PROHANCE- gadoteridol injection, solution
ProHance by
Drug Labeling and Warnings
ProHance by is a Prescription medication manufactured, distributed, or labeled by BRACCO DIAGNOSTICS INC, BRACCO IMAGING SPA, BIPSO GmbH, Labor LS SE & Co. KG, BioChem Labor für biologishe und chemische Analytik GmbH. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
BOXED WARNING
(What is this?)
WARNING: NEPHROGENIC SYSTEMIC FIBROSIS
Gadolinium-based contrast agents (GBCAs) increase the risk for NSF among patients with impaired elimination of the drugs. Avoid use of GBCAs in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating systemic fibrosis affecting the skin, muscle and internal organs.
-
The risk for NSF appears highest among patients with:
- chronic, severe kidney disease (GFR <30 mL/min/1.73m2), or
- acute kidney injury.
- Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (e.g. age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing.
- For patients at highest risk for NSF, do not exceed the recommended ProHance dose and allow a sufficient period of time for elimination of the drug from the body prior to re-administration (see WARNINGS).
-
The risk for NSF appears highest among patients with:
-
DESCRIPTION
ProHance (Gadoteridol) Injection is a nonionic contrast medium for magnetic resonance imaging (MRI), available as a 0.5M sterile clear colorless to slightly yellow aqueous solution in vials and syringes for intravenous injection.
Gadoteridol is the gadolinium complex of 10-(2-hydroxy-propyl)-1,4,7,10- tetraazacyclododecane-1,4,7-triacetic acid with a molecular weight of 558.7, an empirical formula of C17H29N4O7Gd and has the following structural formula:
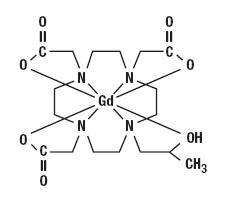
Each mL of ProHance contains 279.3 mg gadoteridol, 0.23 mg calteridol calcium, 1.21 mg tromethamine and water for injection. ProHance contains no antimicrobial preservative.
ProHance has a pH of 6.5 to 8.0. Pertinent physicochemical data are noted below:
PARAMETER Osmolality (mOsmol/kg water) @ 37° C 630 Viscosity (cP) @ 20° C 2.0 @ 37° C 1.3 Specific Gravity @ 25° C 1.140 Density (g/mL) @ 25° C 1.137 Octanol: H2O coefficient -3.68 ± 0.02 ProHance has an osmolality 2.2 times that of plasma (285 mOsmol/kg water) and is hypertonic under conditions of use.
-
CLINICAL PHARMACOLOGY
Pharmacokinetics
The pharmacokinetics of intravenously administered gadoteridol in normal subjects conforms to a two-compartment open model with mean distribution and elimination half-lives (reported as mean ± SD) of about 0.20 ± 0.04 hours and 1.57 ± 0.08 hours, respectively.
Gadoteridol is eliminated in the urine with 94.4 ± 4.8% (mean ± SD) of the dose excreted within 24 hours post-injection. It is unknown if biotransformation or decomposition of gadoteridol occur in vivo.
The renal and plasma clearance rates (1.41 ± 0.33 mL/ min/kg and 1.50 ± 0.35 mL/ min/kg, respectively) of gadoteridol are essentially identical, indicating no alteration in elimination kinetics on passage through the kidneys and that the drug is essentially cleared through the kidney. The volume of distribution (204 ± 58 mL/kg) is equal to that of extracellular water, and clearance is similar to that of substances which are subject to glomerular filtration. Following GBCA administration, gadolinium is present for months or years in brain, bone, skin, and other organs (See WARNINGS- Gadolinium Retention).
It is unknown if protein binding of ProHance occurs in vivo.
Pharmacodynamics
Gadoteridol is a paramagnetic agent and, as such, develops a magnetic moment when placed in a magnetic field. The relatively large magnetic moment produced by the paramagnetic agent results in a relatively large local magnetic field, which can enhance the relaxation rates of water protons in the vicinity of the paramagnetic agent.
In magnetic resonance imaging (MRI), visualization of normal and pathologic brain tissue depends in part on variations in the radiofrequency signal intensity that occur with 1) differences in proton density; 2) differences of the spin-lattice or longitudinal relaxation times (T1); and 3) differences in the spin-spin or transverse relaxation time (T2). When placed in a magnetic field, gadoteridol decreases T1 relaxation times in the target tissues. At recommended doses, the effect is observed with greatest sensitivity in the T1-weighted sequences.
Disruption of the blood-brain barrier or abnormal vascularity allows accumulation of gadoteridol in lesions such as neoplasms, abscesses, and subacute infarcts. The pharmacokinetics of ProHance in various lesions is not known.
-
CLINICAL TRIALS
ProHance was evaluated in two blinded read trials in a total of 133 adults who had an indication for head and neck extracranial or extraspinal magnetic resonance imaging. These 133 adults (74 men, 59 women) had a mean age of 53 with a range of 19 to 76 years. Of these patients, 85% were Caucasian, 13% Black, 2% Asian, and < 1% other. The results of the non-contrast and gadoteridol MRI scans were compared. In this database, approximately 75-82% of the scans were enhanced. 45-48% of the scans provided additional diagnostic information, and 8-25% of the diagnoses were changed. The relevance of the findings to disease sensitivity and specificity has not been fully evaluated.
ProHance was evaluated in a multicenter clinical trial of 103 children who had an indication for a brain or spine MRI. These 103 children, (54 boys and 49 girls) had a mean age of 8.7 years with an age range of 2 to 20 years. Of these 103 children, 54 were between 2 and 12 years of age. Also, of these 103 children, 74% were Caucasian, 11% Black, 12% Hispanic, 2% Asian, and 2% other. The results of the non-contrast and gadoteridol MRI scans were compared. ProHance was given in one single 0.1 mmol/kg dose. Repeat dosing was not studied. In this database, MRI enhancement was noted in approximately 60% of the scans and additional diagnostic information in 30-95% of the scans.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
ProHance is contraindicated in patients with known allergic or hypersensitivity reactions to ProHance (see WARNINGS).
-
WARNINGS
Nephrogenic Systemic Fibrosis (NSF)
Gadolinium-based contrast agents (GBCAs) increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of GBCAs among these patients unless the diagnostic information is essential and not available with non-contrast enhanced MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR <30 mL/min/1.73m2) as well as patients with acute kidney injury. The risk appears lower for patients with chronic, moderate kidney disease (GFR 30-59 mL/min/1.73m2) and little, if any, for patients with chronic, mild kidney disease (GFR 60-89 mL/min/1.73m2). NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. Report any diagnosis of NSF following ProHance administration to Bracco Diagnostics (1-800-257-5181) or FDA (1-800-FDA-1088 or www.fda.gov/medwatch).
Screen patients for acute kidney injury and other conditions that may reduce renal function. Features of acute kidney injury consist of rapid (over hours to days) and usually reversible decrease in kidney function, commonly in the setting of surgery, severe infection, injury or drug-induced kidney toxicity. Serum creatinine levels and estimated GFR may not reliably assess renal function in the setting of acute kidney injury. For patients at risk for chronically reduced renal function (e.g., age > 60 years, diabetes mellitus or chronic hypertension), estimate the GFR through laboratory testing.
Among the factors that may increase the risk for NSF are repeated or higher than recommended doses of a GBCA and the degree of renal impairment at the time of exposure. Record the specific GBCA and the dose administered to a patient. For patients at highest risk for NSF, do not exceed the recommended ProHance dose and allow a sufficient period of time for elimination of the drug prior to re-administration. For patients receiving hemodialysis, physicians may consider the prompt initiation of hemodialysis following the administration of a GBCA in order to enhance the contrast agent’s elimination. The usefulness of hemodialysis in the prevention of NSF is unknown (see CLINICAL PHARMACOLOGY and DOSAGE AND ADMINISTRATION).
Acute Kidney Injury (AKI)
In patients with chronically reduced renal function, acute kidney injury requiring dialysis has occurred with the use of GBCAs. The risk of acute kidney injury may increase with increasing dose of the contrast agent; administer the lowest dose necessary for adequate imaging.
Hypersensitivity Reactions
Severe and fatal hypersensitivity reactions including anaphylaxis have been observed with administration of gadolinium products, including ProHance. Patients with a history of allergy, drug reactions or other hypersensitivity-like disorders should be closely observed during the procedure and for several hours after drug administration. If a reaction occurs, stop ProHance and immediately begin appropriate therapy including resuscitation. (See PRECAUTIONS - General)
Deoxygenated sickle erythrocytes have been shown in in vitro studies to align perpendicular to a magnetic field which may result in vaso-occlusive complications in vivo. The enhancement of magnetic moment by ProHance may possibly potentiate sickle erythrocyte alignment. ProHance in patients with sickle cell anemia and other hemoglobinopathies has not been studied.
Patients with other hemolytic anemias have not been adequately evaluated following administration of ProHance to exclude the possibility of increased hemolysis.
Gadolinium Retention
Gadolinium is retained for months or years in several organs. The highest concentrations (nanomoles per gram of tissue) have been identified in the bone, followed by other organs (e.g. brain, skin, kidney, liver, and spleen). The duration of retention also varies by tissue and is longest in bone. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, gadolinium retention varies among the linear agents with Omniscan (gadodiamide) and Optimark (gadoversetamide) causing greater retention than other linear agents [Eovist (gadoxetate disodium), Magnevist (gadopentetate dimeglumine), MultiHance (gadobenate dimeglumine)]. Retention is lowest and similar among the macrocyclic GBCAs [Dotarem (gadoterate meglumine), Gadavist (gadobutrol), ProHance (gadoteridol)].
Consequences of gadolinium retention in the brain have not been established. Pathologic and clinical consequences of GBCA administration and retention in skin and other organs have been established in patients with impaired renal function (See WARNINGS-Nephrogenic Systemic Fibrosis). There are rare reports of pathologic skin changes in patients with normal renal function. Adverse events involving multiple organ systems have been reported in patients with normal renal function without an established causal link to gadolinium retention (See ADVERSE REACTIONS).
While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients, and patients with inflammatory conditions. Consider the retention characteristics of the agent when choosing a GBCA for these patients. Minimize repetitive GBCA imaging studies, particularly closely spaced studies when possible.
-
PRECAUTIONS
General
Diagnostic procedures that involve the use of contrast agents should be carried out under direction of a physician with the prerequisite training and a thorough knowledge of the procedure to be performed.
Personnel trained in resuscitation techniques and resuscitation equipment should be available.
The possibility of a reaction, including serious, life threatening, or fatal, anaphylactic or cardiovascular reactions, or other idiosyncratic reactions (see ADVERSE REACTIONS), should always be considered, especially in those patients with a history of a known clinical hypersensitivity or a history of asthma or other allergic respiratory disorders.
Gadoteridol is cleared from the body by glomerular filtration. The hepato-biliary enteric pathway of excretion has not been demonstrated with ProHance. Dose adjustments in renal or hepatic impairment have not been studied. Therefore, caution should be exercised in patients with either renal or hepatic impairment.
In a patient with a history of grand mal seizure, the possibility to induce such a seizure by ProHance is unknown.
When ProHance (Gadoteridol) Injection is to be injected using nondisposable equipment, scrupulous care should be taken to prevent residual contamination with traces of cleansing agents. After ProHance is drawn into a syringe, the solution should be used immediately.
Repeat Procedures: Repeated procedures have not been studied. Sequential use during the same diagnostic session has only been studied in central nervous system use. (See Pharmacokinetics under CLINICAL PHARMACOLOGY and Central Nervous System under DOSAGE AND ADMINISTRATION).
Information for patients:
General: Patients scheduled to receive ProHance should be instructed to inform their physician if the patient;
- is pregnant or breast feeding
- has anemia or diseases that affect the red blood cells
- has a history of renal or hepatic disease, seizure, hemoglobinopathies, asthma or allergic respiratory diseases
- has recently received a GBCA.
Nephrogenic Systemic Fibrosis: GBCAs increase the risk for NSF among patients with impaired elimination of the drugs. To counsel patients at risk for NSF:
- Describe the clinical manifestations of NSF
- Describe procedures to screen for the detection of renal impairment
Instruct the patients to contact their physician if they develop signs or symptoms of NSF following ProHance administration, such as burning, itching, swelling, scaling, hardening and tightening of the skin; red or dark patches on the skin; stiffness in joints with trouble moving, bending or straightening the arms, hands, legs or feet; pain in the hip bones or ribs; or muscle weakness.
Gadolinium Retention: Advise patients that gadolinium is retained for months or years in brain, bone, skin, and other organs in patients with normal renal function. The clinical consequences of retention are unknown. Retention depends on multiple factors and is greater following administration of linear GBCAs than following administration of macrocyclic GBCAs. (See WARNINGS-Gadolinium Retention).
-
CARCINOGENESIS, MUTAGENESIS,
AND IMPAIRMENT OF FERTILITY
No animal studies have been performed to evaluate the carcinogenic potential of gadoteridol or potential effects on fertility.
ProHance did not demonstrate genotoxic activity in bacterial reverse mutation assays using Salmonella typhimurium and Escherichia coli, in a mouse lymphoma forward mutation assay, in an in vitro cytogenetic assay measuring chromosomal aberration frequencies in Chinese hamster ovary cells, nor in an in vivo mouse micronucleus assay at intravenous doses up to 5.0 mmol/kg.
Pregnancy
GBCAs cross the placenta and result in fetal exposure and gadolinium retention. The human data on the association between GBCAs and adverse fetal outcomes are limited and inconclusive. Because of the potential risks of gadolinium to the fetus, use ProHance only if imaging is essential during pregnancy and cannot be delayed.
Contrast enhancement is visualized in the human placenta and fetal tissues after maternal GBCA administration.
Cohort studies and case reports on exposure to GBCAs during pregnancy have not reported a clear association between GBCAs and adverse effects in the exposed neonates. However, a retrospective cohort study, comparing pregnant women who had a GBCA MRI to pregnant women who did not have an MRI, reported a higher occurrence of stillbirths and neonatal deaths in the group receiving GBCA MRI. Limitations of this study include a lack of comparison with non-contrast MRI and lack of information about the maternal indication for MRI. Overall, these data preclude a reliable evaluation of the potential risk of adverse fetal outcomes with the use of GBCAs in pregnancy.
GBCAs administered to pregnant non-human primates (0.1 mmol/kg on gestational days 85 and 135) result in measurable gadolinium concentration in the offspring in bone, brain, skin, liver, kidney, and spleen for at least 7 months. GBCAs administered to pregnant mice (2 mmol/kg daily on gestational days 16 through 19) result in measurable gadolinium concentrations in the pups in bone, brain, kidney, liver, blood, muscle, and spleen at one month postnatal age.
ProHance administered to rats at 10 mmol/kg/day (33 times the maximum recommended human dose of 0.3 mmol/kg or 6 times the human dose based on a mmol/m2 comparison) for 12 days during gestation doubled the incidence of postimplantation loss. When rats were administered 6.0 or 10.0 mmol/ kg/day for 12 days, an increase in spontaneous locomotor activity was observed in the offspring. ProHance increased the incidence of spontaneous abortion and early delivery in rabbits administered 6 mmol/ kg/day (20 times the maximum recommended human dose or 7 times the human dose based on a mmol/m2 comparison) for 13 days during gestation.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when ProHance is administered to a nursing woman.
Pediatric Use
Safety and efficacy in children under the age of 2 years have not been established. The safety and efficacy of doses > 0.1 mmol/kg; and sequential and/or repeat procedures has not been studied in children. (See INDICATIONS AND USAGE and DOSAGE AND ADMINISTRATION sections)
-
ADVERSE REACTIONS
The adverse events described in this section were observed in clinical trials involving 1251 patients (670 males and 581 females). Adult patients ranged in age from 18-91 yrs. Pediatric patients ranged from 2-17 years. The racial breakdown was 83% Caucasian, 8% Black, 3% Hispanic, 2% Asian, and 1% other. In 2% of the patients, race was not reported.
The most commonly noted adverse experiences were nausea and taste perversion with an incidence of 1.4%. These events were mild to moderate in severity.
The following additional adverse events occurred in fewer than 1% of the patients:
Body as a Whole: Facial Edema; Neck Rigidity; Pain; Pain at Injection Site; Injection Site Reaction; Chest Pain; Headache; Fever; Itching; Watery Eyes; Abdominal Cramps; Tingling Sensation in Throat; Laryngismus; Flushed Feeling; Vasovagal Reaction; Anaphylactoid Reactions (characterized by cardiovascular, respiratory and cutaneous symptoms) Cardiovascular: Prolonged P-R Interval; Hypotension; Elevated Heart Rate; A-V Nodal Rhythm Digestive: Edematous and/or itching tongue; Gingivitis; Dry Mouth; Loose Bowel; Vomiting Nervous System: Anxiety; Dizziness; Paresthesia; Mental Status Decline; Loss of Coordination in Arm; Staring Episode; Seizure; Syncope Respiratory System: Dyspnea; Rhinitis; Cough Skin and Appendages: Pruritus; Rash; Rash Macular Papular; Urticaria; Hives; Tingling Sensation of Extremity and Digits Special Senses: Tinnitus The following adverse drug reactions have also been reported:
General Disorders and Administration Site Conditions: Adverse events with variable onset and duration have been reported after GBCA administration (See WARNINGS-Gadolinium Retention). These include fatigue, asthenia, pain syndromes, and heterogeneous clusters of symptoms in the neurological, cutaneous, and musculoskeletal systems. Body as a Whole: Generalized Edema; Laryngeal Edema; Malaise; Anaphylactoid Reactions (characterized by cardiovascular, respiratory and cutaneous symptoms, and rarely resulting in Death). Cardiovascular: Cardiac Arrest; Bradycardia; Hypertension; and Death in association with pre-existing cardiovascular disorders. Digestive: Increased Salivation; Dysphagia Nervous System: Stupor; Tremor; Loss of Consciousness Respiratory: Apnea; Wheezing Skin and Appendages: Gadolinium associated plaques, Sweating; and Cyanosis Special Senses: Voice Alteration; Transitory Deafness Urogenital: Urinary Incontinence - OVERDOSAGE
-
DOSAGE AND ADMINISTRATION
Central Nervous System
ADULTS: The recommended dose of ProHance (Gadoteridol) Injection is 0.1 mmol/kg (0.2 mL/kg) administered as a rapid intravenous infusion (10 mL/min-60 mL/min) or bolus (> 60 mL/min). In patients with normal renal function suspected of having poorly enhancing lesions, in the presence of negative or equivocal scans, a supplementary dose of 0.2 mmol/kg (0.4 mL/kg) may be given up to 30 minutes after the first dose.
CHILDREN (2-18 years): The recommended dose of ProHance is 0.1 mmol/kg (0.2 mL/kg) administered as a rapid intravenous infusion (10 mL/min-60 mL/min) or bolus (> 60 mL/min).
The safety and efficacy of doses > 0.1 mmol/kg, and sequential and/or repeat procedures has not been studied.
Extracranial/Extraspinal Tissues
ADULTS: The recommended dose of ProHance is 0.1 mmol/kg (0.2 mL/kg) administered as a rapid intravenous infusion (10 mL/min-60 mL/min) or bolus (> 60 mL/min).
CHILDREN: Safety and efficacy for extracranial/extra-spinal tissues has not been established.
Dose adjustments in renal and liver impairment have not been studied.To ensure complete injection of the contrast medium, the injection should be followed by a 5 mL normal saline flush. The imaging procedure should be completed within 1 hour of the first injection of ProHance (Gadoteridol) Injection.
Parenteral products should be inspected visually for particulate matter and discoloration prior to administration. Do not use the solution if it is discolored or particulate matter is present.
Any unused portion must be discarded in accordance with regulations dealing with the disposal of such materials.
-
HOW SUPPLIED
ProHance (Gadoteridol) Injection is a clear, colorless to slightly yellow solution containing 279.3 mg/mL of gadoteridol in rubber stoppered vials. ProHance is available in boxes of:
Five 5 mL fills in single dose 15 mL vials (NDC: 0270-1111-04) Five 10 mL fills in single dose 30 mL vials (NDC: 0270-1111-01) Five 15 mL fills in single dose 30 mL vials (NDC: 0270-1111-02) Five 20 mL fills in single dose 30 mL vials (NDC: 0270-1111-03) Five 10 mL fills in single dose 20 mL prefilled syringes (NDC: 0270-1111-16) Five 17 mL fills in single dose 20 mL prefilled syringes (NDC: 0270-1111-45) -
STORAGE
ProHance (Gadoteridol) Injection should be stored at 25°C (77°F) excursions permitted to 15-30°C (59-86°F) [See USP Controlled Room Temperature]. Protect from light. DO NOT FREEZE. Should freezing occur in the vial, ProHance should be brought to room temperature before use. If allowed to stand at room temperature for a minimum of 60 minutes, ProHance (Gadoteridol) Injection should return to a clear, colorless to slightly yellow solution. Before use, examine the product to assure that all solids are redissolved and that the container and closure have not been damaged. Should solids persist, discard vial. Frozen syringes should be discarded.
Directions for Use of the ProHance® (Gadoteridol) Injection single dose syringe*
- 1) Screw the threaded tip of the plunger rod clockwise into the cartridge plunger and push forward a few millimeters to break any friction between the cartridge plunger and syringe barrel.
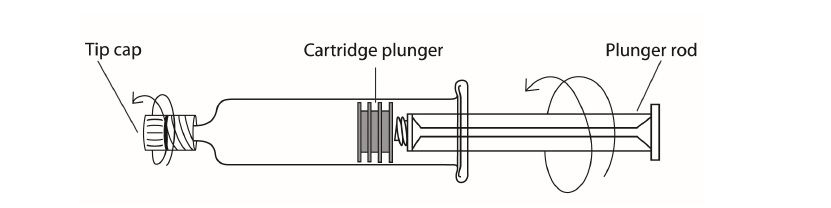
- 2) Holding syringe erect, unscrew the plastic tip cap from the tip of the syringe and attach either a sterile, disposable needle or tubing with a compatible luer lock using a push-twist action.
- 3) Hold the syringe erect and push plunger forward until all of the air is evacuated and fluid either appears at the tip of the needle or the tubing is filled. Following the usual aspiration procedure, complete the injection. To ensure complete delivery of the contrast medium, the injection should be followed by a normal saline flush.
- 4) Properly dispose of the syringe and any other materials used.
*The syringe assembly is a HYPAK SCF® single dose syringe supplied by Becton Dickinson.
- 1) Screw the threaded tip of the plunger rod clockwise into the cartridge plunger and push forward a few millimeters to break any friction between the cartridge plunger and syringe barrel.
- SPL UNCLASSIFIED SECTION
-
SPL UNCLASSIFIED SECTION
Manufactured for
Bracco Diagnostics Inc.
Monroe Township, NJ 08543
by BIPSO GmbH
78224 Singen (Germany)This Medication Guide has been approved by the U.S. Food and Drug Administration Issued: 04/2018
COEB503MEDICATION GUIDE
PROHANCE®(prō-ˈhan(t)s)
(Gadoteridol)
Injection for intravenous useWhat is PROHANCE? - PROHANCE is a prescription medicine called a gadolinium-based contrast agent (GBCA). PROHANCE, like other GBCAs, is used with a magnetic resonance imaging (MRI) scanner.
- An MRI exam with a GBCA, including PROHANCE, helps your doctor to see problems better than an MRI exam without a GBCA.
- Your doctor has reviewed your medical records and has determined that you would benefit from using a GBCA with your MRI exam.
What is the most important information I should know about PROHANCE? - PROHANCE contains a metal called gadolinium. Small amounts of gadolinium can stay in your body including the brain, bones, skin and other parts of your body for a long time (several months to years).
- It is not known how gadolinium may affect you, but so far, studies have not found harmful effects in patients with normal kidneys.
- Rarely, patients have reported pains, tiredness, and skin, muscle or bone ailments for a long time, but these symptoms have not been directly linked to gadolinium.
- There are different GBCAs that can be used for your MRI exam. The amount of gadolinium that stays in the body is different for different gadolinium medicines. Gadolinium stays in the body more after Omniscan or Optimark than after Eovist, Magnevist, or MultiHance. Gadolinium stays in the body the least after Dotarem, Gadavist, or ProHance.
- People who get many doses of gadolinium medicines, women who are pregnant and young children may be at increased risk from gadolinium staying in the body.
- Some people with kidney problems who get gadolinium medicines can develop a condition with severe thickening of the skin, muscles and other organs in the body (nephrogenic systemic fibrosis). Your healthcare provider should screen you to see how well your kidneys are working before you receive PROHANCE.
Do not receive PROHANCE if you have had a severe allergic reaction to PROHANCE. Before receiving PROHANCE, tell your healthcare provider about all your medical conditions, including if you: - have had any MRI procedures in the past where you received a GBCA. Your healthcare provider may ask you for more information including the dates of these MRI procedures.
- are pregnant or plan to become pregnant. It is not known if PROHANCE can harm your unborn baby. Talk to your healthcare provider about the possible risks to an unborn baby if a GBCA such as PROHANCE is received during pregnancy
- have kidney problems, diabetes, or high blood pressure
- have had an allergic reaction to dyes (contrast agents) including GBCAs
What are the possible side effects of PROHANCE? - See “What is the most important information I should know about PROHANCE?”
- Allergic reactions. PROHANCE can cause allergic reactions that can sometimes be serious. Your healthcare provider will monitor you closely for symptoms of an allergic reaction.
These are not all the possible side effects of PROHANCE.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.General information about the safe and effective use of PROHANCE.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your healthcare provider for information about PROHANCE that is written for health professionals.What are the ingredients in PROHANCE?
Active ingredient: gadoteridol
Inactive ingredients: calteridol calcium, tromethamine
Manufactured by: BIPSO GmbH-78224 Singen (Germany)
Manufactured for: Bracco Diagnostics Inc., Monroe Township, NJ 08831
US Patent No. 5,474,756; 5,846,519; and 6,143,274.
For more information, go to www.imaging.bracco.com or call 1-800-257-5181. - PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PROHANCE
gadoteridol injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0270-1111 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength gadoteridol (UNII: 0199MV609F) (gadoteridol - UNII:0199MV609F) gadoteridol 279.3 mg in 1 mL Inactive Ingredients Ingredient Name Strength calteridol calcium (UNII: RPH56VWA1A) .23 mg in 1 mL tromethamine (UNII: 023C2WHX2V) 1.21 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0270-1111-04 5 in 1 BOX 11/16/1992 1 5 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product 2 NDC: 0270-1111-01 5 in 1 BOX 11/16/1992 2 10 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product 3 NDC: 0270-1111-02 5 in 1 BOX 11/16/1992 3 15 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product 4 NDC: 0270-1111-03 5 in 1 BOX 11/16/1992 4 20 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product 5 NDC: 0270-1111-16 5 in 1 BOX 11/16/1992 5 10 mL in 1 SYRINGE, GLASS; Type 0: Not a Combination Product 6 NDC: 0270-1111-45 5 in 1 BOX 11/16/1992 6 17 mL in 1 SYRINGE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020131 11/16/1992 Labeler - BRACCO DIAGNOSTICS INC (849234661) Registrant - BRACCO DIAGNOSTICS INC (849234661) Establishment Name Address ID/FEI Business Operations BRACCO IMAGING SPA 434384007 API MANUFACTURE(0270-1111) Establishment Name Address ID/FEI Business Operations BIPSO GmbH 342104149 MANUFACTURE(0270-1111) , ANALYSIS(0270-1111) Establishment Name Address ID/FEI Business Operations Labor LS SE & Co. KG 314929072 ANALYSIS(0270-1111)
Trademark Results [ProHance]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 PROHANCE 97264281 not registered Live/Pending |
JAMOCHA TECH PRIVATE LIMITED 2022-02-11 |
 PROHANCE 97182054 not registered Live/Pending |
JAMOCHA TECH PRIVATE LIMITED 2021-12-21 |
 PROHANCE 75572308 not registered Dead/Abandoned |
United Agri Products 1998-10-19 |
 PROHANCE 74392033 1820972 Live/Registered |
BRACCO DIAGNOSTICS INC. 1993-05-19 |
 PROHANCE 74222866 not registered Dead/Abandoned |
E. R. Squibb & Sons, Inc. 1991-11-14 |
 PROHANCE 74032581 not registered Dead/Abandoned |
E. R. Squibb & Sons, Inc. 1990-02-26 |
 PROHANCE 73809193 1582205 Live/Registered |
E. R. SQUIBB & SONS, INC. 1989-06-26 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

