ANTISEPTIC SKIN CLEANSER- chlorhexidine gluconate 4% solution
Antiseptic Skin Cleanser by
Drug Labeling and Warnings
Antiseptic Skin Cleanser by is a Otc medication manufactured, distributed, or labeled by Henry Schein, Inc., Xttrium Laboratories, Inc., Xttrium Laboratories. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
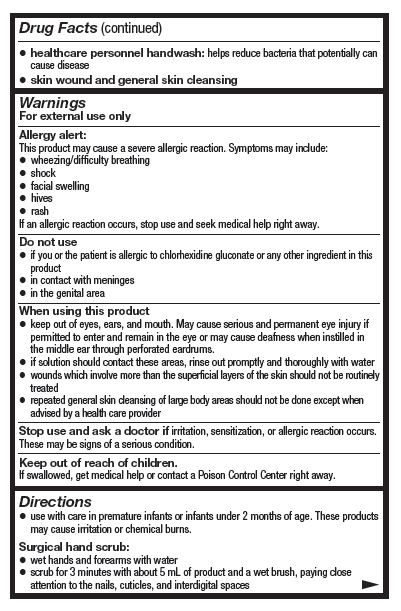
- Active ingredient
- Purpose
- Uses
- Warnings
- Do not use
-
When using this product
- keep out of eyes, ears, and mouth. May cause serious and permanent eye injury if permitted to enter and remain in the eye or may cause deafness when instilled in the middle ear through perforated eardrums.
- if solution should contact these areas, rinse out promptly and thoroughly with water
- wounds which involve more than the superficial layers of the skin should not be routinely treated
- repeated general skin cleansing of large body areas should not be done except when advised by a health care provider
- Stop use and ask a doctor if
- Keep out of reach of children.
-
Directions
- use with care in premature infants or infants under 2 months of age. These products may cause irritation or chemical burns.
Surgical hand scrub:
- wet hands and forwarms with water
- scrub for 3 minutes with about 5 mL of product and a wet brush, paying close attention to the nails, cuticles, and interdigital spaces
- a seperate nail cleaner may be used
- rinse thoroughly
- wash for an additional 3 minutes with 5 mL of product and rinse under running water
- dry thoroughly
Healthcare personnel handwash:
- wet hands with water
- dispense about 5 mL of product into cupped hands and wash in a vigorous manner for 15 seconds
- rinse and dry thoroughly
Skin wound and general skin cleansing:
- thoroughly rinse the area to be cleaned with water
- apply the minimum amount of product necessary to cover the skin or wound area and wash gently
- rinse again thoroughly
- Other information
- Inactive ingredients
- Questions or comments?
-
OTHER SAFETY INFORMATION
Laundering/Cleaning Instructions: Chlorhexidine gluconate skin cleansers will cause stains if used with chlorine releasing products, such as chlorine bleach. Rinse completely and use only non-chlorine detergents.
WARNING: This product can expose you to coconut oil diethanolamine condensate (cocamide diethanolamine), which is known to the State of California to cause cancer. For more information go to www.P65Warnings.ca.gov
Rev 7/2018
- Active ingredient
- Purpose
- Uses
- Warnings
- Do not use
-
When using this product
- keep out of eyes, ears, and mouth. May cause serious and permanent eye injury if permitted to enter and remain in the eye or may cuase deafness when instilled in the middle ear through perforated eardrums.
- if solution should contact these areas, rinse out promptly and thoroughly with water
- wounds which involve more than the superficial layers of the skin should not be routinely treated
- repeated general skin cleansing of large body areas should not be done except when advised by a health care provider
- Stop use and ask a doctor if
- Keeo out of reach of children.
-
Directions
- use with care in premature infants or infants under 2 months of age. These products may cause irritation or chemical burns.
Surgical hand scrub:
- wet hands and forearms with water
- scrub for 3 minutes with about 5 mL of product and a wet brush, paying close attention to the nails, cuticles, and interdigital spaces
- a seperate nail cleaner may be used
- rinse thoroughly
- wash for an additional 3 minutes with 5 mL of product and rinse under running water
- dry thoroughly
Healthcare personnel handwash:
- wet hands with water
- dispense about 5 mL of product into cupped hands and wash in a vigorous manner for 15 seconds
- rinse and dry thoroughly
Skin wound and general skin cleansing:
- thoroughly rinse the area to be cleaned with water
- apply the minimum amount of product necessary to cover the skin or wound area and wash gently
- rinse again thoroughly
- Other information
- Inactive ingredients
- Questions or comments?
-
OTHER SAFETY INFORMATION
Laundering/Cleaning Instructions:
Chlorhexidine gluconate skin cleansers will cause stains if used with chlorine releasing products, such as chlorine bleach. Rinse completely and use only non-chlorine detergents.
WARNING: This product can expose you to coconut oil diethanolamine condensate (cocamide diethanolamine), which is known to the State of California to cause cancer. For more information go to www.P65Warnings.ca.gov
Rev 11/2018
- PRINCIPAL DISPLAY PANEL
- Content of Labeling
-
Package label and principal display panel
NDC: 0404-1061-01
431-0099
HENRY SCHEIN ®
Maxiclens
Antiseptic Skin Cleanser
(Chlorhexidine Gluconate 4%)
FOR EXTERNAL USE ONLY
Net Contents
128 fl oz (1 gal) (3.785 L)
Made in U.S.A.
Distributed By
HENRY SCHEIN INC.
135 Duryea Road
Melville, NY 11747 USA
4MAX1GBTLLBL


-
INGREDIENTS AND APPEARANCE
ANTISEPTIC SKIN CLEANSER
chlorhexidine gluconate 4% solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0404-1061 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORHEXIDINE GLUCONATE (UNII: MOR84MUD8E) (CHLORHEXIDINE - UNII:R4KO0DY52L) CHLORHEXIDINE GLUCONATE 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength COCO DIETHANOLAMIDE (UNII: 92005F972D) GLUCONOLACTONE (UNII: WQ29KQ9POT) HYDROXYETHYL CELLULOSE (2000 CPS AT 1%) (UNII: S38J6RZN16) ISOPROPYL ALCOHOL (UNII: ND2M416302) LAURAMINE OXIDE (UNII: 4F6FC4MI8W) PEG-75 LANOLIN (UNII: 09179OX7TB) WATER (UNII: 059QF0KO0R) TRIDECYL ALCOHOL (UNII: 8I9428H868) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0404-1061-32 946 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/02/2003 2 NDC: 0404-1061-01 3785 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/02/2003 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA019125 04/02/2003 Labeler - Henry Schein, Inc. (012430880) Registrant - Xttrium Laboratories, Inc. (007470579) Establishment Name Address ID/FEI Business Operations Xttrium Laboratories 007470579 manufacture(0404-1061)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.


