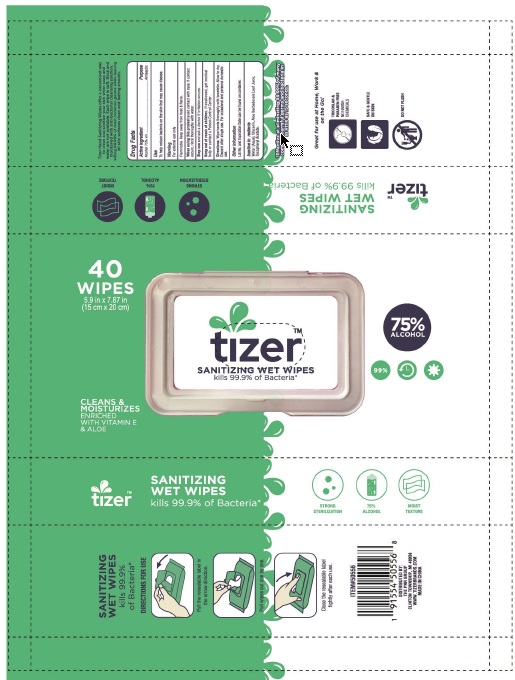
Tizer sanitizing wet wipes by Zhejiang Meizhiyuan Cosmetics Co.,Ltd
Tizer sanitizing wet wipes by
Drug Labeling and Warnings
Tizer sanitizing wet wipes by is a Otc medication manufactured, distributed, or labeled by Zhejiang Meizhiyuan Cosmetics Co.,Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
TIZER SANITIZING WET WIPES- alcohol cloth
Zhejiang Meizhiyuan Cosmetics Co.,Ltd
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
When using this product avoid contact with eyes. If contact occurs, rinse thoroughly with water.
Stop use and ask a doctor if irritation occurs.
| TIZER SANITIZING WET WIPES
alcohol cloth |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Zhejiang Meizhiyuan Cosmetics Co.,Ltd (543036597) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Zhejiang Meizhiyuan Cosmetics Co.,Ltd | 543036597 | manufacture(80204-036) | |
Revised: 12/2022
Document Id: 765337c5-a78c-4c96-b3ba-55639f25d295
Set id: 781b41ad-3f06-4b95-a56a-315bdb5d713f
Version: 5
Effective Time: 20221203