is indicated in pediatric and adult patients as an adjunct to surgery and radioiodine therapy in the management of well-differentiated thyroid cancer.
Limitations of Use:
- LEVOXYL is not indicated for suppression of benign thyroid nodules and nontoxic diffuse goiter in iodine-sufficient patients as there are no clinical benefits and overtreatment with LEVOXYL may induce hyperthyroidism [see Warnings and Precautions (5.4)].
- LEVOXYL is not indicated for treatment of hypothyroidism during the recovery phase of subacute thyroiditis.
-
2 DOSAGE AND ADMINISTRATION
2.1 General Administration Information
Administer LEVOXYL tablets orally as a single daily dose, on an empty stomach, one-half to one hour before breakfast with a full glass of water to avoid choking or gagging [see Adverse Reactions (6)].
Administer LEVOXYL at least 4 hours before or after drugs that are known to interfere with LEVOXYL absorption [see Drug Interactions (7.1)].
Evaluate the need for dose adjustments when regularly administering within one hour of certain foods that may affect LEVOXYL absorption [see Drug Interactions (7.9), Clinical Pharmacology (12.3)].
Administer LEVOXYL to infants and children who cannot swallow intact tablets by crushing the tablet, suspending the freshly crushed tablet in a small amount (5 mL to 10 mL or 1 teaspoon to 2 teaspoons) of water and immediately administering the suspension by spoon or dropper. Do not store the suspension. Do not administer in foods that decrease absorption of LEVOXYL, such as soybean-based infant formula [see Drug Interactions (7.9)].
2.2 General Principles of Dosing
The dose of LEVOXYL for hypothyroidism or pituitary TSH suppression depends on a variety of factors including: the patient's age, body weight, cardiovascular status, concomitant medical conditions (including pregnancy), concomitant medications, co-administered food and the specific nature of the condition being treated [see Dosage and Administration (2.3), Warnings and Precautions (5), Drug Interactions (7)]. Dosing must be individualized to account for these factors and dose adjustments made based on periodic assessment of the patient's clinical response and laboratory parameters [see Dosage and Administration (2.4)].
The peak therapeutic effect of a given dose of LEVOXYL may not be attained for 4 to 6 weeks.
2.3 Dosing in Specific Populations
Primary Hypothyroidism in Adults and in Adolescents in Whom Growth and Puberty Are Complete
Start LEVOXYL at the full replacement dose in otherwise healthy, non-elderly individuals who have been hypothyroid for only a short time (such as a few months). The average full replacement dose of LEVOXYL is approximately 1.6 mcg per kg per day (for example: 100 mcg per day to 125 mcg per day for a 70 kg adult).
Adjust the dose by 12.5 mcg to 25 mcg increments every 4 to 6 weeks until the patient is clinically euthyroid and the serum TSH returns to normal. Doses greater than 200 mcg per day are seldom required. An inadequate response to daily doses of greater than 300 mcg per day is rare and may indicate poor compliance, malabsorption, drug interactions, or a combination of these factors.
For elderly patients or patients with underlying cardiac disease, start with a dose of 12.5 mcg to 25 mcg per day. Increase the dose every 6 to 8 weeks, as needed until the patient is clinically euthyroid and the serum TSH returns to normal. The full replacement dose of LEVOXYL may be less than 1 mcg per kg per day in elderly patients.
In patients with severe longstanding hypothyroidism, start with a dose of 12.5 mcg to 25 mcg per day. Adjust the dose in 12.5 mcg to 25 mcg increments every 2 to 4 weeks until the patient is clinically euthyroid and the serum TSH level is normalized.
Secondary or Tertiary Hypothyroidism
Start LEVOXYL at the full replacement dose in otherwise healthy, non-elderly individuals. Start with a lower dose in elderly patients, patients with underlying cardiovascular disease or patients with severe longstanding hypothyroidism as described above. Serum TSH is not a reliable measure of LEVOXYL dose adequacy in patients with secondary or tertiary hypothyroidism and should not be used to monitor therapy. Use the serum free-T4 level to monitor adequacy of therapy in this patient population. Titrate LEVOXYL dosing per above instructions until the patient is clinically euthyroid and the serum free-T4 level is restored to the upper half of the normal range.
Pediatric Dosage - Congenital or Acquired Hypothyroidism
The recommended daily dose of LEVOXYL in pediatric patients with hypothyroidism is based on body weight and changes with age as described in Table 1. Start LEVOXYL at the full daily dose in most pediatric patients. Start at a lower starting dose in newborns (0 to 3 months) at risk for cardiac failure and in children at risk for hyperactivity (see below). Monitor for clinical and laboratory response [see Dosage and Administration (2.4)].
Table 1: LEVOXYL Dosing Guidelines for Pediatric Hypothyroidism Age Daily Dose Per Kg Body Weight* - * - The dose should be adjusted based on clinical response and laboratory parameters [see Warnings and Precautions (5.4), Drug Interactions (7.10), Use in Specific Populations (8.4)].
0 to 3 months 10 mcg/kg daily to 15 mcg/kg daily 3 to 6 months 8 mcg/kg daily to 10 mcg/kg daily 6 to 12 months 6 mcg/kg daily to 8 mcg/kg daily 1 to 5 years 5 mcg/kg daily to 6 mcg/kg daily 6 to 12 years 4 mcg/kg daily to 5 mcg/kg daily Greater than 12 years but growth and puberty incomplete 2 mcg/kg daily to 3 mcg/kg daily Growth and puberty complete 1.6 mcg/kg daily Newborns (0 to 3 months) at Risk for Cardiac Failure:
Consider a lower starting dose in newborns at risk for cardiac failure. Increase the dose every 4 to 6 weeks as needed based on clinical and laboratory response.
Pediatric Patients at Risk for Hyperactivity:
To minimize the risk of hyperactivity in pediatric patients, start at one-fourth the recommended full replacement dose, and increase on a weekly basis by one-fourth the full recommended replacement dose until the full recommended replacement dose is reached.
Pregnancy
Pre-existing Hypothyroidism: LEVOXYL dose requirements may increase during pregnancy. Measure serum TSH and free-T4 as soon as pregnancy is confirmed and, at minimum, during each trimester of pregnancy. In patients with primary hypothyroidism, maintain serum TSH in the trimester-specific reference range. For patients with serum TSH above the normal trimester-specific range, increase the dose of LEVOXYL by 12.5 mcg daily to 25 mcg daily and measure TSH every 4 weeks until a stable LEVOXYL dose is reached and serum TSH is within the normal trimester-specific range. Reduce LEVOXYL dosage to pre-pregnancy levels immediately after delivery and measure serum TSH levels 4 to 8 weeks postpartum to ensure LEVOXYL dose is appropriate.
New Onset Hypothyroidism: Normalize thyroid function as rapidly as possible. In patients with moderate to severe signs and symptoms of hypothyroidism, start LEVOXYL at the full replacement dose (1.6 mcg per kg body weight per day). In patients with mild hypothyroidism (TSH less than 10 mIU per liter) start LEVOXYL at 1 mcg per kg body weight per day. Evaluate serum TSH every 4 weeks and adjust LEVOXYL dosage until a serum TSH is within the normal trimester-specific range [see Use in Specific Populations (8.1)].
2.4 Monitoring TSH and/or Thyroxine (T4) Levels
Assess the adequacy of therapy by periodic assessment of laboratory tests and clinical evaluation. Persistent clinical and laboratory evidence of hypothyroidism despite an apparent adequate replacement dose of LEVOXYL may be evidence of inadequate absorption, poor compliance, drug interactions, or a combination of these factors.
Adults
In adult patients with primary hypothyroidism, monitor serum TSH levels after an interval of 6 to 8 weeks after any change in dose. In patients on a stable and appropriate replacement dose, evaluate clinical and biochemical response every 6 to 12 months and whenever there is a change in the patient's clinical status.
Pediatrics
In patients with congenital hypothyroidism, assess the adequacy of replacement therapy by measuring both serum TSH and total or free-T4. Monitor TSH and total or free-T4 in children as follows: 2 and 4 weeks after the initiation of treatment, 2 weeks after any change in dosage, and then every 3 to 12 months thereafter following dose stabilization until growth is completed. Poor compliance or abnormal values may necessitate more frequent monitoring. Perform routine clinical examination, including assessment of development, mental and physical growth, and bone maturation, at regular intervals.
While the general aim of therapy is to normalize the serum TSH level, TSH may not normalize in some patients due to in utero hypothyroidism causing a resetting of pituitary-thyroid feedback. Failure of the serum T4 to increase into the upper half of the normal range within 2 weeks of initiation of LEVOXYL therapy and/or of the serum TSH to decrease below 20 mIU per liter within 4 weeks may indicate the child is not receiving adequate therapy. Assess compliance, dose of medication administered, and method of administration prior to increasing the dose of LEVOXYL [see Warnings and Precautions (5.4), Use in Specific Populations (8.4)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Levothyroxine is contraindicated in patients with uncorrected adrenal insufficiency [see Warnings and Precautions (5.3)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Cardiac Adverse Reactions in the Elderly and in Patients with Underlying Cardiovascular Disease
Overtreatment with levothyroxine may cause an increase in heart rate, cardiac wall thickness, and cardiac contractility and may precipitate angina or arrhythmias, particularly in patients with cardiovascular disease and in elderly patients. Initiate LEVOXYL therapy in this population at lower doses than those recommended in younger individuals or in patients without cardiac disease [see Dosage and Administration (2.3), Use in Specific Populations (8.5)].
Monitor for cardiac arrhythmias during surgical procedures in patients with coronary artery disease receiving suppressive LEVOXYL therapy. Monitor patients receiving concomitant LEVOXYL and sympathomimetic agents for signs and symptoms of coronary insufficiency. If cardiovascular symptoms develop or worsen, reduce or withhold the LEVOXYL dose for one week and restart at a lower dose.
5.2 Myxedema Coma
Myxedema coma is a life-threatening emergency characterized by poor circulation and hypometabolism, and may result in unpredictable absorption of levothyroxine sodium from the gastrointestinal tract. Use of oral thyroid hormone drug products is not recommended to treat myxedema coma. Administer thyroid hormone products formulated for intravenous administration to treat myxedema coma.
5.3 Acute Adrenal Crisis in Patients with Concomitant Adrenal Insufficiency
Thyroid hormone increases metabolic clearance of glucocorticoids. Initiation of thyroid hormone therapy prior to initiating glucocorticoid therapy may precipitate an acute adrenal crisis in patients with adrenal insufficiency. Treat patients with adrenal insufficiency with replacement glucocorticoids prior to initiating treatment with LEVOXYL [see Contraindications (4)].
5.4 Prevention of Hyperthyroidism or Incomplete Treatment of Hypothyroidism
LEVOXYL has a narrow therapeutic index. Over- or undertreatment with LEVOXYL may have negative effects on growth and development, cardiovascular function, bone metabolism, reproductive function, cognitive function, emotional state, gastrointestinal function, and on glucose and lipid metabolism. Titrate the dose of LEVOXYL carefully and monitor response to titration to avoid these effects [see Dosage and Administration (2.4)]. Monitor for the presence of drug or food interactions when using LEVOXYL and adjust the dose as necessary [see Drug Interactions (7), Clinical Pharmacology (12.3)].
5.5 Worsening of Diabetic Control
Addition of levothyroxine therapy in patients with diabetes mellitus may worsen glycemic control and result in increased antidiabetic agent or insulin requirements. Carefully monitor glycemic control after starting, changing, or discontinuing LEVOXYL [see Drug Interactions (7.2)].
5.6 Decreased Bone Mineral Density Associated with Thyroid Hormone Over-Replacement
Increased bone resorption and decreased bone mineral density may occur as a result of levothyroxine over-replacement, particularly in post-menopausal women. The increased bone resorption may be associated with increased serum levels and urinary excretion of calcium and phosphorous, elevations in bone alkaline phosphatase, and suppressed serum parathyroid hormone levels. Administer the minimum dose of LEVOXYL that achieves the desired clinical and biochemical response to mitigate against this risk.
-
6 ADVERSE REACTIONS
Common adverse reactions with LEVOXYL therapy are primarily those of hyperthyroidism due to therapeutic overdosage [see Warnings and Precautions (5.4), Overdosage (10)]. They include the following:
General: fatigue, increased appetite, weight loss, heat intolerance, fever, excessive sweating
Central nervous system: headache, hyperactivity, nervousness, anxiety, irritability, emotional lability, insomnia
Musculoskeletal: tremors, muscle weakness and cramps
Cardiovascular: palpitations, tachycardia, arrhythmias, increased pulse and blood pressure, heart failure, angina, myocardial infarction, cardiac arrest
Respiratory: dyspnea
Gastrointestinal: diarrhea, vomiting, abdominal cramps, elevations in liver function tests
Dermatologic: hair loss, flushing
Endocrine: decreased bone mineral density
Reproductive: menstrual irregularities, impaired fertility
Seizures have been reported rarely with levothyroxine therapy.
Adverse Reactions in Pediatric Patients
Pseudotumor cerebri and slipped capital femoral epiphysis have been reported in pediatric patients receiving levothyroxine therapy. Overtreatment may result in craniosynostosis in infants and premature closure of the epiphysis in pediatric patients with resultant compromised adult height.
Hypersensitivity Reactions
Hypersensitivity reactions to inactive ingredients have occurred in patients treated with thyroid hormone products. These include urticaria, pruritus, skin rash, flushing, angioedema, various gastrointestinal symptoms (abdominal pain, nausea, vomiting, and diarrhea), fever, arthralgia, serum sickness and wheezing. Hypersensitivity to levothyroxine itself is not known to occur.
Choking and Gagging on LEVOXYL Tablets
There have been reports of choking, gagging, tablet stuck in throat, and dysphagia with LEVOXYL tablets, predominately when LEVOXYL tablets were not taken with water [see Dosage and Administration (2.1)].
-
7 DRUG INTERACTIONS
7.1 Drugs Known to Affect Thyroid Hormone Pharmacokinetics
Many drugs can exert effects on thyroid hormone pharmacokinetics (e.g., absorption, synthesis, secretion, catabolism, protein binding, and target tissue response) and may alter the therapeutic response to LEVOXYL (see Tables 2 – 5).
Table 2: Drugs That May Decrease T4 Absorption (Hypothyroidism) Potential impact: Concurrent use may reduce the efficacy of LEVOXYL by binding and delaying or preventing absorption, potentially resulting in hypothyroidism. Drug or Drug Class Effect Calcium Carbonate
Ferrous SulfateCalcium carbonate may form an insoluble chelate with levothyroxine, and ferrous sulfate likely forms a ferric-thyroxine complex. Administer LEVOXYL at least 4 hours apart from these agents. Orlistat Monitor patients treated concomitantly with orlistat and LEVOXYL for changes in thyroid function. Bile Acid Sequestrants
-Colesevelam
-Cholestyramine
-Colestipol
Ion Exchange Resins
-Kayexalate
-SevelamerBile acid sequestrants and ion exchange resins are known to decrease levothyroxine absorption. Administer LEVOXYL at least 4 hours prior to these drugs or monitor thyroid-stimulating hormone (TSH) levels. Other drugs:
Proton Pump Inhibitors
Sucralfate
Antacids
- Aluminum & Magnesium Hydroxides
- SimethiconeGastric acidity is an essential requirement for adequate absorption of levothyroxine. Sucralfate, antacids and proton pump inhibitors may cause hypochlorhydria, affect intragastric pH, and reduce levothyroxine absorption. Monitor patients appropriately. Table 3: Drugs That May Alter Thyroxine (T4) and Triiodothyronine (T3) Serum Transport Without Affecting Free Thyroxine (FT4) Concentration (Euthyroidism) Drug or Drug Class Effect Clofibrate
Estrogen-containing Oral Contraceptives
Estrogens (oral)
Heroin/Methadone
5-Fluorouracil
Mitotane
TamoxifenThese drugs may increase serum thyroxine-binding globulin (TBG) concentration. Androgens / Anabolic Steroids
Asparaginase
Glucocorticoids
Slow-Release Nicotinic AcidThese drugs may decrease serum TBG concentration. Potential impact (below): Administration of these agents with LEVOXYL results in an initial transient increase in FT4. Continued administration results in a decrease in serum T4 and normal FT4 and TSH concentrations. Salicylates (>2 g/day) Salicylates inhibit binding of T4 and T3 to TBG and transthyretin. An initial increase in serum FT4 is followed by return of FT4 to normal levels with sustained therapeutic serum salicylate concentrations, although total T4 levels may decrease by as much as 30%. Other drugs:
Carbamazepine
Furosemide (>80 mg IV)
Heparin
Hydantoins
Non-Steroidal Anti-inflammatory Drugs - FenamatesThese drugs may cause protein binding site displacement. Furosemide has been shown to inhibit the protein binding of T4 to TBG and albumin, causing an increased free-T4 fraction in serum. Furosemide competes for T4-binding sites on TBG, prealbumin, and albumin, so that a single high dose can acutely lower the total T4 level. Phenytoin and carbamazepine reduce serum protein binding of levothyroxine, and total and FT4 may be reduced by 20% to 40%, but most patients have normal serum TSH levels and are clinically euthyroid. Closely monitor thyroid hormone parameters. Table 4: Drugs That May Alter Hepatic Metabolism of T4 (Hypothyroidism) Potential impact: Stimulation of hepatic microsomal drug-metabolizing enzyme activity may cause increased hepatic degradation of levothyroxine, resulting in increased LEVOXYL requirements. Drug or Drug Class Effect Phenobarbital
RifampinPhenobarbital has been shown to reduce the response to thyroxine. Phenobarbital increases L-thyroxine metabolism by inducing uridine 5'-diphospho-glucuronosyltransferase (UGT) and leads to a lower T4 serum levels. Changes in thyroid status may occur if barbiturates are added or withdrawn from patients being treated for hypothyroidism. Rifampin has been shown to accelerate the metabolism of levothyroxine. Table 5: Drugs That May Decrease Conversion of T4 to T3 Potential impact: Administration of these enzyme inhibitors decreases the peripheral conversion of T4 to T3, leading to decreased T3 levels. However, serum T4 levels are usually normal but may occasionally be slightly increased. Drug or Drug Class Effect Beta-adrenergic antagonists
(e.g., Propranolol >160 mg/day)In patients treated with large doses of propranolol (>160 mg/day), T3 and T4 levels change slightly, TSH levels remain normal, and patients are clinically euthyroid. It should be noted that actions of particular beta-adrenergic antagonists may be impaired when the hypothyroid patient is converted to the euthyroid state. Glucocorticoids (e.g., Dexamethasone ≥4 mg/day) Short-term administration of large doses of glucocorticoids may decrease serum T3 concentrations by 30% with minimal change in serum T4 levels. However, long-term glucocorticoid therapy may result in slightly decreased T3 and T4 levels due to decreased TBG production (see above). Other:
Amiodarone)Amiodarone inhibits peripheral conversion of levothyroxine (T4) to triiodothyronine (T3) and may cause isolated biochemical changes (increase in serum free-T4, and decreased or normal free-T3) in clinically euthyroid patients. 7.2 Antidiabetic Therapy
Addition of LEVOXYL therapy in patients with diabetes mellitus may worsen glycemic control and result in increased antidiabetic agent or insulin requirements. Carefully monitor glycemic control, especially when LEVOXYL is started, changed, or discontinued [see Warnings and Precautions (5.5)].
7.3 Oral Anticoagulants
LEVOXYL increases the response to oral anticoagulant therapy. Therefore, a decrease in the dose of anticoagulant may be warranted with correction of the hypothyroid state or when the LEVOXYL dose is increased. Closely monitor coagulation tests to permit appropriate and timely dosage adjustments.
7.4 Digitalis Glycosides
LEVOXYL may reduce the therapeutic effects of digitalis glycosides. Serum digitalis glycoside levels may be decreased when a hypothyroid patient becomes euthyroid, necessitating an increase in the dose of digitalis glycosides.
7.5 Antidepressant Therapy
Concurrent use of tricyclic (e.g., amitriptyline) or tetracyclic (e.g., maprotiline) antidepressants and LEVOXYL may increase the therapeutic and toxic effects of both drugs, possibly due to increased receptor sensitivity to catecholamines. Toxic effects may include increased risk of cardiac arrhythmias and central nervous system stimulation. LEVOXYL may accelerate the onset of action of tricyclics. Administration of sertraline in patients stabilized on LEVOXYL may result in increased LEVOXYL requirements.
7.6 Ketamine
Concurrent use of ketamine and LEVOXYL may produce marked hypertension and tachycardia. Closely monitor blood pressure and heart rate in these patients.
7.7 Sympathomimetics
Concurrent use of sympathomimetics and LEVOXYL may increase the effects of sympathomimetics or thyroid hormone. Thyroid hormones may increase the risk of coronary insufficiency when sympathomimetic agents are administered to patients with coronary artery disease.
7.8 Tyrosine-Kinase Inhibitors
Concurrent use of tyrosine-kinase inhibitors such as imatinib may cause hypothyroidism. Closely monitor TSH levels in such patients.
7.9 Drug-Food Interactions
Consumption of certain foods may affect LEVOXYL absorption thereby necessitating adjustments in dosing [see Dosage and Administration (2.1)]. Soybean flour (infant formula), cotton seed meal, walnuts, and dietary fiber may bind and decrease the absorption of LEVOXYL from the GI tract. Grapefruit juice may delay the absorption of levothyroxine and reduce its bioavailability.
7.10 Drug-Laboratory Test Interactions
Consider changes in TBG concentration when interpreting T4 and T3 values. Measure and evaluate unbound (free) hormone and/or determine the free T4 index (FT4I) in this circumstance. Pregnancy, infectious hepatitis, estrogens, estrogen-containing oral contraceptives, and acute intermittent porphyria increase TBG concentrations. Nephrosis, severe hypoproteinemia, severe liver disease, acromegaly, androgens, and corticosteroids decrease TBG concentration. Familial hyper- or hypo-thyroxine-binding globulinemias have been described, with the incidence of TBG deficiency approximating 1 in 9000.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Experience with levothyroxine use in pregnant women, including data from post-marketing studies, have not reported increased rates of major birth defects or miscarriages (see Data). There are risks to the mother and fetus associated with untreated hypothyroidism in pregnancy. Since thyroid-stimulating hormone (TSH) levels may increase during pregnancy, TSH should be monitored and LEVOXYL dosage adjusted during pregnancy (see Clinical Considerations). There are no animal studies conducted with levothyroxine during pregnancy. LEVOXYL should not be discontinued during pregnancy and hypothyroidism diagnosed during pregnancy should be promptly treated.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Maternal hypothyroidism during pregnancy is associated with a higher rate of complications, including spontaneous abortion, gestational hypertension, pre-eclampsia, stillbirth, and premature delivery. Untreated maternal hypothyroidism may have an adverse effect on fetal neurocognitive development.
Dose Adjustments During Pregnancy and the Postpartum Period
Pregnancy may increase LEVOXYL requirements. Serum TSH level should be monitored and the LEVOXYL dosage adjusted during pregnancy. Since postpartum TSH levels are similar to preconception values, the LEVOXYL dosage should return to the pre-pregnancy dose immediately after delivery [see Dosage and Administration (2.3)].
Data
Human Data
Levothyroxine is approved for use as a replacement therapy for hypothyroidism. There is a long experience of levothyroxine use in pregnant women, including data from post-marketing studies that have not reported increased rates of fetal malformations, miscarriages or other adverse maternal or fetal outcomes associated with levothyroxine use in pregnant women.
8.2 Lactation
Risk Summary
Limited published studies report that levothyroxine is present in human milk. However, there is insufficient information to determine the effects of levothyroxine on the breastfed infant and no available information on the effects of levothyroxine on milk production. Adequate levothyroxine treatment during lactation may normalize milk production in hypothyroid lactating mothers. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for LEVOXYL and any potential adverse effects on the breastfed infant from LEVOXYL or from the underlying maternal condition.
8.4 Pediatric Use
The initial dose of LEVOXYL varies with age and body weight. Dosing adjustments are based on an assessment of the individual patient's clinical and laboratory parameters [see Dosage and Administration (2.3, 2.4)].
In children in whom a diagnosis of permanent hypothyroidism has not been established, discontinue LEVOXYL for a trial period, but only after the child is at least 3 years of age. Obtain serum T4 and TSH levels at the end of the trial period, and use laboratory test results and clinical assessments to guide diagnosis and treatment, if warranted.
Congenital Hypothyroidism [see Dosage and Administration (2.3, 2.4)]
Rapid restoration of normal serum T4 concentrations is essential for preventing the adverse effects of congenital hypothyroidism on intellectual development as well as on overall physical growth and maturation. Therefore, initiate LEVOXYL therapy immediately upon diagnosis. Levothyroxine is generally continued for life in these patients.
Closely monitor infants during the first 2 weeks of LEVOXYL therapy for cardiac overload, arrhythmias, and aspiration from avid suckling.
Closely monitor patients to avoid undertreatment or overtreatment. Undertreatment may have deleterious effects on intellectual development and linear growth. Overtreatment is associated with craniosynostosis in infants, may adversely affect the tempo of brain maturation, and may accelerate the bone age and result in premature epiphyseal closure and compromised adult stature.
Acquired Hypothyroidism in Pediatric Patients
Closely monitor patients to avoid undertreatment and overtreatment. Undertreatment may result in poor school performance due to impaired concentration and slowed mentation and in reduced adult height. Overtreatment may accelerate the bone age and result in premature epiphyseal closure and compromised adult stature.
Treated children may manifest a period of catch-up growth, which may be adequate in some cases to normalize adult height. In children with severe or prolonged hypothyroidism, catch-up growth may not be adequate to normalize adult height.
8.5 Geriatric Use
Because of the increased prevalence of cardiovascular disease among the elderly, initiate LEVOXYL at less than the full replacement dose [see Dosage and Administration (2.3), Warnings and Precautions (5.1)]. Atrial arrhythmias can occur in elderly patients. Atrial fibrillation is the most common of the arrhythmias observed with levothyroxine overtreatment in the elderly.
-
10 OVERDOSAGE
The signs and symptoms of overdosage are those of hyperthyroidism [see Warnings and Precautions (5.4), Adverse Reactions (6)]. In addition, confusion and disorientation may occur. Cerebral embolism, shock, coma, and death have been reported. Seizures occurred in a 3-year-old child ingesting 3.6 mg of levothyroxine. Symptoms may not necessarily be evident or may not appear until several days after ingestion of levothyroxine sodium.
Reduce the LEVOXYL dose or temporarily discontinued if signs or symptoms of overdosage occur. Initiate appropriate supportive treatment as dictated by the patient's medical status.
For current information on the management of poisoning or overdosage, contact the National Poison Control Center at 1-800-222-1222 or www.poison.org.
-
11 DESCRIPTION
LEVOXYL contains the active ingredient, levothyroxine, asynthetic crystalline levothyroxine (T4) in sodium salt form. It is chemically designated as L-3,3',5,5'-tetraiodothyronine monosodium hydrate. Synthetic T4 is identical in chemical structure to the T4 produced in the human thyroid gland. Levothyroxine sodium has an empirical formula of C15H10I4N NaO4 ∙ H2O, molecular weight of 798.85 g/mol (anhydrous), and structural formula as shown:
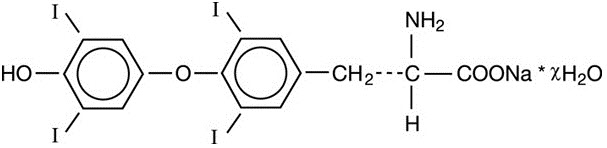
LEVOXYL tablets for oral administration are supplied in the following strengths: 25 mcg, 50 mcg, 75 mcg, 88 mcg, 100 mcg, 112 mcg, 125 mcg, 137 mcg, 150 mcg, 175 mcg, and 200 mcg.
Inactive Ingredients
Calcium sulfate dihydrate, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and sodium bicarbonate. The following are the coloring additives per tablet strength:
Strength
(mcg)Color additive(s) 25 FD&C Yellow No. 6 Aluminum Lake 50 None 75 FD&C Blue No. 1 Aluminum Lake, D&C Red No. 30 Aluminum Lake 88 FD&C Yellow No. 6 Aluminum Lake, FD&C Blue No. 1 Aluminum Lake, D&C Yellow No. 10 Aluminum Lake 100 FD&C Yellow No. 6 Aluminum Lake, D&C Yellow No. 10 Aluminum Lake 112 FD&C Yellow No. 6 Aluminum Lake, FD&C Red No. 40 Aluminum Lake, D&C Red No. 30 Aluminum Lake 125 FD&C Red No. 40 Aluminum Lake, D&C Yellow No. 10 Aluminum Lake 137 FD&C Blue No. 1 Aluminum Lake 150 FD&C Blue No. 1 Aluminum Lake, D&C Red No. 30 Aluminum Lake 175 FD&C Blue No. 1 Aluminum Lake, D&C Yellow No. 10 Aluminum Lake 200 D&C Red No. 30 Aluminum Lake, D&C Yellow No. 10 Aluminum Lake -
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Thyroid hormones exert their physiologic actions through control of DNA transcription and protein synthesis. Triiodothyronine (T3) and L-thyroxine (T4) diffuse into the cell nucleus and bind to thyroid receptor proteins attached to DNA. This hormone nuclear receptor complex activates gene transcription and synthesis of messenger RNA and cytoplasmic proteins.
The physiological actions of thyroid hormones are produced predominantly by T3, the majority of which (approximately 80%) is derived from T4 by deiodination in peripheral tissues.
12.2 Pharmacodynamics
Oral levothyroxine sodium is a synthetic T4 hormone that exerts the same physiologic effect as endogenous T4, thereby maintaining normal T4 levels when a deficiency is present.
12.3 Pharmacokinetics
Absorption
Absorption of orally administered T4 from the gastrointestinal tract ranges from 40% to 80%. The majority of the levothyroxine dose is absorbed from the jejunum and upper ileum. The relative bioavailability of LEVOXYL tablets, compared to an equal nominal dose of oral levothyroxine sodium solution, is approximately 95% to 98%. T4 absorption is increased by fasting, and decreased in malabsorption syndromes and by certain foods such as soybeans. Dietary fiber decreases bioavailability of T4. Absorption may also decrease with age. In addition, many drugs and foods affect T4 absorption [see Drug Interactions (7)].
Distribution
Circulating thyroid hormones are greater than 99% bound to plasma proteins, including thyroxine-binding globulin (TBG), thyroxine-binding prealbumin (TBPA), and thyroxine-binding albumin (TBA), whose capacities and affinities vary for each hormone. The higher affinity of both TBG and TBPA for T4 partially explains the higher serum levels, slower metabolic clearance, and longer half-life of T4 compared to T3. Protein-bound thyroid hormones exist in reverse equilibrium with small amounts of free hormone. Only unbound hormone is metabolically active. Many drugs and physiologic conditions affect the binding of thyroid hormones to serum proteins [see Drug Interactions (7)]. Thyroid hormones do not readily cross the placental barrier [see Use in Specific Populations (8.1)].
Elimination
Metabolism
T4 is slowly eliminated (Table 7). The major pathway of thyroid hormone metabolism is through sequential deiodination. Approximately 80 % of circulating T3 is derived from peripheral T4 by monodeiodination. The liver is the major site of degradation for both T4 and T3, with T4 deiodination also occurring at a number of additional sites, including the kidney and other tissues. Approximately 80% of the daily dose of T4 is deiodinated to yield equal amounts of T3 and reverse T3 (rT3). T3 and rT3 are further deiodinated to diiodothyronine. Thyroid hormones are also metabolized via conjugation with glucuronides and sulfates and excreted directly into the bile and gut where they undergo enterohepatic recirculation.
Excretion
Thyroid hormones are primarily eliminated by the kidneys. A portion of the conjugated hormone reaches the colon unchanged and is eliminated in the feces. Approximately 20% of T4 is eliminated in the stool. Urinary excretion of T4 decreases with age.
Table 7: Pharmacokinetic Parameters of Thyroid Hormones in Euthyroid Patients Hormone Ratio in Thyroglobulin Biologic Potency t½
(days)Protein Binding
(%)*- * Includes TBG, TBPA, and TBA
- † 3 to 4 days in hyperthyroidism, 9 to 10 days in hypothyroidism
Levothyroxine (T4) 10–20 1 6–7† 99.96 Liothyronine (T3) 1 4 ≤2 99.5 - 13 NONCLINICAL TOXICOLOGY
-
16 HOW SUPPLIED/STORAGE AND HANDLING
LEVOXYL (levothyroxine sodium) tablets are oval, color-coded and, potency marked available as follows:
Strength
(mcg)Color Tablet Markings NDC - bottles of 100 NDC - bottles of 1000 25 Orange 25 60793-850-01 60793-850-10 50 White 50 60793-851-01 60793-851-10 75 Purple 75 60793-852-01 60793-852-10 88 Olive 88 60793-853-01 60793-853-10 100 Yellow 100 60793-854-01 60793-854-10 112 Rose 112 60793-855-01 60793-855-10 125 Light Brown 125 60793-856-01 60793-856-10 137 Dark Blue 137 60793-857-01 60793-857-10 150 Blue 150 60793-858-01 60793-858-10 175 Turquoise 175 60793-859-01 60793-859-10 200 Pink 200 60793-860-01 60793-860-10 -
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Dosing and Administration
- Instruct patients to take LEVOXYL only as directed by their healthcare provider.
- Instruct patients to take LEVOXYL as a single dose, preferably on an empty stomach, one-half to one hour before breakfast with a full glass of water to avoid choking or gagging.
- Inform patients that agents such as iron and calcium supplements and antacids can decrease the absorption of levothyroxine. Instruct patients not to take LEVOXYL tablets within 4 hours of these agents.
- Instruct patients to notify their healthcare provider should they become pregnant or breastfeeding or are thinking of becoming pregnant, while taking LEVOXYL.
Important Information
- Inform patients that it may take several weeks before they notice an improvement in symptoms.
- Inform patients that the levothyroxine in LEVOXYL is intended to replace a hormone that is normally produced by the thyroid gland. Generally, replacement therapy is to be taken for life.
- Inform patients that LEVOXYL should not be used as a primary or adjunctive therapy in a weight control program.
- Instruct patients to notify their healthcare provider if they are taking any other medications, including prescription and over-the-counter preparations.
- Instruct patients to notify their healthcare provider of any other medical conditions you may have, particularly heart disease, diabetes, clotting disorders, and adrenal or pituitary gland problems as the dose of medications used to control these other conditions may need to be adjusted while taking LEVOXYL. If they have diabetes, instruct patients to monitor their blood and/or urinary glucose levels as directed by their physician and immediately report any changes to their physician. If patients are taking anticoagulants, their clotting status should be checked frequently.
- Instruct patients to notify their physician or dentist if they are taking LEVOXYL prior to any surgery.
Adverse Reactions
- Instruct patients to notify their healthcare provider if they experience any of the following symptoms: rapid or irregular heartbeat, chest pain, shortness of breath, leg cramps, headache, nervousness, irritability, sleeplessness, tremors, change in appetite, weight gain or loss, vomiting, diarrhea, excessive sweating, heat intolerance, fever, changes in menstrual periods, hives or skin rash, or any other unusual medical event.
- Inform patients that partial hair loss may occur rarely during the first few months of LEVOXYL therapy; this is usually temporary.
- SPL UNCLASSIFIED SECTION
-
PATIENT PACKAGE INSERT
-
PRINCIPAL DISPLAY PANEL - 25 mcg Tablet Bottle Label
NDC: 60793-850-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets25 mcg
100 Tablets
Rx only
-
PRINCIPAL DISPLAY PANEL - 50 mcg Tablet Bottle Label
NDC: 60793-851-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets50 mcg
100 Tablets
Rx only
-
PRINCIPAL DISPLAY PANEL - 75 mcg Tablet Bottle Label
NDC: 60793-852-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets75 mcg
100 Tablets
Rx only
-
PRINCIPAL DISPLAY PANEL - 88 mcg Tablet Bottle Label
NDC: 60793-853-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets88 mcg
100 Tablets
Rx only
-
PRINCIPAL DISPLAY PANEL - 100 mcg Tablet Bottle Label
NDC: 60793-854-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets100 mcg
100 Tablets
Rx only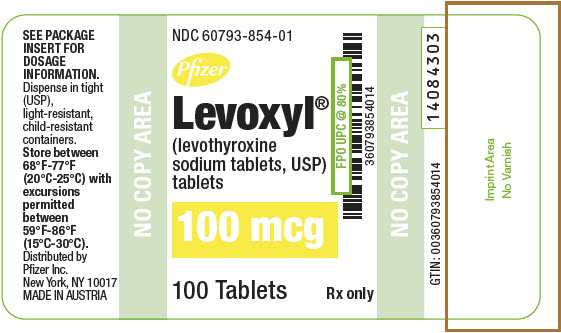
-
PRINCIPAL DISPLAY PANEL - 112 mcg Tablet Bottle Label
NDC: 60793-855-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets112 mcg
100 Tablets
Rx only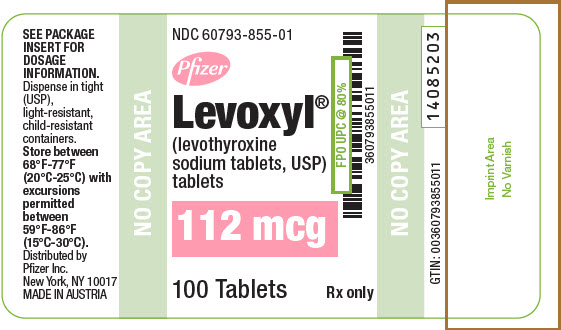
-
PRINCIPAL DISPLAY PANEL - 125 mcg Tablet Bottle Label
NDC: 60793-856-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets125 mcg
100 Tablets
Rx only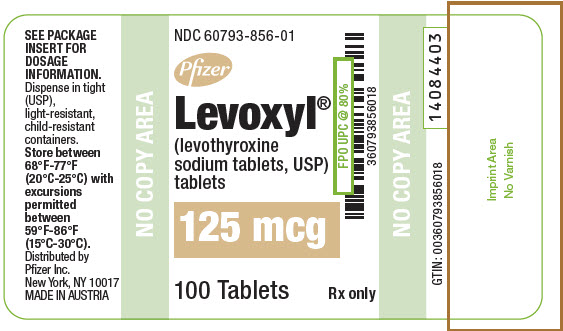
-
PRINCIPAL DISPLAY PANEL - 137 mcg Tablet Bottle Label
NDC: 60793-857-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets137 mcg
100 Tablets
Rx only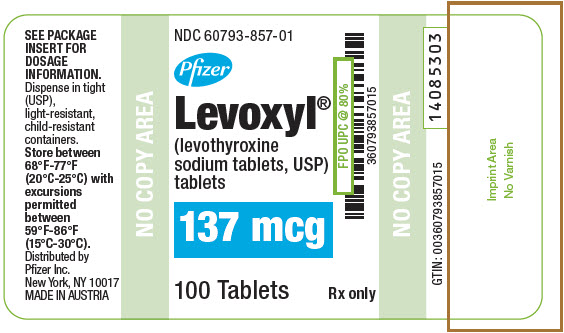
-
PRINCIPAL DISPLAY PANEL - 150 mcg Tablet Bottle Label
NDC: 60793-858-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets150 mcg
100 Tablets
Rx only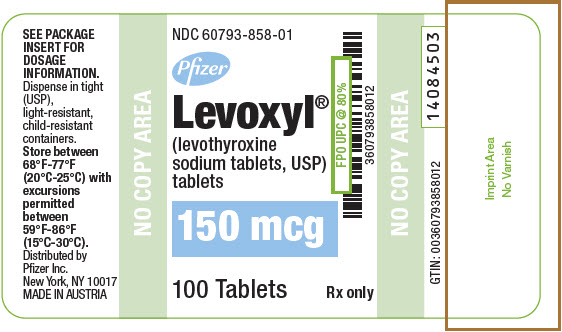
-
PRINCIPAL DISPLAY PANEL - 175 mcg Tablet Bottle Label
NDC: 60793-859-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets175 mcg
100 Tablets
Rx only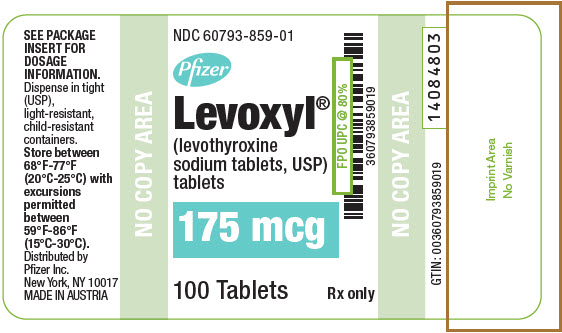
-
PRINCIPAL DISPLAY PANEL - 200 mcg Tablet Bottle Label
NDC: 60793-860-01
Pfizer
Levoxyl®
(levothyroxine
sodium tablets, USP)
tablets200 mcg
100 Tablets
Rx only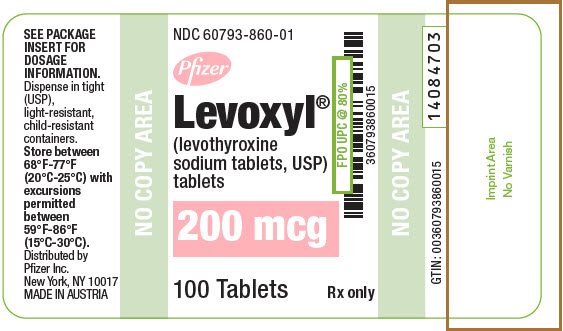
-
INGREDIENTS AND APPEARANCE
LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-850 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 25 ug Inactive Ingredients Ingredient Name Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) Product Characteristics Color ORANGE Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;25 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-850-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-850-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-851 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 50 ug Inactive Ingredients Ingredient Name Strength CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) SODIUM BICARBONATE (UNII: 8MDF5V39QO) Product Characteristics Color WHITE Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;50 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-851-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-851-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-852 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 75 ug Inactive Ingredients Ingredient Name Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) Product Characteristics Color PURPLE Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;75 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-852-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-852-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-853 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 88 ug Inactive Ingredients Ingredient Name Strength CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) SODIUM BICARBONATE (UNII: 8MDF5V39QO) Product Characteristics Color GREEN (Olive) Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;88 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-853-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-853-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-854 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 100 ug Inactive Ingredients Ingredient Name Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) Product Characteristics Color YELLOW Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;100 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-854-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-854-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-855 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 112 ug Inactive Ingredients Ingredient Name Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) Product Characteristics Color PINK (ROSE) Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;112 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-855-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-855-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-856 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 125 ug Inactive Ingredients Ingredient Name Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) Product Characteristics Color BROWN Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;125 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-856-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-856-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-857 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 137 ug Inactive Ingredients Ingredient Name Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) Product Characteristics Color BLUE (DARK BLUE) Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;137 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-857-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-857-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-858 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 150 ug Inactive Ingredients Ingredient Name Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) Product Characteristics Color BLUE Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;150 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-858-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-858-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-859 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 175 ug Inactive Ingredients Ingredient Name Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) Product Characteristics Color TURQUOISE Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;175 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-859-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-859-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 LEVOXYL
levothyroxine sodium tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60793-860 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM ANHYDROUS 200 ug Inactive Ingredients Ingredient Name Strength SODIUM BICARBONATE (UNII: 8MDF5V39QO) CALCIUM SULFATE DIHYDRATE (UNII: 4846Q921YM) Product Characteristics Color PINK Score 2 pieces Shape OVAL Size 10mm Flavor Imprint Code Levoxyl;dp;200 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60793-860-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 2 NDC: 60793-860-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021301 05/25/2001 Labeler - Pfizer Laboratories Div Pfizer Inc (134489525) Establishment Name Address ID/FEI Business Operations UPM Pharmaceuticals 032125469 ANALYSIS(60793-850, 60793-851, 60793-852, 60793-853, 60793-854, 60793-855, 60793-856, 60793-857, 60793-858, 60793-859, 60793-860) , LABEL(60793-850, 60793-851, 60793-852, 60793-853, 60793-854, 60793-855, 60793-856, 60793-857, 60793-858, 60793-859, 60793-860) , MANUFACTURE(60793-850, 60793-851, 60793-852, 60793-853, 60793-854, 60793-855, 60793-856, 60793-857, 60793-858, 60793-859, 60793-860) , PACK(60793-850, 60793-851, 60793-852, 60793-853, 60793-854, 60793-855, 60793-856, 60793-857, 60793-858, 60793-859, 60793-860)
Trademark Results [LEVOXYL]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 LEVOXYL 74534308 2016089 Live/Registered |
KING PHARMACEUTICALS RESEARCH AND DEVELOPMENT, LLC 1994-06-07 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

