SIKLOS- hydroxyurea tablet, film coated
Siklos by
Drug Labeling and Warnings
Siklos by is a Prescription medication manufactured, distributed, or labeled by Medunik, Addmedica. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use SIKLOS safely and effectively. See full prescribing information for SIKLOS.
SIKLOS (hydroxyurea) tablets, for oral use
Initial U.S. Approval: 1967WARNING: MYELOSUPPRESSION and MALIGNANCIES
See full prescribing information for complete boxed warning.
- Myelosuppression: SIKLOS may cause severe myelosuppression. Do not give if bone marrow function is markedly depressed. Monitor blood counts at baseline and throughout treatment. Interrupt treatment and reduce dose as necessary. (5.1)
- Malignancies: Hydroxyurea is carcinogenic. Advise sun protection and monitor patients for malignancies. (5.2)
INDICATIONS AND USAGE
SIKLOS is an antimetabolite, indicated to reduce the frequency of painful crises and to reduce the need for blood transfusions in pediatric patients, 2 years of age and older, with sickle cell anemia with recurrent moderate to severe painful crises. (1)
DOSAGE AND ADMINISTRATION
- Initial dose: 20 mg/kg once daily. Monitor blood counts every two weeks. (2.1)
- The dose may be increased by 5 mg/kg/day every 8 weeks, or sooner if a severe painful crisis occurs, until a maximum tolerated dose or 35 mg/kg/day is reached if blood counts are in an acceptable range. (2.1)
- Discontinue SIKLOS until hematologic recovery if blood counts are considered toxic. Resume treatment after reducing the dose by 5 mg/kg/day from the dose associated with hematological toxicity. (2.1)
- Renal impairment: Reduce the dose of SIKLOS by 50% in patients with creatinine clearance less than 60 mL/min. (2.2, 8.6, 12.3)
DOSAGE FORMS AND STRENGTHS
Tablets: 100 mg and functionally triple-scored 1,000 mg tablet (3)
CONTRAINDICATIONS
Patients who have demonstrated a previous hypersensitivity to hydroxyurea or any other component of its formulation. (4)
WARNINGS AND PRECAUTIONS
- Embryo-Fetal toxicity: Can cause fetal harm. Advise of potential risk to a fetus and use of effective contraception. (5.3, 8.1, 8.3)
- Cutaneous vasculitic toxicities (incl. leg ulcers): Institute treatment and discontinue SIKLOS and/or reduce dose if this occurs. (5.4)
- Risks with concomitant use of antiretroviral drugs: Pancreatitis, hepatotoxicity, and neuropathy have occurred. Monitor for signs and symptoms patients with HIV infection using antiretroviral drugs; discontinue SIKLOS, and implement treatment. (5.5)
- Concomitant use with live virus vaccine: increased risk of severe infections.
ADVERSE REACTIONS
USE IN SPECIFIC POPULATIONS
- Lactation: Advise women to stop breastfeeding while taking SIKLOS.
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 5/2018
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: MYELOSUPPRESSION and MALIGNANCIES
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing
2.2 Dose Modifications for Renal Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Myelosuppression
5.2 Malignancies
5.3 Embryo-Fetal Toxicity
5.4 Vasculitic Toxicities (including Leg Ulcers)
5.5 Risks with Concomitant Use of Antiretroviral Drugs
5.6 Risks with Concomitant Use of Live Virus Vaccine
5.7 Macrocytosis
5.8 Test Interference
6 ADVERSE REACTIONS
6.1 Clinical Trial Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Increased Toxicity with Concomitant Use of Antiretroviral Drugs
7.2 Concomitant Use of Live Virus Vaccine
7.3 Test Interference
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.6 Renal Impairment
8.7 Hepatic impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage
16.3 Handling and Disposal
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: MYELOSUPPRESSION and MALIGNANCIES
Myelosuppression: SIKLOS may cause severe myelosuppression. Monitor blood counts at baseline and throughout treatment. Interrupt treatment and reduce dose as necessary [see Warnings and Precautions (5.1)].
Malignancies: Hydroxyurea is carcinogenic. Advise sun protection and monitor patients for malignancies [see Warnings and Precautions (5.2)].
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing
The recommended SIKLOS dosing is described in Table 1.
Table 1: Dosing Recommendation Based on Blood Count Dosing Regimen Dose Dose Modification Criteria Monitoring Parameters Initial Recommended Dosing 20 mg/kg once daily based on patient's actual or ideal weight, whichever is less. Monitor the patient's blood count every 2 weeks [see Warnings and Precautions (5.1)]. Dosing Adjustment Based on Blood Counts in an acceptable range Increase dose 5 mg/kg/day every 8 weeks or if a painful crisis occurs.
Give until mild myelosuppression (absolute neutrophil count 2,000/uL to 4,000/uL) is achieved, up to a maximum of 35 mg/kg/day.Increase dosing only if blood counts are in an acceptable range.
Increase dosing if a painful crisis occurs.
Do not increase if myelosuppression occurs.Blood Counts Acceptable Range:
- neutrophils greater than or equal to 2,000 cells/mm3
- platelets greater than or equal to 80,000/mm3
- hemoglobin greater than 5.3 g/dL
- reticulocytes greater than or equal to 80,000/mm3 if the hemoglobin concentration less than 9 g/dLDosing Adjustment Based on Blood Counts in a toxic range Discontinue treatment. If blood counts are considered toxic, discontinue SIKLOS until hematologic recovery. Blood Counts Toxic Range:
- neutrophils less than 2,000 cells/mm3
younger patients with lower baseline counts may safely tolerate absolute neutrophil counts down to 1,250/mm3.
- platelets less than 80,000/mm3
- hemoglobin less than 4.5 g/dL
- reticulocytes less than 80,000/mm3 if the hemoglobin concentration less than 9 g/dLDosing After Hematologic Recovery Reduce dose by 5 mg/kg/day. Reduce the dose from the dose associated with hematologic toxicity.
May titrate up or down every 8 weeks in 5 mg/kg/day increments.
The patient should be at a stable dose with no hematologic toxicity for 24 weeks.
Discontinue the treatment permanently if a patient develops hematologic toxicity twice.Siklos is available in 100 mg and 1,000 mg tablets. The 1,000 mg tablets have 3 score lines and can be split into 4 parts (each 250 mg). Therefore, the two strengths can be used to deliver doses of 1,000 mg, 750 mg, 500 mg, 250 mg, 100 mg, and combinations thereof. Calculate the rounded doses to the nearest 50 mg or 100 mg strength based on clinical judgment.
Do not split the SIKLOS 100 mg tablets into smaller parts.
Patients must be able to follow directions regarding drug administration and their monitoring and care.
Fetal hemoglobin (HbF) levels may be used to evaluate the efficacy of SIKLOS in clinical use. Obtain HbF levels every three to four months. Monitor for an increase in HbF of at least two-fold over the baseline value.
Administration:
The tablet should be taken once daily, with a glass of water. For patients who are not able to swallow the tablets, these can be dispersed immediately before use in a small quantity of water in a teaspoon.
SIKLOS is a cytotoxic drug. Follow applicable special handling and disposal procedures [see References (15)].
2.2 Dose Modifications for Renal Impairment
Reduce the dose of SIKLOS by 50% in patients with creatinine clearance of less than 60 mL/min or with end-stage renal disease (ESRD) [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)]. Obtain the creatinine clearance using a 24-hour urine collection.
Creatinine Clearance
(mL/min)Recommended SIKLOS Initial Dose
(mg/kg daily)- * On dialysis days, administer SIKLOS to patients with ESRD following hemodialysis
Greater than or equal to 60 20 Less than 60 or ESRD* 10 Monitor the hematologic parameters closely in these patients.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
SIKLOS is contraindicated in:
- - Patients who have demonstrated a previous hypersensitivity to hydroxyurea or any other component of its formulation [see Adverse Reactions (6)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Myelosuppression
Hydroxyurea causes severe myelosuppression. Do not initiate treatment with hydroxyurea in patients if bone marrow function is markedly depressed. Bone marrow suppression may occur, and leukopenia is generally its first and most common manifestation. Thrombocytopenia and anemia occur less often, and are seldom seen without a preceding leukopenia.
Some patients, treated at the recommended initial dose of 20 mg/kg/day, have experienced severe or life-threatening myelosuppression. Due to the change in body weight requiring modification of daily dose, pediatric patients have an increased risk of myelosuppression at the time of dose adjustment.
Evaluate hematologic status prior to and during treatment with SIKLOS. Provide supportive care and modify dose or discontinue SIKLOS as needed. Recovery from myelosuppression is usually observed within 15 days when therapy is interrupted. Resume therapy after interruption at a lower dose [see Dosage and Administration (2.1)].
5.2 Malignancies
Hydroxyurea is a human carcinogen. In patients receiving long-term hydroxyurea for myeloproliferative disorders (a condition for which Siklos is not approved), secondary leukemia has been reported. Skin cancer has also been reported in patients receiving long-term hydroxyurea. Advise protection from sun exposure and monitor for the development of secondary malignancies.
5.3 Embryo-Fetal Toxicity
Based on the mechanism of action and findings in animals, SIKLOS can cause fetal harm when administered to a pregnant woman. Hydroxyurea was embryotoxic and teratogenic in rats and rabbits at doses 0.8 times and 0.3 times, respectively, the maximum recommended human daily dose on a mg/m2 basis. Advise pregnant women of the potential risk to a fetus [see Use in Specific Populations (8.1)].
Advise females of reproductive potential to use effective contraception during and after treatment with SIKLOS for at least 6 months after therapy. Advise males of reproductive potential to use effective contraception during and after treatment with SIKLOS for at least 6 months after therapy [see Use in Specific Populations (8.1, 8.3)].
5.4 Vasculitic Toxicities (including Leg Ulcers)
Cutaneous vasculitic toxicities, including vasculitic ulcerations and gangrene, have occurred in patients with myeloproliferative disorders during therapy with hydroxyurea. These vasculitic toxicities were reported most often in patients with a history of, or currently receiving, interferon therapy. Due to potentially severe clinical outcomes for the cutaneous vasculitic ulcers reported in patients with myeloproliferative disease (a condition for which SIKLOS is not approved), treatment with SIKLOS should be discontinued and/or its dose reduced if cutaneous vasculitic ulcerations develop. Rarely, ulcers are caused by leukocytoclastic vasculitis.
Avoid use of SIKLOS in patients with wounds on the legs (leg ulcers).
5.5 Risks with Concomitant Use of Antiretroviral Drugs
Pancreatitis, hepatotoxicity, and peripheral neuropathy have occurred when hydroxyurea was administered concomitantly with antiretroviral drugs, including didanosine and stavudine [see Drug Interactions (7.1)].
5.6 Risks with Concomitant Use of Live Virus Vaccine
Avoid use of live virus vaccine in patients taking SIKLOS. Concomitant use of hydroxyurea with a live virus vaccine may potentiate the replication of the vaccine virus and/or may increase the adverse reactions of the vaccine virus and result in severe infections [see Drug Interactions (7.2)]. Patient's antibody response to vaccines may be decreased. Consider consultation with a specialist.
5.7 Macrocytosis
SIKLOS may cause macrocytosis, which is self-limiting, and is often seen early in the course of treatment. The morphologic change resembles pernicious anemia, but is not related to vitamin B12 or folic acid deficiency. This may mask the diagnosis of pernicious anemia. Prophylactic administration of folic acid is recommended.
5.8 Test Interference
Interference with Uric Acid, Urea, or Lactic Acid Assays is possible, rendering falsely elevated results of these in patients treated with hydroxyurea [see Drug Interactions (7.3)].
-
6 ADVERSE REACTIONS
- Myelosuppression [see Warnings and Precautions (5.1)]
- Malignancies [see Warnings and Precautions (5.2)]
- Embryo-fetal toxicity [see Boxed Warning and Warnings and Precautions (5.3)]
- Vasculitic toxicities (including Leg Ulcers) [see Warnings and Precautions (5.4)]
- Risks with concomitant use of antiretroviral drugs [see Warnings and Precautions (5.5)]
- Risk with concomitant use of live virus vaccine [see Warnings and Precautions (5.6)]
- Macrocytosis [see Warnings and Precautions (5.7)]
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of SIKLOS has been assessed in 405 pediatric patients with sickle cell disease from 2-18 years of age in the European Sickle Cell Disease prospective Cohort study ESCORT-HU.
The most frequently reported adverse reactions in ESCORT-HU were infections and myelosuppression, with mild to moderate neutropenia as the most common manifestation.
Other adverse reactions include skin and subcutaneous disorders (skin depigmentation/melanonychia, skin rash, alopecia), gastrointestinal disorders, vitamin D deficiency and headache.
At least one serious adverse reaction was reported in 32.6 % of the 405 pediatric patients with sickle cell disease in ESCORT-HU. The most frequent serious adverse reactions were infections (17.8 %), and blood and lymphatic system disorders (9.1 %). This included serious neutropenia (3.2%), thrombocytopenia (3.0%) and anemia (3.0%). Other reported serious adverse reactions were gastrointestinal disorders (3.2 %), fever (2.5 %) and nervous system disorders (4.0 %), including headache (2.7%).
Table 2: Most frequent (greater than or equal to 2.0%) adverse reactions reported in pediatric patients enrolled in ESCORT-HU Global Safety Set (N=405) Total Intensity Mild Moderate Severe n % n % n % n % n: number of patients with an adverse reaction At least one adverse reaction 261 64.4 Infections 161 39.8 120 29.6 88 21.7 18 4.4 Other Infections 92 22.7 66 16.3 32 7.9 3 0.7 Bacterial 65 16.0 24 5.9 37 9.1 10 2.5 Viral 40 9.9 23 5.7 14 3.5 3 0.7 Parvovirus B19 15 3.7 7 1.7 5 1.2 2 0.5 Blood and lymphatic system disorders 85 21.0 51 12.6 59 14.6 14 3.5 Neutropenia 51 12.6 26 6.4 31 7.7 4 1.0 Thrombocytopenia 30 7.4 16 4.0 15 3.7 2 0.5 Anemia 17 4.2 4 1.0 8 2.0 7 1.7 Gastrointestinal disorders 53 13.1 29 7.2 30 7.4 4 1.0 Other Gastrointestinal Disorders 30 7.4 13 3.2 15 3.7 2 0.5 Constipation 10 2.5 5 1.2 5 1.2 0 0 Nausea 10 2.5 4 1.0 4 1.0 2 0.5 Metabolic and nutrition disorders 44 10.9 24 5.9 21 5.2 1 0.2 Deficiency of vitamin D 25 6.2 19 4.7 7 1.7 1 0.2 Other Metabolic and nutrition disorders 8 2.0 3 0.7 4 1.0 1 0.2 Weight gain 8 2.0 1 0.2 7 1.7 0 0 Nervous system disorders 45 11.1 19 4.7 19 4.7 8 2.0 Headache 30 7.4 15 3.7 7 1.7 4 1.0 Other Nervous system disorders 11 2.7 2 0.5 4 1.0 4 1.0 General disorders 41 10.1 22 5.4 17 4.2 4 1.0 Fever 31 7.7 20 4.9 12 3.0 2 0.5 Skin and subcutaneous tissue disorders 38 9.4 29 7.2 14 3.5 1 0.2 Skin reactions 15 3.7 8 2.0 7 1.7 1 0.2 Other Skin and subcutaneous tissue disorders 13 3.2 8 2.0 5 1.2 0 0 Other Not SCD-related reactions 23 5.7 16 4.0 3 0.7 1 0.2 Other Not SCD-related reactions 23 5.7 16 4.0 3 0.7 1 0.2 Respiratory thoracic and mediastinal disorders 11 2.7 6 1.5 3 0.7 2 0.5 Renal and urinary disorders 8 2.0 2 0.5 4 1.0 0 0 6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of SIKLOS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Infections and infestations: Parvovirus B19 infection
- Blood and lymphatic system disorders: bone marrow depression including neutropenia (<2.0 × 109/l), reticulocytopenia (<80 × 109/l), macrocytosis, thrombocytopenia (<80 × 109/l), anemia (hemoglobin <4.5g/dl)
- Nervous system disorders: headache, dizziness
- Gastrointestinal disorders: nausea, gastrointestinal disturbances, vomiting, gastrointestinal ulcer, severe hypomagnesemia
- Hepatobiliary disorders: elevation of hepatic enzymes
- Skin and subcutaneous tissue disorders: skin reactions (oral, ungula and cutaneous pigmentation), oral mucositis, rash, melanonychia, alopecia, leg ulcers, cutaneous dryness
- Reproductive system and breast disorders: oligospermia, azoospermia, amenorrhea
- General disorders: fever
- Investigations: weight gain
-
7 DRUG INTERACTIONS
7.1 Increased Toxicity with Concomitant Use of Antiretroviral Drugs
Pancreatitis
Pancreatitis (including fatal cases) have occurred in patients with HIV infection during therapy with hydroxyurea and didanosine, with or without stavudine. Hydroxyurea is not indicated for the treatment of HIV infection; however, if patients with HIV infection are treated with hydroxyurea, and in particular, in combination with didanosine and/or stavudine, monitor closely for signs and symptoms of pancreatitis. Permanently discontinue therapy with hydroxyurea in patients who develop signs and symptoms of pancreatitis.
Hepatotoxicity
Hepatotoxicity and hepatic failure resulting in death have been reported during postmarketing surveillance in patients with HIV infection treated with hydroxyurea and other antiretroviral drugs. Fatal hepatic events were reported most often in patients treated with the combination of hydroxyurea, didanosine, and stavudine. Avoid this combination.
7.2 Concomitant Use of Live Virus Vaccine
Concomitant use of SIKLOS with a live virus vaccine may potentiate the replication of the vaccine virus and/or may increase the adverse reactions of the vaccine virus, because normal defense mechanisms may be suppressed by SIKLOS therapy. Vaccination with a live vaccine in a patient taking SIKLOS may result in severe infections. Generally, the patient's antibody response to vaccines may be decreased. Treatment with SIKLOS and concomitant immunization with live virus vaccines should only be performed if benefits clearly outweigh potential risks. Consider consultation with a specialist.
7.3 Test Interference
Interference with Uric Acid, Urea, or Lactic Acid Assays
Studies have shown that there is an analytical interference of SIKLOS with the enzymes (urease, uricase, and lactate dehydrogenase) used in the determination of urea, uric acid, and lactic acid, rendering falsely elevated results of these in patients treated with SIKLOS.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
SIKLOS can cause fetal harm based on findings from animal studies and the drug's mechanism of action [see Clinical Pharmacology (12.1)]. There are no studies with the use of SIKLOS in pregnant women, and limited available data on SIKLOS use during pregnancy are insufficient to inform drug-associated risks. Drugs which affect DNA synthesis, such as hydroxyurea, may be potential mutagenic agents. In animal reproduction studies, administration of hydroxyurea to pregnant rats and rabbits during organogenesis produced embryotoxic and teratogenic effects at doses 0.8 times and 0.3 times, respectively, the maximum recommended human daily dose on a mg/m² basis. In rats and rabbits, fetal malformations were observed with partially ossified cranial bones, absence of eye sockets, hydrocephaly, bipartite sternebrae, and missing lumbar vertebrae. Embryotoxicity was characterized by decreased fetal viability, reduced live litter sizes, and developmental delays (see Data). Advise pregnant women of the potential risk to a fetus (see Clinical Considerations).
Background risk of major birth defects and miscarriage for the indicated population are unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2%–4% and 15%–20%, respectively.
Clinical Considerations
Fetal/Neonatal adverse reactions
Although the data on a limited number of exposed pregnancies indicate no adverse effects on pregnancy or on the health of the fetus/newborn, patients on SIKLOS should be made aware of the potential risks to the fetus.
Based on the limited amount of available information, in case of an exposure to SIKLOS of pregnant female patients or pregnant partners of male patients, treated by SIKLOS, a careful follow-up with adequate clinical, biological and ultrasonographic examinations should be considered.
Data
Human Data
According to a retrospective analysis of a cohort of 123 adult patients treated with hydroxyurea, twenty-three pregnancies have been reported from 15 women treated with hydroxyurea and partners of 3 men not using barrier contraception treated with hydroxyurea. Most (61%) had no adverse developmental outcomes. In the other cases with known evolution, pregnancy had been interrupted either voluntarily or upon medical advice.
In retrospective cohorts of 352 children and adolescents with sickle cell disease older than 2 years treated with hydroxyurea for a period of up to 12 years, 3 pregnancies under hydroxyurea were reported with no adverse developmental outcomes.
From post-marketing data of SIKLOS, 3 pregnancies have been reported while the father was treated with SIKLOS and 16 pregnancies have been reported in 15 females treated with SIKLOS. Among the 13 cases with known evolution, 5 pregnancies had no adverse developmental outcomes, 4 led to premature birth, and 4 were early terminated.
Animal Data
Hydroxyurea has been demonstrated to be a potent teratogen in a wide variety of animal models, including mice, hamsters, cats, miniature swine, dogs, and monkeys at doses within 1-fold of the human dose given on a mg/m2 basis. Hydroxyurea is embryotoxic and causes fetal malformations (partially ossified cranial bones, absence of eye sockets, hydrocephaly, bipartite sternebrae, missing lumbar vertebrae) at 180 mg/kg/day (about 0.8 times the maximum recommended human daily dose on a mg/m2 basis) in rats and at 30 mg/kg/day (about 0.3 times the maximum recommended human daily dose on a mg/m2 basis) in rabbits. Embryotoxicity was characterized by decreased fetal viability, reduced live litter sizes, and developmental delays. Hydroxyurea crosses the placenta. Single doses of ≥375 mg/kg (about 1.7 times the maximum recommended human daily dose on a mg/m2 basis) to rats caused growth retardation and impaired learning ability.
8.2 Lactation
Risk Summary
It is not known whether SIKLOS is excreted in human milk, the effects of SIKLOS on the breastfed child, or the effects of SIKLOS on milk production. Because of the potential for serious adverse reactions in a breastfed child from SIKLOS, including carcinogenicity, advise patients not to breastfeed during treatment with SIKLOS.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
SIKLOS can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Verify the pregnancy status of females of reproductive potential prior to initiating SIKLOS therapy.
Contraception
Females
Advise females of reproductive potential to use effective contraception during and after treatment with SIKLOS for at least 6 months after therapy. Advise females to immediately report pregnancy.
Males
SIKLOS may damage spermatozoa and testicular tissue, resulting in possible genetic abnormalities. Males with female sexual partners of reproductive potential should use effective contraception during and after treatment with SIKLOS for at least 6 months after therapy [see Nonclinical Toxicology (13.1)].
Infertility
Males
Based on findings in animals and humans, male fertility may be compromised by treatment with SIKLOS. Azoospermia or oligospermia, sometimes reversible, has been observed in men. Before the start of therapy, inform male patients about the possibility of sperm conservation [see Adverse Reactions (6) and Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of SIKLOS have been established in pediatric patients aged 2-18 years with sickle cell anemia with recurrent moderate to severe painful crises. Use of SIKLOS in these age groups is supported by evidence from a non-interventional cohort study, the European Sickle Cell Disease prospective Cohort study, ESCORT-HU, in which 405 pediatric patients ages 2 to <18 were enrolled. Among the 405 pediatric patients treated with SIKLOS, 274 were children (2-11) and 108 were adolescents (12-16) [see Clinical Studies (14)].
Continuous follow-up of the growth of treated children is recommended.
Pediatric patients aged 2-16 years had a higher risk of neutropenia than patients more than 16 years old.
The safety and effectiveness of SIKLOS have not been established in pediatric patients less than 2 years of age.
8.6 Renal Impairment
The exposure to SIKLOS is higher in patients with creatinine clearance of less than 60 mL/min. Reduce dosage and closely monitor the hematologic parameters when SIKLOS is to be administered to these patients [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
Acute mucocutaneous toxicity has been reported in patients receiving hydroxyurea at doses several times above the therapeutic dose. Soreness, violet erythema, oedema on palms and soles followed by scaling of hand and feet, severe generalized hyperpigmentation of the skin and stomatitis have been observed. In patients with sickle cell anemia, neutropenia was reported in isolated cases of hydroxyurea overdose (1.43 times and 8.57 times of the maximum recommended dose of 35 mg/kg b.w./day). Monitor blood counts weekly until recovery. Treatment of overdose consists of gastric lavage, followed by symptomatic treatment and control of bone marrow function.
-
11 DESCRIPTION
SIKLOS (hydroxyurea) is an antimetabolite that is available for oral use as 100 mg film-coated tablet and functionally triple-scored 1,000 mg film-coated tablet containing 100 and 1,000 mg of hydroxyurea, respectively. Inactive ingredients include silicified microcrystalline cellulose, sodium stearyl fumarate, and film-coating agent amino methacrylate copolymer.
Hydroxyurea is a white crystalline powder. It has a molecular weight of 76.05. Its structural formula is:
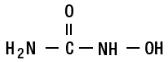
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The precise mechanism by which hydroxyurea produces its cytotoxic and cytoreductive effects is not known. However, various studies support the hypothesis that hydroxyurea causes an immediate inhibition of DNA synthesis by acting as a ribonucleotide reductase inhibitor, without interfering with the synthesis of ribonucleic acid or of protein.
The mechanisms by which SIKLOS produces its beneficial effects in patients with sickle cell Anemia (SCA) are uncertain. Known pharmacologic effects of SIKLOS that may contribute to its beneficial effects include increasing hemoglobin F levels in red blood cells (RBCs), decreasing neutrophils, increasing the water content of RBCs, increasing deformability of sickled cells, and altering the adhesion of RBCs to endothelium.
12.2 Pharmacodynamics
The correlation between hydroxyurea concentrations, reduction of crisis rate, and increase in HbF, is not known.
12.3 Pharmacokinetics
Absorption
Following oral administration, hydroxyurea reaches peak plasma concentrations in 1 to 4 hours. Mean peak plasma concentrations and AUCs increase more than proportionally with increase of dose.
Distribution
Hydroxyurea distributes throughout the body with a volume of distribution approximating total body water. Hydroxyurea concentrates in leukocytes and erythrocytes.
Elimination
Specific Populations
Patients with Renal Impairment
The effect of renal impairment on the pharmacokinetics of hydroxyurea was assessed in adult patients with sickle cell anemia and renal impairment. Patients with normal renal function (creatinine clearance [CrCl] >80 mL/min), mild (CrCl 50-80 mL/min), moderate (CrCl =30-<50 mL/min), or severe (<30 mL/min) renal impairment received a single oral dose of 15 mg/kg hydroxyurea. Creatinine clearance values were obtained using 24-hour urine collections. Patients with ESRD received two doses of 15 mg/kg separated by 7 days; the first was given following a 4-hour hemodialysis session, the second prior to hemodialysis. The exposure to hydroxyurea (mean AUC) in patients with CrCl <60 mL/min and those with ESRD was 64% higher than in patients with normal renal function (CrCl >60 mL/min). Reduce the dose of SIKLOS when it is administered to patients with creatinine clearance of <60 mL/min or with ESRD following hemodialysis [see Dosage and Administration (2.2) and Use in Specific Populations (8.6)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Conventional long-term studies to evaluate the carcinogenic potential of hydroxyurea have not been performed. However, hydroxyurea is presumed to be a transspecies carcinogen. Intraperitoneal administration of 125 to 250 mg/kg hydroxyurea (about 0.6-1.2 times the maximum recommended human oral daily dose on a mg/m2 basis) thrice weekly for 6 months to female rats increased the incidence of mammary tumors in rats surviving to 18 months compared to control. Hydroxyurea is mutagenic in vitro to bacteria, fungi, protozoa, and mammalian cells. Hydroxyurea is clastogenic in vitro (hamster cells, human lymphoblasts) and in vivo (SCE assay in rodents, mouse micronucleus assay). Hydroxyurea causes the transformation of rodent embryo cells to a tumorigenic phenotype [see Warnings and Precautions (5.2, 5.3)].
Hydroxyurea administered to male rats at 60 mg/kg /day (about 0.3 times the maximum recommended human daily dose on a mg/m2 basis) produced testicular atrophy, decreased spermatogenesis and significantly reduced their ability to impregnate females [see Use in Specific Populations (8.3)].
-
14 CLINICAL STUDIES
The efficacy of SIKLOS was assessed in the European Sickle Cell Disease Cohort study (ESCORT HU) [NCT02516579]. This is an open-label single-arm study of 405 pediatric patients with sickle cell disease from 2-18 years of age, of which 141 had not been previously treated with hydroxyurea prior to enrollment. Evaluable patients had at least 12 months follow-up (median [range] 23 months [12,80]). Median (range) hemoglobin F percentages were 5.6% (1.3, 15.0) at baseline and 12.8% (2.1, 37.2) at least 6 months (the value closest to 6 months collected between 5 and 14 months) after initiation of SIKLOS treatment, with median (range) change of 5.9% (-2.2, 34.7) in 47 patients. Median (range) hemoglobin levels were 8.2 g/dL (3.7, 14.2) at baseline, 8.8 g/dL (0.7, 13.1) at 6 months (the value closest to 6 months collected between 5 and 7 months), and 8.9 g/dL (5.5, 13.2) at 12 months (the value closest to 12 months collected between 10 and 14 months) after initiation of SIKLOS treatment. The median (range) change was 0.5 g/dL (-4.6, 6.1) in 63 patients at 6 months (the post-baseline value closest to 6 months collected between 5 and 7 months) and 0.7 g/dL (-6.4, 6.0) in 83 patients at 12 months (the post-baseline value closest to 12 months collected between 10 and 14 months) after initiation of SIKLOS treatment.
Among pediatric patients not previously treated with hydroxyurea prior to enrollment and analyzable for efficacy (N=141), the percentage of patients with at least one vaso-occlusive episode, one episode of acute chest syndrome, one hospitalization due to SCD or one blood transfusion decreased after 12 months of SIKLOS treatment (Table 3).
Table 3: Comparison of SCD Events in the First Year of Treatment with SIKLOS with SCD Events in the 12 Months Prior to Enrollment – ESCORT HU Trial (N=141) SCD events Patients under 18 years old previously not treated with hydroxyurea with at least 12 months follow-up data available for clinical efficacy (N=141) In the 12 months prior to enrolment After 12 months of Siklos® treatment Change Number of patients with at least one vaso-occlusive episode (in 120 evaluable patients) No 37 (30.8%) 69 (57.5%) Yes 83 (69.2%) 51 (42.5%) Number of vasoocclusive episodes over 12 months (in 113 evaluable patients) Median (range) 2.0 (0.0, 10.0) 0.0 (0.0, 7.0) -1.0 (-10.0, 5.0) Number of patients with at least one episode of acute chest syndrome (in 123 evaluable patients) No 94 (76.4%) 116 (94.3%) Yes 29 (23.6%) 7 (5.7%) Number of episodes of acute chest syndrome over 12 months (in 123 evaluable patients) Median (range) 0.0 (0.0, 2.0) 0.0 (0.0, 1.0) 0.0 (-2.0, 1.0) Number of patients with at least one hospitalization related to SCD (in 110 evaluable patients) No 27 (24.5%) 64 (58.2%) Yes 83 (75.5%) 46 (41.8%) Number of hospitalizations related to SCD over 12 months (in 106 evaluable patients) Median (range) 2.0 (0.0, 6.0) 0.0 (0.0, 7.0) -1.0 (-6.0, 6.0) Number of days of hospitalizations related to SCD over 12 months (in 100 evaluable patients) Median (range) 8.0 (0.0, 58.0) 0.0 (0.0, 100.0) -3.0 (-58.0, 86.0) Number of patients with at least one blood transfusion (in 122 evaluable patients) No 66 (54.1%) 94 (77.0%) Yes 56 (45.9%) 28 (23.0%) - 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
SIKLOS (hydroxyurea) film-coated tablet is supplied in high density polyethylene (HDPE) bottle with polypropylene child-resistant cap with a desiccant unit containing 30 (SIKLOS 1,000 mg) or 60 (SIKLOS 100 mg) film coated tablets. Each bottle containing SIKLOS 100 mg tablets or SIKLOS 1000 mg tablets is supplied in a carton.
SIKLOS is supplied in the following strengths:
- 100 mg off-white round, film-coated tablet, embossed with 100 on one side.
- 1,000 mg off-white, capsule-shaped, film-coated, functionally triple-scored tablet with scoring on both sides which can be divided into four equal parts embossed with "T" on one side.Bottles of 30 Bottles of 60 100 mg N/A NDC: 71770-100-60 1,000 mg NDC: 71770-120-30 N/A 16.2 Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Keep tightly closed.
Broken 1,000 mg tablets must be stored in the bottle and must be used within three months.
16.3 Handling and Disposal
SIKLOS is a cytotoxic drug. Follow applicable special handling and disposal procedures [see References (15)].
To decrease the risk of contact, advise caregivers to wear disposable gloves when handling SIKLOS or bottles containing SIKLOS. Wash hands with soap and water before and after contact with the bottle or tablets when handling SIKLOS. Avoid exposure to crushed tablets. If contact with crushed tablets occurs on the skin, wash affected area immediately and thoroughly with soap and water. If contact with crushed tablets occurs on the eye(s), the affected area should be flushed thoroughly with water or isotonic eyewash designated for that purpose for at least 15 minutes.
Powder spilled from the broken tablet should be wiped up with a damp disposable towel which must be thrown away in a closed container such as a plastic bag to avoid ingestion of powder by other people. The spill areas should then be cleaned using a detergent solution followed by clean water.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient or caregiver to read the FDA-approved patient labeling (Instructions for Use and Medication Guide).
- There is a risk of myelosuppression. Emphasize the importance of monitoring blood counts every two weeks throughout the duration of therapy to patients taking SIKLOS [see Warnings and Precautions (5.1)]. Advise patients to report signs and symptoms of infection or bleeding immediately.
- Advise patients that there is a risk of cutaneous vasculitic toxicities and secondary malignancies including leukemia. Advise use of sun protection [see Warnings and Precautions (5.1)].
- Advise females of reproductive potential of the potential risk to a fetus should they become pregnant while taking SIKLOS. Advise patients to inform their healthcare provider of a known or suspected pregnancy. Advise females and males of reproductive potential to use contraception during and after treatment with SIKLOS [see Warnings and Precautions (5.3) and Use in Specific Populations (8.1,8.3)].
- Advise females to discontinue breastfeeding during treatment with SIKLOS [see Use in Specific Populations (8.2)].
- Advise male patients of potential risk to fertility.
- Advise patients with HIV infection to contact their physician for signs and symptoms of pancreatitis, hepatic events, and peripheral neuropathy [see Warnings and Precautions (5.5)].
- Because SIKLOS 1,000 mg tablets are scored, advise patients on how to take SIKLOS properly.
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
This Medication Guide has been approved by the U.S. Food and Drug Administration. Issued: 05/2018 MEDICATION GUIDE
SIKLOS (See – k – los)
(hydroxyurea)
tabletsWhat is the most important information I should know about SIKLOS?
SIKLOS can cause serious side effects including:
- Low blood cell counts are common with SIKLOS, including low red blood cells, white blood cells, and platelets, and can be severe and life-threatening. If your white blood cell count becomes very low, you are at increased risk for infection. Your healthcare provider will check your blood cell counts before and during treatment with SIKLOS. Your healthcare provider may change your dose or tell you to stop taking SIKLOS if you have low blood cell counts. Tell your healthcare provider right away if you get any of the following symptoms:
- fever or chills
- body aches
- feeling very tired
- shortness of breath
- unusual headache
- bleeding or unexplained bruising
- Cancer. Some people have developed cancer, such as leukemia and skin cancer, after taking SIKLOS for a long time. Your healthcare provider will check you for cancer. You should protect your skin from the sun using sunblock, hats, and sun-protective clothing.
-
SIKLOS can harm your unborn baby.
For females taking SIKLOS who can become pregnant:- You should talk with your healthcare provider about the risks of SIKLOS to your unborn baby.
- You should use effective birth control during treatment with SIKLOS and for at least 6 months after treatment with SIKLOS.
- Your healthcare provider will perform a pregnancy test before you start treatment with SIKLOS. Tell your healthcare provider right away if you become pregnant or think you may be pregnant.
- For males taking SIKLOS: SIKLOS can affect your sperm. If you have a female sexual partner who can become pregnant, you should use effective birth control during treatment with SIKLOS and for at least 6 months after treatment.
SIKLOS may cause fertility problems in males. Talk to your healthcare provider if this is a concern for you.
See "What are the possible side effects of SIKLOS?" for more information about side effects.
What is SIKLOS?
SIKLOS is a prescription medicine that is used to reduce the frequency of painful crises and reduce the need for blood transfusions in children, 2 years of age and older, with sickle cell anemia with recurrent moderate to severe painful crises.
It is not known if SIKLOS is safe and effective in children less than 2 years of age.
Do not take SIKLOS if you are allergic to hydroxyurea or any of the ingredients in SIKLOS. See the end of this Medication Guide for a list of the ingredients in SIKLOS. Before taking SIKLOS, tell your healthcare provider about all of your medical conditions, including if you:
- have kidney problems or are receiving hemodialysis
- have liver problems
- have human immunodeficiency virus (HIV) or take HIV medicines. Taking SIKLOS with certain HIV medicines can cause serious reactions and may lead to death.
- have increased levels of uric acid in your blood (hyperuricemia)
- have a history of receiving interferon therapy or are currently receiving interferon therapy
- have leg wounds or ulcers
- plan to receive any vaccinations. You should not receive "live vaccines" during treatment with SIKLOS.
- are pregnant or plan to become pregnant. See "What is the most important information I should know about SIKLOS?"
- are breastfeeding or plan to breastfeed. It is not known if SIKLOS can pass into your breast milk. Do not breastfeed during treatment with SIKLOS.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How should I take SIKLOS?
Read the Instructions for Use at the end of this Medication Guide for step-by-step instructions on how to prepare a dose of SIKLOS. If you have any questions, talk to your healthcare provider or pharmacist.
- Take SIKLOS exactly as your healthcare provider tells you to take it.
- Take SIKLOS 1 time a day at the same time each day.
- Swallow the tablet with a glass of water.
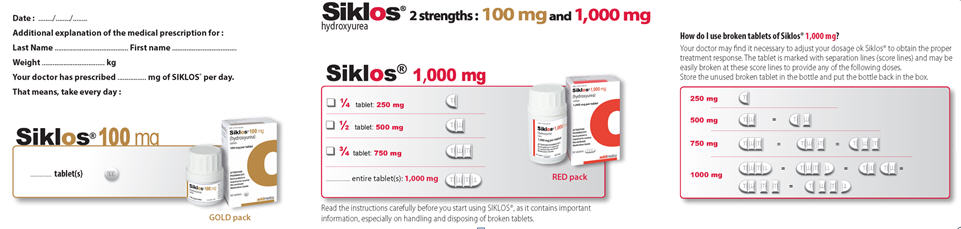
- SIKLOS is supplied as 100 mg tablets and 1,000 mg tablets. The SIKLOS 1,000 mg tablet has three separation lines (score lines) and can be broken at these score lines to provide smaller doses. Each 1,000 mg tablet can be divided into 4 equal parts (each part is 250 mg).
- Do not break the SIKLOS 100 mg tablets into smaller parts.
- Your healthcare provider will tell you how many tablets or parts of a tablet you should take. If you are not able to swallow SIKLOS tablets, you can dissolve your prescribed dose in a small amount of water in a teaspoon and swallow right away.
- SIKLOS tablets must be handled with care. To decrease the risk of exposure, you or your caregivers should do the following when handling SIKLOS:
- Wear disposable gloves when handling SIKLOS or bottles containing SIKLOS. Wash your hands with soap and water before and after handling SIKLOS tablets or bottles containing SIKLOS.
- Avoid contact with crushed tablets. If contact with crushed tablets happens on the skin, wash the skin area right away and thoroughly with soap and water. If contact with crushed tablets happens in the eyes, flush the eyes thoroughly with water or isotonic eyewash used for that purpose for at least 15 minutes.
Powder spilled from the broken tablet should be wiped up with a damp disposable towel which must be thrown away in a closed container such as a plastic bag to avoid ingestion of powder by other people. The spill areas should then be cleaned using a detergent solution followed by clean water.
- If you take too much SIKLOS, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of SIKLOS?
SIKLOS may cause serious side effects, including:
See "What is the most important information I should know about SIKLOS?"
- Skin ulcers, including leg ulcers have happened in people who take SIKLOS. This has happened most often in people who receive interferon therapy or have a history of interferon therapy. Your healthcare provider will decrease your dose or stop treatment with SIKLOS if you develop any skin ulcers.
- Enlarged red blood cells (macrocytosis). Macrocytosis is common in people who take SIKLOS and can make it difficult to detect a decrease of folic acid. Your healthcare provider may prescribe a folic acid supplement for you.
The most common side effects of SIKLOS include:
- infections
- headache
- fever
- skin problems including:
- skin reactions
- dry skin
- changes in skin and nail color
- stomach and intestine (gastrointestinal) problems including:
- nausea
- constipation
- decrease in vitamin D
- weight gain
These are not all the possible side effects of SIKLOS.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store SIKLOS?
- Store SIKLOS at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep the SIKLOS bottle tightly closed.
- Broken 1,000 mg SIKLOS tablets must be stored in the bottle and must be used within three months.
Keep SIKLOS and all medicines out of the reach of children.
General information about the safe and effective use of SIKLOS
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use SIKLOS for a condition for which it was not prescribed. Do not give SIKLOS to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about SIKLOS that is written for health professionals.
What are the ingredients of SIKLOS?
Active ingredient: hydroxyurea
Inactive ingredients: silicified microcrystalline cellulose, sodium stearyl fumarate, and film-coating agent amino methacrylate copolymer.
Distributed by: MEDUNIK USA.
For more information, call 1-844-884-5520. -
INSTRUCTIONS FOR USE
INSTRUCTIONS FOR USE
SIKLOS (See – k – los)
(hydroxyurea)
tabletsRead this Instructions for Use before you start taking SIKLOS and each time you get a refill. There may be new information. This Instructions for Use does not take the place of talking to your healthcare provider about your medical condition or treatment. You and your healthcare provider should talk about SIKLOS when you start taking it and at regular checkups.
Important Information:
- Wash your hands with soap and water before and after handling SIKLOS tablets or bottles containing SIKLOS.
- Wear disposable gloves when handling SIKLOS tablets or bottles containing SIKLOS.
- Take SIKLOS 1 time a day at the same time each day.
- Powder spilled from a broken tablet should be wiped up right away with a damp disposable paper towel and thrown away in a closed container, such as a plastic bag to avoid harm to other people. The spill area should then be cleaned using a detergent solution followed by clean water.
- When the tablet is broken, avoid touching the broken surfaces.
- If contact with crushed tablets happens on the skin, wash the skin area right away and thoroughly with soap and water.
- If contact with crushed tablets happens in the eyes, flush the eyes thoroughly with water or isotonic eyewash used for that purpose for at least 15 minutes.
SIKLOS is supplied in 2 different strengths:

SIKLOS 1,000 mg tablet has three separation lines (score lines) and can be broken at these score lines to provide smaller doses. Each 1,000 mg tablet can be divided into 4 equal parts (each part is 250 mg).

Do not break the SIKLOS 100 mg into smaller tablets.
SIKLOS 1,000 mg Tablet Breaking Instructions:
You will need the following supplies to break a SIKLOS 1,000 mg tablet:
- SIKLOS 1,000 mg tablets
- A damp disposable paper towel
- A tablet cutter
- Disposable gloves
Step 1: Place a damp disposable paper towel on a flat surface where the tablets will be broken. Step 2: Wash and dry your hands before handling SIKLOS tablets or bottles containing the tablets.
Step 3: Check your prescribed dose. You may need more than 1 tablet to get your prescribed dose. Step 4: Put on disposable gloves. 
Step 5: Remove the SIKLOS 1,000 mg tablet out of the bottle needed to get your dose. 
Step 6: Use your index fingers and thumbs to hold each end of the SIKLOS 1,000 mg tablet. 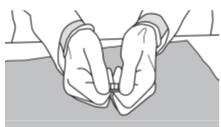
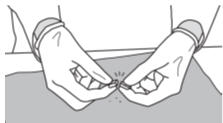
Step 7: While holding the ends of the SIKLOS 1,000 mg tablet, push down on the tablet to break the tablet on the score line to get your prescribed dose.
SIKLOS 1,000 mg tablets can be broken as:- 1/4 of a tablet for a dose of 250 mg of SIKLOS:

- 1/2 of a tablet for a dose of 500 mg of SIKLOS:

- 3/4 of a tablet for a dose of 750 mg of SIKLOS:

- a whole tablet for a dose of 1,000 mg of SIKLOS (no breaking needed):

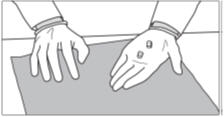
Step 8: Take your prescribed dose by swallowing it with a glass of water.
Important: If you have difficulty swallowing SIKLOS tablets, please stop here and follow the instructions below, "For people who cannot swallow SIKLOS tablets".
Step 9: Throw away the damp disposable paper towel in the trash. Pull off disposable gloves and throw away in the trash.
Wash and dry your hands.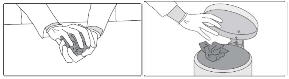
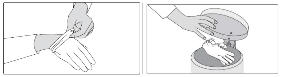
Step 10: Store any unused broken tablet in the bottle and put the bottle back in the box. Broken tablets must be used within three months. For people who cannot swallow SIKLOS tablets You will need the following supplies to prepare and take your dose by dissolving the tablet: - Your bottle of SIKLOS 1,000 mg tablets or SIKLOS 100 mg tablets
- Note: If you have been prescribed SIKLOS 1,000 mg and need to break your tablets, use the SIKLOS 1,000 mg Tablet Breaking Instructions above to get your prescribed dose before you begin the steps below.
- A teaspoon
- Water to dissolve tablets
Step 1: Get your prescribed dose of SIKLOS tablets. Put your prescribed dose of SIKLOS tablets onto the teaspoon. 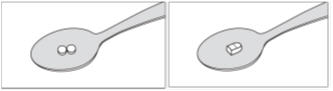
SIKLOS 100 mg tablet SIKLOS 1,000 mg tablet (broken)
Step 2: Add a small amount of water to the teaspoon. The tablet dissolves within about 1 minute. 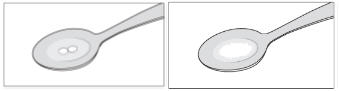
SIKLOS 100 mg tablet
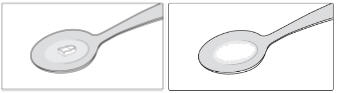
SIKLOS 1,000 mg tablet (broken)
Step 3: Swallow the mixture right away. 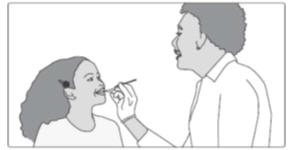
Step 4: After you take your prescribed dose of SIKLOS tablets, drink a glass of water. When you are finished drinking a glass of water, continue to Step 9 and Step 10 above. 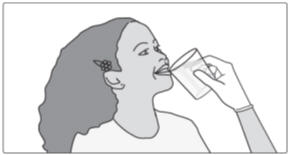
Storing your SIKLOS tablets:
- Store SIKLOS at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep the SIKLOS bottle tightly closed.
Keep SIKLOS and all medicines out of the reach of children.
Distributed by: MEDUNIK USA.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Issued: 05/2018Special form to illustrate daily dose and how to handle tablets
-
PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Carton
NDC: 71770-100-60
Siklos® 100 mg
(hydroxyurea)
tablets100 mg per tablet
ATTENTION
PHARMACIST:
Each patient is
required to receive
the enclosed
Medication Guide60 tablets
addmedica

-
PRINCIPAL DISPLAY PANEL - 1,000 mg Tablet Bottle Carton
NDC: 71770-120-30
Siklos® 1,000 mg
(hydroxyurea)
tablets1,000 mg per tablet
ATTENTION
PHARMACIST:
Each patient is
required to receive
the enclosed
Medication Guide30 tablets
addmedica

-
INGREDIENTS AND APPEARANCE
SIKLOS
hydroxyurea tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71770-100 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength hydroxyurea (UNII: X6Q56QN5QC) (hydroxyurea - UNII:X6Q56QN5QC) hydroxyurea 100 mg Inactive Ingredients Ingredient Name Strength Microcrystalline Cellulose (UNII: OP1R32D61U) Silicon dioxide (UNII: ETJ7Z6XBU4) Sodium stearyl fumarate (UNII: 7CV7WJK4UI) DIMETHYLAMINOETHYL METHACRYLATE - BUTYL METHACRYLATE - METHYL METHACRYLATE COPOLYMER (UNII: 905HNO1SIH) Isopropyl alcohol (UNII: ND2M416302) Product Characteristics Color WHITE (white to off-white) Score no score Shape ROUND (biconvex) Size 6mm Flavor Imprint Code 100 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71770-100-60 1 in 1 CARTON 02/06/2018 1 60 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208843 02/06/2018 SIKLOS
hydroxyurea tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71770-120 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength hydroxyurea (UNII: X6Q56QN5QC) (hydroxyurea - UNII:X6Q56QN5QC) hydroxyurea 1000 mg Inactive Ingredients Ingredient Name Strength Microcrystalline Cellulose (UNII: OP1R32D61U) Silicon dioxide (UNII: ETJ7Z6XBU4) Sodium stearyl fumarate (UNII: 7CV7WJK4UI) DIMETHYLAMINOETHYL METHACRYLATE - BUTYL METHACRYLATE - METHYL METHACRYLATE COPOLYMER (UNII: 905HNO1SIH) Isopropyl alcohol (UNII: ND2M416302) Product Characteristics Color WHITE (white to off-white) Score 4 pieces Shape OVAL (capsule-shaped) Size 22mm Flavor Imprint Code T Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71770-120-30 1 in 1 CARTON 02/06/2018 1 30 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208843 02/06/2018 Labeler - Medunik (080318531) Registrant - Addmedica (280967790)
Trademark Results [Siklos]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 SIKLOS 86229715 4887561 Live/Registered |
ADDMEDICA 2014-03-24 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.