ACTIDOM DA- chlorpheniramine maleate, phenylephrine hydrochloride solution
ACTIDOM DA by
Drug Labeling and Warnings
ACTIDOM DA by is a Otc medication manufactured, distributed, or labeled by ACTIPHARMA, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
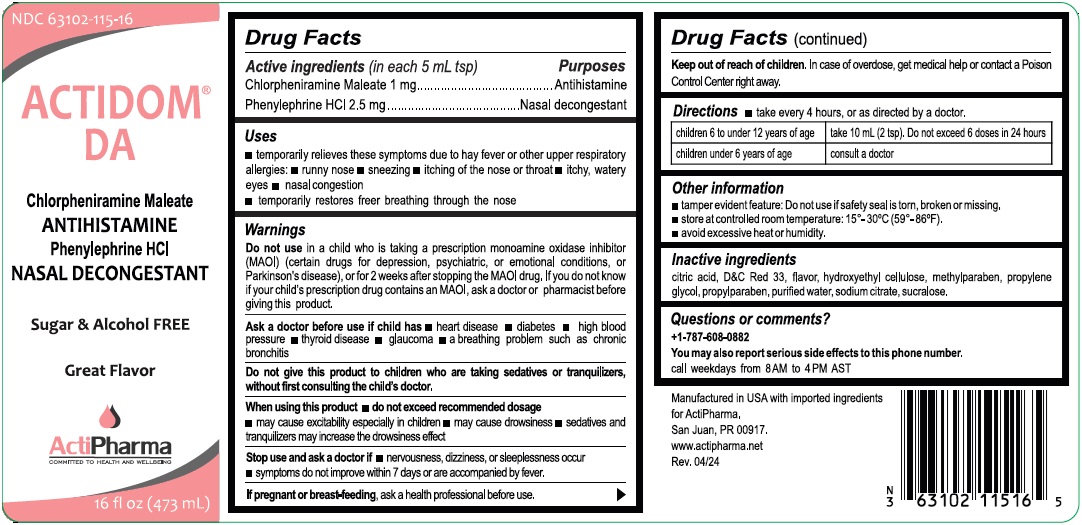
- Drug Facts
- Active ingredients (in each 5 mL tsp)
- Purposes
- Uses
-
Warnings
Do not use in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your child's prescription drug contains an MAOI, ask a doctor or pharmacist before giving this product.
Ask a doctor before use if child has heart disease diabetes high blood pressure thyroid disease glaucoma a breathing problem such as chronic bronchitis
Do not give this product to children who are taking sedatives or tranquilizers, without first consulting the child's doctor.
When using this product do not exceed recommended dosage
may cause excitability especially in children may cause drowsiness sedatives and tranquilizers may increase the drowsiness effectStop use and ask a doctor if nervousness, dizziness, or sleeplessness occur
symptoms do not improve within 7 days or are accompanied by fever.If pregnant or breast-feeding, ask a health professional before use.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- Packaging
-
INGREDIENTS AND APPEARANCE
ACTIDOM DA
chlorpheniramine maleate, phenylephrine hydrochloride solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 63102-115 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 1 mg in 5 mL PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 2.5 mg in 5 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) D&C RED NO. 33 (UNII: 9DBA0SBB0L) HYDROXYETHYL CELLULOSE, UNSPECIFIED (UNII: T4V6TWG28D) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color pink (LIGHT PINK) Score Shape Size Flavor BANANA, STRAWBERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 63102-115-16 474 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 01/20/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 01/20/2022 Labeler - ACTIPHARMA, LLC (079340948)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.