PLENVU- polyethylene glycol 3350, sodium sulfate, sodium chloride, potassium chloride, ascorbic acid, sodium ascorbate kit
Plenvu by
Drug Labeling and Warnings
Plenvu by is a Prescription medication manufactured, distributed, or labeled by Salix Pharmaceuticals, Inc, Norgine Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use PLENVU safely and effectively.
See full prescribing information for PLENVU.
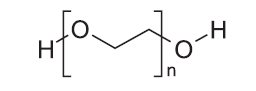
PLENVU® (polyethylene glycol 3350, sodium ascorbate, sodium sulfate,
ascorbic acid, sodium chloride and potassium chloride for oral solution)
Initial U.S. Approval: 2006INDICATIONS AND USAGE
PLENVU is an osmotic laxative indicated for cleansing of the colon in preparation for colonoscopy in adults. (1)
DOSAGE AND ADMINISTRATION
Preparation and Administration:
Two doses of PLENVU are required for a complete preparation for colonoscopy, using a “Two-Day” or “One-Day” dosing regimen. (2.1)
PLENVU must be reconstituted in water prior to ingestion. (2.1)
Additional clear liquids must be consumed after each dose of PLENVU in both dosing regimens. (2.1, 5.1)
Do not take oral medications within 1 hour of starting each dose. (2.1, 7.2)
Dosing Regimen:
Two-Day: Dose 1 the evening before the colonoscopy (approximately 4 pm to 8 pm) and Dose 2 the next morning (approximately12 hours after the start of Dose 1). (2.1, 2.2)
One-Day: Dose 1 the morning of the colonoscopy (approximately 3 am to 7 am) and Dose 2 a minimum of 2 hours after the start of Dose 1. (2.1, 2.3)
For complete information on dosing, preparation and administration see full prescribing information. (2.1, 2.2, 2.3)
DOSAGE FORMS AND STRENGTHS
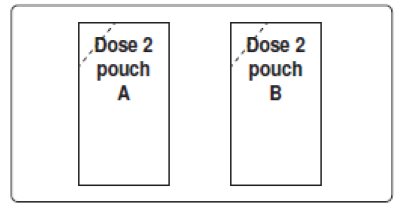
For Oral Solution: First dose: one pouch labeled Dose 1; Second dose: two pouches labeled Dose 2 Pouch A and Dose 2 Pouch B.
Dose 1 contains 100 grams of polyethylene glycol (PEG) 3350, NF; 9 grams of sodium sulfate, USP; 2 grams of sodium chloride, USP/NF; and 1 gram of potassium chloride, USP/NF. (3)
Dose 2 Pouch A contains 40 grams of PEG 3350, NF; 3.2 grams of sodium chloride, USP/NF; and 1.2 grams of potassium chloride, USP/NF. (3)
Dose 2 Pouch B contains 48.11 grams of sodium ascorbate, USP/NF; and 7.54 grams of ascorbic acid, USP/NF. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
Risk of fluid and electrolyte abnormalities: Encourage adequate hydration, assess concurrent medications, and consider laboratory assessments prior to and after use. (5.1, 5.2, 7.1)
Cardiac arrhythmias: Consider pre-dose and post-colonoscopy ECGs in patients at increased risk. (5.2)
Seizures: Use caution in patients with a history of seizures and patients at increased risk of seizure, including medications that lower the seizure threshold. (5.3, 7.1)
Patients with renal impairment or taking concomitant medications that affect renal function: Use caution, ensure adequate hydration and consider testing. (5.4, 7.1, 8.6)
Mucosal ulcerations: Consider potential for mucosal ulcerations when interpreting colonoscopy findings in patients with known or suspected inflammatory bowel disease. (5.5)
Suspected GI obstruction or perforation: Rule out diagnosis before administration. (4, 5.6)
Patients at risk for aspiration: Observe during administration. (5.7)
Glucose-6-phosphate dehydrogenase deficiency (G6PD): Use with caution. (5.8)
Risks in patients with phenylketonuria: Contains phenylalanine. (5.9)
Hypersensitivity reactions, including anaphylaxis: Inform patients to seek immediate medical care if symptoms occur. (5.10)
ADVERSE REACTIONS
Most common adverse reactions (>2%) are nausea, vomiting, dehydration and abdominal pain/discomfort. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Salix Pharmaceuticals, at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Drugs that increase risks due to fluid and electrolyte change. (7.1)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 5/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Preparation and Administration Instructions
2.2 Two-Day Split-Dosing Regimen
2.3 One-Day Morning Dosing Regimen
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Serious Fluid and Electrolyte Abnormalities
5.2 Cardiac Arrhythmias
5.3 Seizures
5.4 Use in Patients with Renal Impairment
5.5 Colonic Mucosal Ulceration, Ischemic Colitis and Ulcerative Colitis
5.6 Use in Patients with Significant Gastrointestinal Disease
5.7 Aspiration
5.8 Glucose-6-Phosphate Dehydrogenase (G6PD) Deficiency
5.9 Risks in Patients with Phenylketonuria
5.10 Hypersensitivity Reactions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Drugs That May Increase Risks Due to Fluid and Electrolyte Abnormalities
7.2 Potential for Reduced Drug Absorption
7.3 Stimulant Laxatives
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Preparation and Administration Instructions
- Correct fluid and electrolyte abnormalities before treatment with PLENVU [see Warnings and Precautions (5.1)].
- Two doses of PLENVU are required for a complete preparation for colonoscopy. The time interval between the two doses depends on the regimen prescribed and the planned timing of the colonoscopy procedure [see Dosage and Administration (2.2, 2.3)].
- The “Two-Day Split-Dosing” method consists of two separate doses: the first dose is taken the evening before the colonoscopy and the second dose is taken the next day, the morning of the day of the colonoscopy [see Dosage and Administration (2.2)].
- The “One-Day Morning Dosing” method consists of two separate doses: both doses are taken in the morning of the day of the colonoscopy, with a minimum of 2 hours between the start of the first dose and the start of the second dose [see Dosage and Administration (2.3)].
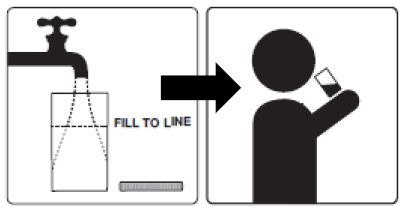
- PLENVU must be reconstituted in water prior to ingestion. It may take 2 to 3 minutes for complete dissolution.
- Additional clear liquids (including water) must be consumed in both dosing regimens [see Dosage and Administration (2.2, 2.3), Warnings and Precautions (5.1)].
- Consume only clear liquids (no solid food) from the start of PLENVU treatment until after the colonoscopy.
- Do not eat or drink alcohol, milk, anything colored red or purple or any other foods containing pulp material.
- Do not take other laxatives while taking PLENVU.
- Do not take oral medications within 1 hour before or after starting each dose of PLENVU [see Drug Interactions (7.2)].
- Ensure completion of Dose 2, including all additional liquids, at least 2 hours before the colonoscopy.
2.2 Two-Day Split-Dosing Regimen
The Two-Day Split-Dosing Regimen commences in the evening of the day before the colonoscopy.
Instruct adult patients that on the day before the clinical procedure, they can consume a light breakfast followed by a light lunch, which must be completed at least 3 hours prior to the start of the first PLENVU dose.
Instruct patients to take two separate doses in conjunction with clear liquids as follows:
Dose 1 – In the evening before the colonoscopy, between approximately 4 pm and 8 pm:
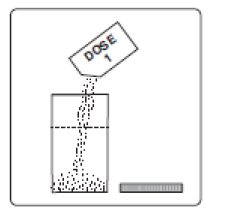
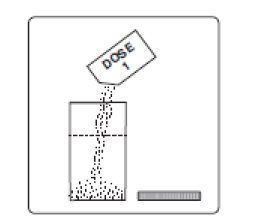
1. Empty the contents of Dose 1 into the mixing container that comes with PLENVU.
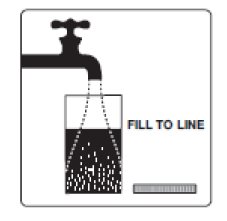
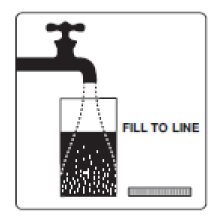
2. Add water to the fill line on the mixing container (at least 16 fluid ounces). Do not add other ingredients to the PLENVU solution.
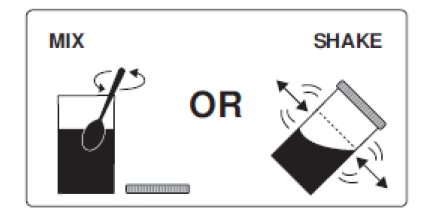
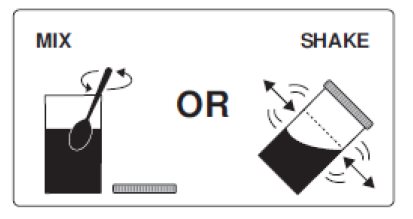
3. Thoroughly mix with a spoon or shake with lid on securely until completely dissolved (which may take 2 to 3 minutes).
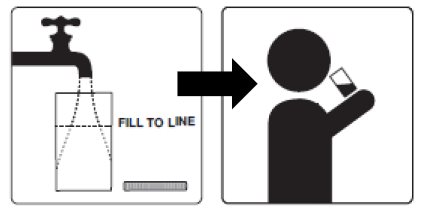
4. Drink over the next 30 minutes. Be sure to drink all of the solution.
5. Refill the mixing container to the fill line (at least 16 fluid ounces) with clear liquids and drink over the next 30 minutes.
6. Consume additional clear liquids during the evening.
7. If severe bloating, abdominal distention, or abdominal pain occurs following the first dose, delay the second dose until the symptoms resolve.
Dose 2 – The next morning, on the day of the colonoscopy, approximately 12 hours after the start of Dose 1 (between approximately 4 am and 8 am):
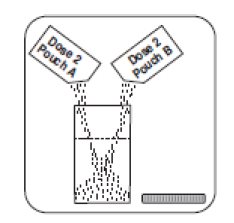
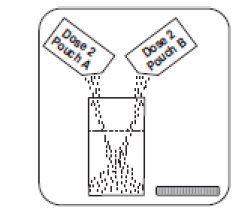
1. Empty the contents of Dose 2 Pouch A and Dose 2 Pouch B into the mixing container that comes with PLENVU.
2. Add water to the fill line on the mixing container (at least 16 fluid ounces). Do not add other ingredients to the PLENVU solution.
3. Thoroughly mix with a spoon or shake with lid on securely until completely dissolved (which may take 2 to 3 minutes). Drink over the next 30 minutes. Be sure to drink all of the solution.
4. Refill the mixing container to the fill line (at least 16 fluid ounces) with clear liquids and drink over the next 30 minutes.
5. Consume additional water or clear liquids up to 2 hours before the colonoscopy or as prescribed by your doctor. Then stop drinking liquids until after the colonoscopy.
Stop drinking PLENVU temporarily or drink each portion at longer intervals if severe bloating, abdominal discomfort or distention occurs, until these symptoms resolve.
2.3 One-Day Morning Dosing Regimen
The One-Day Morning Dosing Regimen commences in the morning of the day of the colonoscopy.
Instruct adult patients that on the day before the clinical procedure, they can consume a light breakfast followed by a light lunch, and clear broth soup and/or plain yogurt for dinner, which should be completed by approximately 8 pm.
Instruct patients to take two separate doses in conjunction with clear liquids as follows:
Dose 1 – On the day of the colonoscopy, between approximately 3 am and 7 am:
1. Empty the contents of Dose 1 into the mixing container that comes with PLENVU.
2. Add water to the fill line on the mixing container (at least 16 fluid ounces). Do not add other ingredients to the PLENVU solution.
3. Thoroughly mix with a spoon or shake with lid on securely until completely dissolved (which may take 2 to 3 minutes).
4. Drink over the next 30 minutes. Be sure to drink all of the solution.
5. Refill the mixing container to the fill line (at least 16 fluid ounces) with clear liquids and drink over the next 30 minutes.
6. If severe bloating, abdominal distention, or abdominal pain occurs following the first dose, delay the second dose until the symptoms resolve.Dose 2 – On the day of the colonoscopy, a minimum of 2 hours after the start of Dose 1:
1. Empty the contents of Dose 2 Pouch A and Dose 2 Pouch B into the mixing container that comes with PLENVU.
2. Add water to the fill line on the mixing container (at least 16 fluid ounces). Do not add other ingredients to the PLENVU solution.
3. Thoroughly mix with a spoon or shake with lid on securely until completely dissolved (which may take 2 to 3 minutes). Drink over the next 30 minutes. Be sure to drink all of the solution.
4. Refill the mixing container to the fill line (at least 16 fluid ounces) with clear liquids and drink over the next 30 minutes.
5. Consume additional water or clear liquids up to 2 hours before the colonoscopy or as prescribed by your doctor. Then stop drinking liquids until after the colonoscopy.
Stop drinking PLENVU temporarily or drink each portion at longer intervals if severe bloating, abdominal discomfort or distention occurs, until these symptoms resolve.Storage:
After reconstitution, keep PLENVU solution at room temperature, between 68°F to 77°F (20°C to 25°C) [see USP Controlled Room Temperature]. The solution may also be stored in a refrigerator. Use within 6 hours after it is mixed in water. -
3 DOSAGE FORMS AND STRENGTHS
PLENVU (polyethylene glycol 3350, sodium ascorbate, sodium sulfate, ascorbic acid, sodium chloride and potassium chloride for oral solution) is supplied as a white to yellow powder for reconstitution.
First dose: one pouch labeled Dose 1; Second dose: two pouches labeled Dose 2 Pouch A and Dose 2 Pouch B.
Dose 1 contains 100 grams of polyethylene glycol (PEG) 3350, NF; 9 grams of sodium sulfate, USP; 2 grams of sodium chloride, USP/NF; and 1 gram of potassium chloride, USP/NF.Dose 2 Pouch A contains 40 grams of PEG 3350, NF; 3.2 grams of sodium chloride, USP/NF; and 1.2 grams of potassium chloride, USP/NF.
Dose 2 Pouch B contains 48.11 grams of sodium ascorbate, USP/NF; and 7.54 grams of ascorbic acid, USP/NF.
-
4 CONTRAINDICATIONS
PLENVU is contraindicated in the following conditions:
Gastrointestinal (GI) obstruction [see Warnings and Precautions (5.6)]
Bowel perforation [see Warnings and Precautions (5.6)]
Gastric retention
Ileus
Toxic megacolon
Hypersensitivity to any ingredient in PLENVU [see Warnings and Precautions (5.10)] -
5 WARNINGS AND PRECAUTIONS
5.1 Serious Fluid and Electrolyte Abnormalities
Advise patients to hydrate adequately before, during, and after the use of PLENVU. If a patient develops significant vomiting or signs of dehydration after taking PLENVU, consider performing post-colonoscopy laboratory tests (electrolytes, creatinine, and BUN).
Bowel Preparations can cause fluid and electrolyte disturbances, which can lead to serious adverse reactions including cardiac arrhythmias, seizures, and renal impairment. Correct fluid and electrolyte abnormalities before treatment with PLENVU. PLENVU should be used with caution in patients using concomitant medications that increase the risk of electrolyte abnormalities [such as diuretics, angiotensin converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs)] [see Drug Interactions (7.1)]. Consider performing pre-dose and post-colonoscopy laboratory tests (sodium, potassium, calcium, creatinine, and BUN) in patients receiving these concomitant medications.
5.2 Cardiac Arrhythmias
There have been rare reports of serious arrhythmias (including atrial fibrillation) associated with the use of ionic osmotic laxative products for bowel preparation. These occur predominantly in patients with underlying cardiac risk factors and electrolyte disturbances. Use caution when prescribing PLENVU for patients at increased risk of arrhythmias (e.g., patients with a history of prolonged QT, uncontrolled arrhythmias, recent myocardial infarction, unstable angina, congestive heart failure, cardiomyopathy or electrolyte imbalance). Consider pre-dose and post-colonoscopy ECGs in patients at increased risk of serious cardiac arrhythmias.
5.3 Seizures
There have been rare reports of generalized tonic-clonic seizures and/or loss of consciousness associated with use of bowel preparation products in patients with no prior history of seizures. The seizure cases were associated with electrolyte abnormalities (e.g., hyponatremia, hypokalemia, hypocalcemia, and hypomagnesemia) and low serum osmolality. The neurologic abnormalities resolved with correction of fluid and electrolyte abnormalities.
Use caution when prescribing PLENVU for patients with a history of seizures and in patients at increased risk of seizure, such as patients taking medications that lower the seizure threshold (e.g., tricyclic antidepressants), patients withdrawing from alcohol or benzodiazepines, or patients with known or suspected hyponatremia. [see Drug Interactions (7.1)].
5.4 Use in Patients with Renal Impairment
Use PLENVU with caution in patients with renal impairment or patients taking concomitant medications that affect renal function (such as diuretics, ACE inhibitors, angiotensin receptor blockers, or nonsteroidal anti-inflammatory drugs) [see Drug Interactions (7.1)]. These patients may be at risk for renal injury. Advise these patients of the importance of adequate hydration before, during and after the use of PLENVU, and consider performing pre-dose and post-colonoscopy laboratory tests (electrolytes, creatinine, and BUN) in these patients [see Use in Specific Populations (8.6)].
5.5 Colonic Mucosal Ulceration, Ischemic Colitis and Ulcerative Colitis
Osmotic laxatives may produce colonic mucosal aphthous ulcerations, and there have been reports of more serious cases of ischemic colitis requiring hospitalization. Concurrent use of stimulant laxatives and PLENVU may increase the risk and is not recommended. Consider the potential for mucosal ulcerations resulting from the bowel preparation when interpreting colonoscopy findings in patients with known or suspected inflammatory bowel disease.
5.6 Use in Patients with Significant Gastrointestinal Disease
If gastrointestinal obstruction or perforation is suspected, perform appropriate diagnostic studies to rule out these conditions before administering PLENVU [see Contraindications (4)]. Use with caution in patients with severe ulcerative colitis.
5.7 Aspiration
Patients with impaired gag reflex or other swallowing abnormalities are at risk for regurgitation or aspiration of PLENVU. Observe these patients during the administration of PLENVU. Use with caution in these patients.
5.8 Glucose-6-Phosphate Dehydrogenase (G6PD) Deficiency
Since PLENVU contains sodium ascorbate and ascorbic acid, PLENVU should be used with caution in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency, especially G6PD deficiency patients with an active infection, with a history of hemolysis, or taking concomitant medications known to precipitate hemolytic reactions.
5.9 Risks in Patients with Phenylketonuria
Phenylalanine can be harmful to patients with phenylketonuria (PKU). PLENVU contains phenylalanine, a component of aspartame. Each PLENVU treatment contains 491 mg of phenylalanine. Before prescribing PLENVU to a patient with PKU, consider the combined daily amount of phenylalanine from all sources, including PLENVU.
5.10 Hypersensitivity Reactions
PLENVU contains PEG and may cause serious hypersensitivity reactions including anaphylaxis, angioedema, rash, urticaria, and pruritus [see Adverse Reactions (6.1, 6.2)]. Inform patients of the signs and symptoms of anaphylaxis, and instruct them to seek immediate medical care should signs and symptoms occur.
-
6 ADVERSE REACTIONS
The following serious or otherwise important adverse reactions for bowel preparations are described elsewhere in the labeling:
Serious Fluid and Electrolyte Abnormalities [see Warnings and Precautions (5.1)]
Cardiac Arrhythmias [see Warnings and Precautions (5.2)]
Seizures [see Warnings and Precautions (5.3)]
Patients with Renal Impairment [see Warnings and Precautions (5.4)]
Colonic Mucosal Ulceration, Ischemic Colitis and Ulcerative Colitis [see Warnings and Precautions (5.5)]
Patients with Significant Gastrointestinal Disease [see Warnings and Precautions (5.6)]
Aspiration [see Warnings and Precautions (5.7)]
Glucose-6-Phosphate Dehydrogenase (G6PD) Deficiency [see Warnings and Precautions (5.8)]
Risks in Patients with Phenylketonuria [see Warnings and Precautions (5.9)]
Hypersensitivity Reactions [see Warnings and Precautions (5.10)]6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of PLENVU as a Two-Day Split-Dosing and One-Day Morning Dosing Regimen was evaluated in two randomized, parallel group, multicenter, investigator-blinded clinical trials (Two-Day Split-Dosing in the NOCT and MORA trials and One-Day Morning Dosing in the MORA trial) in 1351 adult patients undergoing colonoscopy. The mean age of the study population was 56 years (range 18 to 86 years), 92% of patients were Caucasian and 51% were female. In the NOCT trial, 61% of patients had mild renal impairment. In the MORA trial, 67% had mild renal impairment and 5% had moderate renal impairment. Patients with severe renal impairment were not enrolled in the clinical trials of PLENVU [see Clinical Studies (14)].
The most common adverse reactions (>2%) in the PLENVU treatment groups in both trials were: nausea, vomiting, dehydration and abdominal pain/discomfort.
Table 1 and Table 2 display adverse reactions reported in at least 1% of patients in one or more treatment group(s) in the NOCT and MORA trials, respectively. Since diarrhea was considered as a part of the efficacy assessment, it was not defined as an adverse reaction in these trials.
Table 1: Common Adverse Reactions* in Patients Undergoing Colonoscopy in the NOCT Trial by Treatment Group - * Trisulfate: Two 6 ounce bottles of oral solution each containing sodium sulfate 17.5 grams, potassium sulfate 3.13 grams, magnesium sulfate 1.6 grams
- † Includes signs and symptoms of dehydration, including dizziness, dry mouth, orthostatic hypotension, pre-syncope, syncope, and thirst
- ‡ Includes abdominal discomfort, abdominal pain, lower abdominal pain, upper abdominal pain, and abdominal tenderness
- § Decreased or abnormal GFR
- ¶ Includes increased anion gap, decreased blood bicarbonate, hypomagnesemia, hyperosmolarity, hypokalemia, hyperkalemia, hypercalcemia, hypernatremia, hyperosmolar state, hyperuricemia, hypocalcemia, and hypophosphatemia
Preferred Term
PLENVU
Two-Day Split Dosing
Regimen
(N = 275)
%Trisulfate
Two-Day Split Dosing
Regimen*
(N = 271)
%Nausea
7
2
Vomiting
6
3
Dehydration†
4
2
Abdominal Pain/Discomfort‡
2
2
Decline in Glomerular Filtration Rate (GFR)§
2
2
Electrolyte Abnormalities¶
2
1
Fatigue
2
1
Headache
2
1
Abdominal Distension
1
1
Gastritis
1
1
Hiatus Hernia
1
0
Nasopharyngitis
1
1
* Reported in at least 1% of patients in either treatment group
N = Total number of patients in the treatment group, n = the number of patients with the adverse reaction
Table 2: Common Adverse Reactions* in Patients Undergoing Colonoscopy in the MORA Trial by Treatment Group - * 2 Liter PEG Plus Electrolytes: Two doses each containing PEG 3350 100 grams, sodium sulfate 7.5 grams, sodium chloride 2.691 grams, potassium chloride 1.015 grams, sodium ascorbate 5.9 grams, and ascorbic acid 4.7 grams
- † Includes signs and symptoms of dehydration, including dizziness, dry mouth, orthostatic hypotension, pre-syncope, syncope, and thirst
- ‡ Includes abdominal discomfort, abdominal pain, lower abdominal pain, upper abdominal pain, and abdominal tenderness
- § Includes increased anion gap, decreased blood bicarbonate, hypomagnesemia, increased blood osmolarity, hypokalemia, hyperkalemia, hypercalcemia, hypernatremia, hyperosolar state, hyperuricemia, hypocalcemia, and hypophosphatemia
Preferred Term
PLENVU
One-Day Morning
Dosing Regimen
(N = 271)
%
PLENVU
Two-Day Split
Dosing Regimen
(N = 265)
%
2 Liter PEG +
Electrolytes
Two-Day Split-
Dosing Regimen*
(N = 269)
%
Vomiting
7
4
1
Nausea
6
6
3
Dehydration†
4
3
2
Abdominal Pain/Discomfort‡
3
2
3
Hypertension
2
1
0
Headache
1
2
2
Electrolyte Abnormalities§
1
1
0
* Reported in at least 1% of patients in either treatment group
N = Total number of patients in the treatment group; n = the number of patients with the adverse reaction
Electrolyte Changes
Increases in serum sodium, chloride, calcium, magnesium, phosphate, and urate were noted in more patients treated with PLENVU compared with control in one or both trials. The majority of these changes were transient and not clinically significant. Associated decreases in bicarbonate and increases in serum osmolality were also noted.
Renal Function
Decreases in creatinine clearance and increases in blood urea nitrogen (BUN) were also noted in more patients treated with PLENVU compared to control in both trials. Changes of a magnitude indicative of possible acute renal injury, or worsening of baseline chronic renal impairment, were noted infrequently and occurred at a similar incidence in both PLENVU and comparator arms.
Adverse reactions in patients with mild renal impairment were similar to those in patients with normal renal function.
Less Common Adverse Reactions
Less common adverse reactions (less than 1%) in the NOCT and MORA trials include: anorectal discomfort, hypersensitivity reaction (including rash), migraine, somnolence, asthenia, chills, pains, aches, palpitation, sinus tachycardia, hot flush, and transient increase in liver enzymes.
An additional 235 patients were exposed to the One-Day Morning Dosing Regimen of PLENVU in a third clinical trial, utilizing a comparator not approved in the United States. The adverse reaction profile for patients receiving PLENVU in that trial was similar to what is described above.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of another oral formulation of polyethylene glycol 3350, sodium ascorbate, sodium sulfate, ascorbic acid, sodium chloride and potassium chloride or other polyethylene glycol (PEG)-based bowel preparations. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity: urticaria/rash, pruritus, dermatitis, rhinorrhea dyspnea, chest and throat tightness, fever, angioedema, anaphylaxis and anaphylactic shock [see Contraindications (4)]
Cardiovascular: arrhythmia, atrial fibrillation, peripheral edema, asystole, and acute pulmonary edema after aspiration
Gastrointestinal: upper gastrointestinal bleeding from a Mallory-Weiss tear, esophageal perforation [usually with gastroesophageal reflux disease (GERD)]
Nervous system: tremor, seizure
-
7 DRUG INTERACTIONS
7.1 Drugs That May Increase Risks Due to Fluid and Electrolyte Abnormalities
Use caution when prescribing PLENVU for patients with conditions and/or who are using medications that increase the risk of fluid and electrolyte disturbances or may increase the risk of renal impairment, seizures, arrhythmias, or QT prolongation in the setting of fluid and electrolyte abnormalities [see Warnings and Precautions (5.1, 5.2, 5.3, 5.4)].
Consider additional patient evaluations as appropriate.
7.2 Potential for Reduced Drug Absorption
PLENVU can reduce the absorption of other coadministered drugs. Administer oral medications at least 1 hour before the start of administration of each dose of PLENVU [see Dosage and Administration (2.1)].
7.3 Stimulant Laxatives
Concurrent use of stimulant laxatives and PLENVU may increase the risk of mucosal ulceration or ischemic colitis. Avoid use of stimulant laxatives (e.g., bisacodyl, sodium picosulfate) while taking PLENVU [see Warnings and Precautions (5.5)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data with PLENVU in pregnant women to inform a drug-associated risk for adverse developmental outcomes. Animal reproduction studies have not been conducted with PLENVU.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data available to assess the presence of PLENVU in human milk, the effects on the breastfed child or the effects on milk production. The lack of clinical data during lactation precludes a clear determination of the risk of PLENVU to a child during lactation; therefore, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for PLENVU and any potential adverse effects on the breastfed child from PLENVU or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of PLENVU in pediatric patients has not been established.
8.5 Geriatric Use
Of the approximately 1000 patients in clinical trials receiving PLENVU, 217 (21%) patients were over 65 years of age. No overall differences in safety or effectiveness were observed between geriatric patients and younger patients, and other reported clinical experience has not identified differences in responses between geriatric patients and younger patients. However, elderly patients are more likely to have decreased hepatic, renal or cardiac function and may be more susceptible to adverse reactions resulting from fluid and electrolyte abnormalities [see Warnings and Precautions (5.1)].
8.6 Renal Impairment
Use PLENVU with caution in patients with renal impairment or patients taking concomitant medications that may affect renal function [see Drug Interactions (7.1)]. These patients may be at risk for renal injury. Advise these patients of the importance of adequate hydration before, during and after the use of PLENVU, and consider performing baseline and post-colonoscopy laboratory tests (electrolytes, creatinine, and BUN) in these patients [see Warnings and Precautions (5.4)].
- 10 OVERDOSAGE
-
11 DESCRIPTION
The active ingredients contained in PLENVU are provided in Table 3.
PLENVU (polyethylene glycol 3350, sodium ascorbate, sodium sulfate, ascorbic acid, sodium chloride and potassium chloride for oral solution) is an osmotic laxative consisting of three pouches (one for Dose 1, one for Dose 2 Pouch A and one for Dose 2 Pouch B) containing white to yellow powder for reconstitution.
Dose 1 contains 100 grams of PEG 3350, NF; 9 grams of sodium sulfate, USP; 2 grams of sodium chloride, USP/NF; and 1 gram of potassium chloride, USP/NF; and the following excipients: sucralose, NF; encapsulated citric acid; and mango flavoring. When Dose 1 is dissolved in water to a volume of 16 fluid ounces, PLENVU Dose 1 (PEG 3350, sodium sulfate, sodium chloride and potassium chloride) is an oral solution having a mango flavor.
Each Dose 2 Pouch A contains 40 grams of PEG 3350, NF; 3.2 grams of sodium chloride, USP/NF; and 1.2 grams of potassium chloride, USP/NF and the following excipients: aspartame, NF and fruit punch flavoring.
Each Dose 2 Pouch B contains 48.11 grams of sodium ascorbate, USP/NF; and 7.54 grams of ascorbic acid, USP/NF.
When Dose 2 Pouch A and Dose 2 Pouch B are dissolved together in water to a volume of 16 fluid ounces, PLENVU Dose 2 (sodium ascorbate, PEG 3350, ascorbic acid, sodium chloride and potassium chloride) is an oral solution having a fruit punch flavor.
The entire, reconstituted, 32 fluid ounces of PLENVU bowel preparation contains 140 grams of PEG 3350, 48.11 grams of sodium ascorbate, 9 grams of sodium sulfate, 7.54 grams of ascorbic acid, 5.2 grams of sodium chloride and 2.2 grams of potassium chloride and the following excipients: aspartame, sucralose, encapsulated citric acid, mango and fruit punch flavorings.
A mixing container for reconstitution is enclosed.
Phenylketonurics: Contains Phenylalanine 491 mg per treatment.
Contains no ingredient made from a gluten-containing grain (wheat, barley, or rye).
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The primary mode of action is osmotic action of the components of PLENVU (PEG 3350 plus sodium sulfate components in Dose 1, and sodium ascorbate and ascorbic acid plus PEG 3350 components in Dose 2) which induce the laxative effect. The physiological consequence is increased water retention in the lumen of the colon, resulting in loose stools.
12.2 Pharmacodynamics
The osmotic effect of the unabsorbed PEG, ascorbate and sulfate ions, when ingested, produces a copious watery diarrhea.
The first bowel movement may happen about 1 to 2 hours after the start of PLENVU intake.
12.3 Pharmacokinetics
The plasma pharmacokinetic parameters for PEG 3350, ascorbate and sulfate are shown in Table 4.
Table 4: Plasma Pharmacokinetic Data Following Two-Day Split-Dosing Regimen of 140 grams PEG 3350, 33.9 grams Sodium Ascorbate, 9 grams Sodium Sulfate, 20.1 grams Ascorbic Acid, 4.8 grams Sodium Chloride and 2.3 grams Potassium Chloride in Healthy Subjects* (N=21)† - * Four-day study with controlled diet including fasting from 2 pm on Day 1 to 2 pm on Day 2.
- † Product studied contains the same amount of PEG 3350 and sodium sulfate, although the amount of sodium ascorbate and ascorbic acid are slightly different, compared to PLENVU.
- ‡ Baseline-corrected
PK Parameter
PEG 3350
Mean (SD)Ascorbate‡
Mean (SD)Sulfate‡
Mean (SD)
Cmax [mcg/mL]
2.7 (1.17)
70.8 (22.37)
17.6 (4.80)
tmax [h]
3.0 (0.61)
16.8 (0.75)
8.1 (5.51)
AUC(0-tlast)
[(mcg/mL)*h]
17.3 (7.19)
433.1 (157.29)
206.2 (74.32)
Vd [l]
48,481 (29,811)
1,026 (675)
231 (205)
t1/2 [h]
4.1 (2.34)
7.2 (6.16)
-
- 5.2 (15.19)
SD = standard deviation; Cmax = maximum concentration; tmax = time to maximum concentration from start of dosing; AUC (0-tlast) = area under the curve from t0 to tlast; Vd = volume of distribution; t1/2=half-life.
A pharmacokinetic study measured up to 85% to 99% of a 140 grams oral PEG 3350 dose in excreted feces.
A pharmacokinetic study measured up to 69% of a 50 grams oral ascorbate dose in excreted feces and up to 5% of the 50 grams oral ascorbate dose is recovered in the urine (with up to 0.07% as the ascorbate metabolite, oxalic acid).
Sulfate is endogenous and also present in the diet. A pharmacokinetic study measured up to 69% to 73% of a 9 grams oral sodium sulfate dose in excreted feces, with approximately 43% recovered in the urine.
-
14 CLINICAL STUDIES
Study Design
The colon cleansing efficacy, safety and tolerability of PLENVU was evaluated in two randomized, parallel-group, multicenter, investigator-blinded trials in adult patients scheduled to undergo a screening, surveillance, or diagnostic colonoscopy. The overall patient population consisted of 49% male and 51% female patients, mean age of 56 years (range 18 to 86 years), 92% Caucasian, 5% Black and 2% Asian. In general, the demographic characteristics were balanced across the trials.
In Study NER1006-01/2014 (referred to as NOCT; NCT02254486) and Study NER1006-02/2014 (referred to as MORA; NCT02273167), the bowel cleansing efficacy of PLENVU was compared to two different comparators (Table 5) using two different PLENVU dosing regimen(s):
PLENVU Two-Day Split-Dosing Regimen allows for an overnight gap between doses (Dose 1 taken in the evening before the colonoscopy, between approximately 4 pm and 8 pm, and Dose 2 the next morning, on the day of the colonoscopy, approximately 12 hours after the start of Dose 1).
PLENVU One-Day Morning Dosing Regimen gives both doses the morning of the day of colonoscopy (Dose 1 between approximately 3 am and 7 am, and Dose 2 a minimum of 2 hours after the start of Dose 1).
Table 5: Treatment Regimens by Trial Trial
PLENVU Regimen(s)
Comparator Regimens
NOCT
Two-Day Split-Dosing
Trisulfate bowel cleansing solution
administered as a Two-Day Split-Dosing Regimen:
[Trisulfate (Two 6 ounce bottles each containing sodium sulfate 17.5 grams, potassium sulfate 3.13 grams, and
magnesium sulfate 1.6 grams)]
MORA
Two-Day Split-Dosing
and
One-Day Morning Dosing
2 liter PEG + electrolytes (2 L PEG+E) preparation
administered as a Two-Day Split-Dosing Regimen:Two doses, each containing PEG 3350 100 grams, sodium sulfate 7.5 grams, sodium chloride 2.691 grams,
potassium chloride 1.015 grams, sodium ascorbate
5.9 grams, and ascorbic acid 4.7 grams
Primary Endpoint
The primary efficacy endpoint in both trials was the proportion of patients achieving “overall bowel cleansing success,” which was defined by a result of Grade A or B (Grades A or B [see Table 6] corresponding to full visualization of the bowel mucosa on the Harefield Cleansing Scale [HCS]), as assessed on withdrawal of colonoscope. The HCS segmental scores were initially evaluated by the colonoscopist at the site, who was blinded to treatment, and evaluated for endpoint analysis by central readers (gastroenterologists) using video recordings of the colonoscopy.
Table 6: Harefield Cleansing Scale - * Colon ascendens, Colon transversum, Colon descendens, Colon sigmoideum, Rectum
Overall Grade
Description
A
All five segments* scored 3 or 4
(Mucosa is fully visualized without cleaning.)
B
One or more segments scored 2, remaining segments
scored 3 or 4
(Mucosa is fully visualized.)
C
One or more segments scored 1, remaining segments
scored 2, 3 or 4
D
One or more segments scored 0
Segmental Score
Description
4
Empty and clean
3
Clear liquid
2
Brown liquid/fully removable semisolid stools
1
Semisolid, only partially removable stools
0
Irremovable, heavy, hard stools
Statistical Analysis
The modified Intent-to-Treat (mITT) population was used as the primary population for the efficacy analyses and was defined as all randomized patients with the exception of any patient who (i) was randomized but subsequently failed to meet entry criteria and (ii) in whom it was confirmed (from their patient diary) that the same patient did not receive any study drug.Non-inferiority was assessed using a one-sided 97.5% confidence interval (CI) for the difference in proportions of patients for the overall bowel cleansing success endpoint. Non-inferiority was demonstrated if the difference between PLENVU and the comparator was above the predefined non-inferiority margin set at -10%.
Efficacy Results
The results for the overall bowel cleansing success endpoint in the mITT population in NOCT are shown in Table 7. The Two-Day Split-Dosing regimen of PLENVU was shown to be non-inferior (NI) to the trisulfate solution comparator.Table 7: Overall Bowel Cleansing Success Rate of PLENVU versus Trisulfate in NOCT Primary Endpoint
(N=556)
PLENVU
Two-Day
Split-Dosing
(N=276)
n (% = n/N*100)
Trisulfate
Two-Day
Split-Dosing
(N=280)
n (% = n/N*100)
PLENVU® - Trisulfate
Difference (%)
(97.5% One-Sided Lower
Confidence Interval)
Overall Colon
Cleansing Success
Rate
235
(85.1%)
238
(85.0%)
0.1%
(-8.2%)
The results for the overall bowel cleansing success endpoint in the mITT population in MORA are shown in
Table 8. Both the PLENVU Two-Day Split-Dosing regimen and the PLENVU One-Day Morning Dosing
regimen were shown to be non-inferior (NI) to the 2 L PEG+E treatment comparator.
Table 8: Overall Bowel Cleansing Success Rate of PLENVU versus 2 L PEG+E in MORA Primary
Endpoint(N=822)
PLENVU
Two-Day
Split-Dosing
(N=275)
n (% = n/N*100)
PLENVU
One-Day Morning
Dosing
(N=275)
n (% = n/N*100)
2 L PEG+E
Two-Day Split-
Dosing
(N=272)
n (% = n/N*100)
PLENVU® Regimen - 2 L
PEG+E
Difference (%)
(97.5% One-Sided Lower
Confidence Interval)
Overall Colon
Cleansing Success
Rate
253
(92.0%)
245
(89.1%)
238
(87.5%)
Two-Day
Split-Dosing4.5%
(-4.0%)
One-Day
Morning Dosing1.6%
(-6.9%)
-
16 HOW SUPPLIED/STORAGE AND HANDLING
PLENVU (polyethylene glycol 3350, sodium ascorbate, sodium sulfate, ascorbic acid, sodium chloride and potassium chloride for oral solution) is supplied as a white to yellow powder for reconstitution.
Dose 1 contains 100 grams of PEG 3350, NF; 9 grams of sodium sulfate, USP; 2 grams of sodium chloride, USP/NF; and 1 gram of potassium chloride, USP/NF: NDC: 65649-400-01.
Dose 2 Pouch A contains 40 grams of PEG 3350, NF; 3.2 grams of sodium chloride, USP/NF; and 1.2 grams of potassium chloride, USP/NF: NDC: 65649-400-01.
Dose 2 Pouch B contains 48.11 grams of sodium ascorbate, USP/NF; and 7.54 grams of ascorbic acid, USP/NF: NDC: 65649-400-01.
PLENVU, single-use inner carton: The inner carton contains three pouches labeled Dose 1, Dose 2 Pouch A and Dose 2 Pouch B: NDC: 65649-400-01.
PLENVU, single-use outer carton: Each outer carton contains the inner carton, prescribing information and patient information and a disposable mixing container with lid for reconstitution of PLENVU: NDC: 65649-400-01.
Storage
Store pack at room temperature, between 68°F to 77°F (20°C to 25°C) with excursions permitted to 59°F to 86°F (15°C to 30°C) [see USP Controlled Room Temperature]. The pack may be stored in a refrigerator.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
Instruct patients:
That two doses of PLENVU are required for a complete preparation for colonoscopy either as a Two-Day Split-Dosing or One-Day Morning Dosing Regimen [see Instructions for Use].
Not to take other laxatives while they are taking PLENVU.
That PLENVU contains 491 mg of phenylalanine per treatment [see Warnings and Precautions (5.9)].
That each pouch needs to be reconstituted in water before ingestion and that they should drink additional clear liquids. Examples of clear liquids can be found in the Instructions for Use.
Not to take oral medications within one hour of starting each dose of PLENVU.
To follow the directions in the Instructions for Use, for either the Two-Day Split-Dosing or the One-Day Morning Dosing Regimen, as prescribed.
To consume additional clear liquids before, during, and after the use of PLENVU to prevent dehydration [see Warnings and Precautions (5.1)].
To contact their healthcare provider if they develop significant vomiting or signs of dehydration after taking PLENVU or if they experience altered consciousness or seizures [see Warnings and Precautions (5.1, 5.2, 5.3, 5.4)].
Not eat or drink alcohol, milk, anything colored red or purple or any other foods containing pulp material.
To stop drinking PLENVU temporarily or drink each portion at longer intervals if they develop severe abdominal discomfort or distention until these symptoms diminish. If severe symptoms persist, tell patients to contact their healthcare provider.
Distributed by:
Salix Pharmaceuticals, a division of
Bausch Health US, LLC
Bridgewater, NJ 08807 USAManufactured by:
Norgine Limited
7 Tir-y-berth Industrial Estate
New Road, Tir-y-berth
Hengoed, CF82 8SJ
United Kingdom (GBR)U.S. Patent Numbers: 8,999,313; 9,326,969; 9,592,252; 9,707,297 and 10,016,504
PLENVU is a registered trademark of the Norgine group of companies used under license.
© 2019 Salix Pharmaceuticals, Inc. or its affiliates
- 9643001 70014352
-
MEDICATION GUIDE
PLENVU® (plen-vu)
(polyethylene glycol 3350, sodium ascorbate, sodium sulfate, ascorbic acid, sodium chloride and potassium chloride for oral solution)Read this Medication Guide and Instructions for Use before your colonoscopy and again before you start taking PLENVU.
What is the most important information I should know about PLENVU?
PLENVU and other bowel preparations can cause serious side effects, including:Serious loss of body fluid (dehydration) and changes in blood salts (electrolytes) in your blood. These changes can cause:
- o abnormal heartbeats that can cause death.
- o seizures. This can happen even if you have never had a seizure.
- o kidney problems.
Your chance of having fluid loss and changes in body salts with PLENVU is higher if you:
have heart problems.
have kidney problems.
take water pills (diuretics), high blood pressure medicine or non-steroidal anti-inflammatory drugs (NSAIDs).- vomiting
- urinating less often than normal
- dizziness
- headache
Tell your healthcare provider right away if you have any of these symptoms of serious loss of body fluid (dehydration) while taking PLENVU:
See “What are the possible side effects of PLENVU?” for more information about side effects.
What is PLENVU?
PLENVU is a prescription medicine used by adults to clean the colon before a colonoscopy. PLENVU cleans your colon by causing you to have diarrhea (loose stools). Cleaning your colon helps your healthcare provider see the inside of your colon more clearly during your colonoscopy.
It is not known if PLENVU is safe and effective in children.
Do not take PLENVU if your healthcare provider has told you that you have:
- a blockage in your intestine (bowel obstruction).
- an opening in the wall of your stomach or intestine (bowel perforation).
- problems with food and fluid emptying from your stomach (gastric retention).
- a problem with food moving too slowly through your intestines (ileus).
- a very dilated intestine (toxic megacolon).
- an allergy to any of the ingredients in PLENVU. See the end of this leaflet for a complete list of ingredients in PLENVU.
Before taking PLENVU, tell your healthcare provider about all of your medical conditions, including if you:
- have problems with serious loss of body fluid (dehydration) and changes in blood salts (electrolytes).
- have heart problems.
- have seizures or take medicines for seizures.
- have kidney problems or take medicines for kidney problems.
- have stomach or bowel problems, including ulcerative colitis.
- have problems with swallowing, gastric reflux or if you inhale food or fluid into your lungs when eating or drinking (aspirate).
- have a condition called glucose-6-phosphate dehydrogenase (G6PD) deficiency that destroys red blood cells.
- are withdrawing from drinking alcohol.
- have phenylketonuria (PKU). PLENVU contains phenylalanine.
- are allergic to any of the ingredients in PLENVU.
- are pregnant or plan to become pregnant. It is not known if PLENVU will harm your unborn baby. Talk to your healthcare provider if you are pregnant.
- are breastfeeding or plan to breastfeed. It is not known if PLENVU passes into your breast milk. You and your healthcare provider should decide if you will take PLENVU while breastfeeding.
Tell your doctor about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal supplements.
PLENVU may affect how other medicines work. Do not take medicines by mouth 1 hour before or after the start of PLENVU.
Especially tell your healthcare provider if you take:
- medicines to treat a blood salt (electrolyte) imbalance.
- medicines for blood pressure or heart problems.
- medicines for seizures (antiepileptics).
- medicines for kidney problems.
- water pills (diuretics).
- non-steroidal anti-inflammatory drugs (NSAIDs).
- laxatives. Do not take other laxatives while taking PLENVU.
- medicines for depression or other mental health problems.
Ask your healthcare provider or pharmacist for a list of these medicines if you are not sure if you are taking any of the medicines listed above.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take PLENVU?
See the “Instructions for Use” for dosing instructions. You must read, understand, and follow these instructions to take PLENVU the right way.
- Take PLENVU exactly as your healthcare provider tells you to take it. Your healthcare provider will tell you to take the Two-Day Split-Dosing option or the One-Day Morning Dosing option.
- Drink clear liquids before, during, and after you take PLENVU, up until 2 hours before your colonoscopy, to help prevent fluid loss (dehydration) and changes in blood salt (electrolyte) levels.
- Do not eat solid food while taking PLENVU until after your colonoscopy.
- It is important for you to drink the additional amount of clear liquids listed in the Instructions for Use.
-
You may have stomach-area (abdomen) bloating after your first dose of PLENVU.
- o If you have severe stomach-area (abdomen) discomfort or bloating, stop drinking PLENVU for a short time or wait a longer time between each dose of PLENVU until your stomach-area symptoms improve. If your stomach-area discomfort or bloating continues, tell your healthcare provider.
- Your first bowel movement may happen about 1 to 2 hours after you start taking PLENVU.
- If you take too much PLENVU, call your healthcare provider.
What are the possible side effects of PLENVU?
PLENVU can cause serious side effects including:
- Changes in certain blood tests. Your healthcare provider may do blood tests after you take PLENVU to check your blood for changes. Tell your healthcare provider if you have any symptoms of too much fluid loss, including:
- o vomiting
- o heart problems
- o seizures
- o dizziness
- o Kidney problems
- o dry mouth
- o feel faint, weak or lightheaded especially when you stand up (orthostatic hypotension)
- Ulcers of the bowel or bowel problems (ischemic colitis):Tell your healthcare provider right away if you have severe stomach-area (abdomen) pain or rectal bleeding.
- Serious allergic reactions. Symptoms of a serious allergic reaction may include:
- o Skin rash
- o Raised red patched
- o Kidney problems
- o itching
- o swelling of the face, lips, tongue and throat
The most common side effects of PLENVU include:
- o nausea
- o vomiting
- o dehydration
- o stomach pain or discomfort
These are not all the possible side effects of PLENVU.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store PLENVU?
- Store PLENVU (before opening and after mixed) at room temperature, between 68°F to 77°F (20°C to 25°C). PLENVU may also be stored in a refrigerator.
- Use PLENVU within 6 hours after mixing with water.
Keep PLENVU and all medicines out of the reach of children.
General information about the safe and effective use of PLENVU.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use PLENVU for a condition for which it was not prescribed. Do not give PLENVU to other people, even if they are going to have the same procedure you are. It may harm them.
You can ask your pharmacist or healthcare provider for information that is written for healthcare professionals.What are the ingredients in PLENVU?
Active ingredient:
Dose 1: PEG 3350, sodium sulfate, sodium chloride, potassium chloride
Dose 2 Pouch A: PEG 3350, sodium chloride, potassium chloride
Dose 2 Pouch B: sodium ascorbate, ascorbic acidInactive ingredients:
Dose 1: sucralose, encapsulated citric acid, mango flavoring
Dose 2 Pouch A: aspartame, fruit punch flavoringDistributed by:
Salix Pharmaceuticals, a division of
Bausch Health US, LLC
Bridgewater, NJ 08807 USAManufactured by:
Norgine Limited
7 Tir-y-berth Industrial Estate
New Road, Tir-y-berth
Hengoed, CF82 8SJ
United Kingdom (GBR)U.S. Patent Numbers: 8,999,313; 9,326,969; 9,592,252; 9,707,297 and 10,016,504
PLENVU is a registered trademark of the Norgine group of companies used under license.
© 2019 Salix Pharmaceuticals, Inc. or its affiliates
For more information, go to www.PLENVU.com or call 1-800-321-4576.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
- Revised: May 2019 9643001 70014352
-
Instructions for Use
PLENVU®
(polyethylene glycol 3350, sodium ascorbate, sodium sulfate, ascorbic acid, sodium chloride and potassium chloride for oral solution)There are two different options for taking PLENVU. Your healthcare provider will tell you to take the Two-Day Split-Dosing option or the One-Day Morning Dosing option.
The following are provided with the pack:
An outer carton:
An inner carton:
A mixing container with removable lid:
Prescribing Information and Patient Information
The inner carton contains:
Dose 1
Dose 2 Pouch A and Dose 2
Pouch B (to be taken together)Additional supplies:
- Water (to mix with PLENVU).
- Scissors (optional to cut the pouches on the dotted line).
- Spoon (optional to mix the water and PLENVU together. You can also mix the water and PLENVU by putting the lid on securely and shaking it. See step 1c below).
Important Information on PLENVU:
You must drink all of Dose 1 (one pouch) and Dose 2 (two pouches) of PLENVU for either dosing option. Make sure you finish Dose 2 at least 2 hours before your colonoscopy. Do not add any other ingredients to PLENVU.
PLENVU must be mixed with water.
Drink clear liquids before, during, and after you take PLENVU, up until 2 hours before your colonoscopy, to help prevent fluid loss (dehydration), and changes in blood salt (electrolyte) levels.
It is important for you to drink the additional amount of clear liquids listed here in the Instructions for Use.
Examples of clear liquids are:
- o water
- o clear broth soups
- o herbal tea, black tea or coffee
- o watered down (diluted) (from concentrate) clear fruit juices (without pulp) including apple juice or white grape juice
- o clear soda
- o gelatin (without added fruit or topping)
- o popsicles (without pieces of fruit or fruit pulp)
- o strained limeade or lemonade
Do not eat or drink alcohol, milk, anything colored red or purple or any foods that have pulp.
Do not take other laxatives while taking PLENVU.
Do not take any medicines by mouth (oral) within 1 hour before or after starting each dose of PLENVU.
Do not eat solid food while taking PLENVU until after your colonoscopy.
For the Two-Day Split-Dosing schedule:
- o On the day before the colonoscopy you can eat a light breakfast followed by a light lunch.
- o You must finish eating lunch at least 3 hours before you start taking PLENVU.
- o After you start taking PLENVU you can only drink clear liquids.
For the One-Day Morning Dosing schedule:
- o On the day before the colonoscopy you can eat a light breakfast followed by a light lunch. For dinner you may have clear broth soup and/or plain yogurt.
- o You should finish dinner by about 8 pm.
- o After you start taking PLENVU you can only drink clear liquids.
Do not eat the morning of your colonoscopy.
Two-Day Split-Dosing Schedule
Take Dose 1 in the evening sometime between 4 pm and 8 pm.
Take Dose 2 the next morning, on the day of the colonoscopy. This should be about 12 hours (between about 4 am and 8 am) after you started Dose 1. Make sure you finish Dose 2 at least 2 hours before your colonoscopy.Follow steps 1 through 3 on how to mix with a spoon or shake with lid on securely and take PLENVU:
Step 1a: Empty Dose 1 into the mixing container that comes with your PLENVU.
Step 1b: Add water to the fill line. You will need to add least 16 ounces.
Step 1c: Mix the water and PLENVU together with a spoon or put lid on mixing container securely and shake the water and PLENVU, until it is completely dissolved. This can take 2 to 3 minutes.
Step 2: Drink the entire contents of the mixing container over the next 30 minutes.
If you feel like you have severe stomach pain or discomfort you can stop taking PLENVU for a short time and then continue taking it or you can take smaller sips of PLENVU so that you space out your dose longer than 30 minutes. If you still have severe stomach pain, call your healthcare provider.
Step 3: Rinse the mixing container with water. Refill to the fill line with clear liquids. This will be at least 16 ounces. Drink the entire contents of the mixing container over the next 30 minutes. Continue to drink additional clear liquids during the evening, this is important to help avoid dehydration. For a list of clear liquids, see examples at the top of the Instructions for Use.
After taking PLENVU if you have any bloating or feeling like your stomach is upset, wait to take Dose 2 until your stomach feels better.
For Dose 2: Rinse the mixing container with water. Repeat Steps 1, 2 and 3 but this time for Dose 2 you will empty two dose pouches (Dose 2 Pouch A and Dose 2 Pouch B) into the mixing container at the same time.
After drinking your 16 ounces of water mixed with PLENVU and the 16 ounces of the clear liquids, it is important that you drink additional clear liquids to help avoid dehydration. For a list of clear liquids, see examples at the top of the Instructions for Use. You must stop drinking all liquids at least 2 hours before your colonoscopy.
One-Day Morning Dosing Schedule
Take Dose 1 the morning of your colonoscopy sometime between 3 am and 7 am.
Take Dose 2 about two hours after you start Dose 1. Make sure you finish Dose 2 at least 2 hours before your colonoscopy.
Follow the steps 1 through 3 on how to mix with a spoon or shake with lid on securely and take PLENVU:
Step 1a: Empty Dose 1 into the mixing container that comes with your PLENVU.
Step 1b: Add water to the fill line. You will need to add least 16 ounces.
Step 1c: Mix the water and PLENVU together with a spoon or put lid on mixing container securely and shake the water and PLENVU, until it is completely dissolved. This can take 2 to 3 minutes.
Step 2: Drink the entire contents of the mixing container over the next 30 minutes.
If you feel like you have severe stomach pain or discomfort you can stop taking PLENVU for a short time and then continue taking it or you can take smaller sips of PLENVU so that you space out your dose longer than 30 minutes. If you still have severe stomach pain, call your healthcare provider.
Step 3: Rinse the mixing container with water. Refill to fill line with clear liquids. This will be at least 16 ounces. Drink the entire contents of the mixing container over the next 30 minutes.
After drinking your 16 ounces of water mixed with PLENVU and the 16 ounces of the clear liquids, it is important that you drink additional clear liquids in the morning before dose 2 to help avoid dehydration. For a list of clear liquids, see examples at the top of the Instructions for Use. You must stop drinking all liquids at least 2 hours before your colonoscopy.
After taking PLENVU if you have any bloating or feeling like your stomach is upset, wait to take Dose 2 until your stomach feels better.
For Dose 2: Rinse the mixing container with water. Repeat Steps 1, 2 and 3 but this time for Dose 2 you will empty two dose pouches (Dose 2 Pouch A and Dose 2 Pouch B) into the mixing container at the same time.
After drinking your 16 ounces of water mixed with PLENVU and the 16 ounces of the clear liquids, it is important that you drink additional clear liquids to help avoid dehydration. For a list of clear liquids, see examples at the top of the Instructions for Use. You must stop drinking all liquids at least 2 hours before your colonoscopy.
Distributed by:
Salix Pharmaceuticals, a division of
Bausch Health US, LLC
Bridgewater, NJ 08807 USAManufactured by:
Norgine Limited
7 Tir-y-berth Industrial Estate
New Road, Tir-y-berth
Hengoed, CF82 8SJ
United Kingdom (GBR)
U.S. Patent Numbers: 8,999,313; 9,326,969; 9,592,252; 9,707,297 and 10,016,504PLENVU is a registered trademark of the Norgine group of companies used under license.
© 2019 Salix Pharmaceuticals, Inc. or its affiliates
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: May 2019
- 9643001 70014352
-
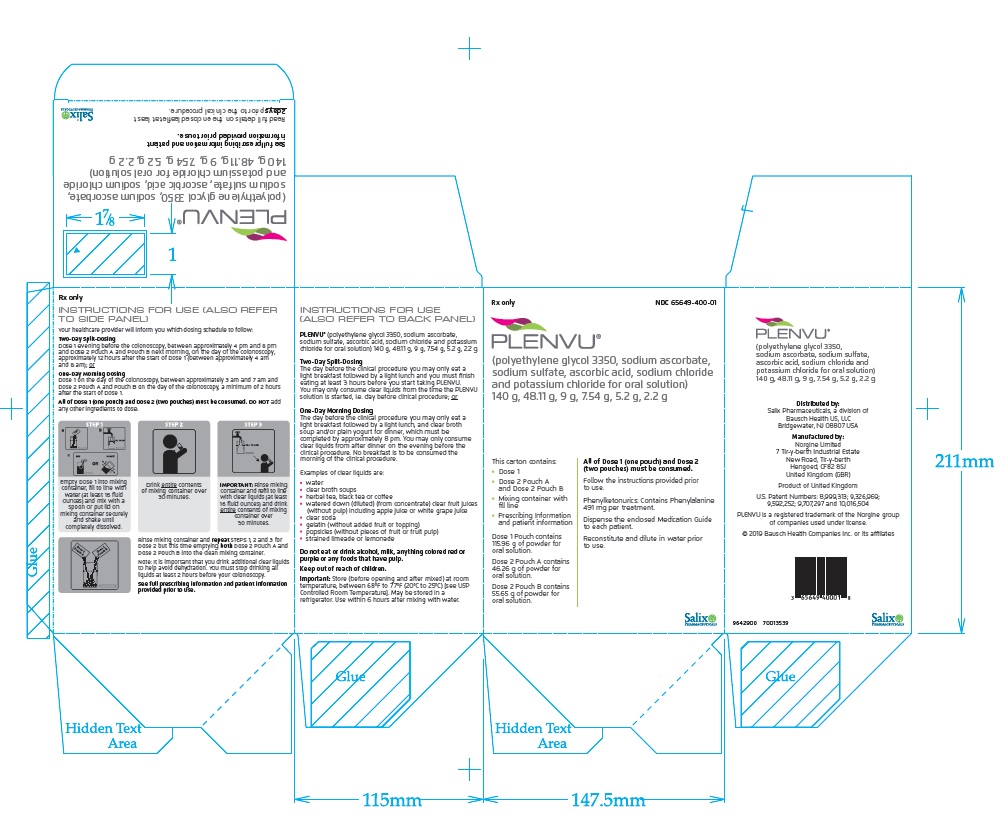
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – Plenvu Outer Carton
Rx only
NDC 65649-400-01
PLENVU®(polyethylene glycol 3350, sodium ascorbate,
sodium sulfate, ascorbic acid, sodium chloride
and potassium chloride for oral solution)
140 g, 48.11 g, 9 g, 7.54 g, 5.2 g, 2.2 gThis carton contains:
- Dose 1
-
Dose 2 Pouch A
and Dose 2 Pouch B -
Mixing container with
fill line -
Prescribing Information
and patient information
Dose 1 Pouch contains
115.96 g of powder for
oral solution.Dose 2 Pouch A contains
46.26 g of powder for
oral solution.Dose 2 Pouch B contains
55.65 g of powder for
oral solution.All of Dose 1 (one pouch) and Dose 2
(two pouches) must be consumed.Follow the instructions provided prior
to use.Phenylketonurics: Contains Phenylalanine
491 mg per treatment.Dispense the enclosed Medication Guide
to each patient.Reconstitute and dilute in water prior
to use.Salix
PHARMACEUTICALS -
INGREDIENTS AND APPEARANCE
PLENVU
polyethylene glycol 3350, sodium sulfate, sodium chloride, potassium chloride, ascorbic acid, sodium ascorbate kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 65649-400 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 65649-400-02 1 in 1 CARTON; Type 0: Not a Combination Product 05/04/2018 2 NDC: 65649-400-01 1 in 1 CARTON; Type 0: Not a Combination Product 05/04/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 POUCH 115.96 Part 2 1 POUCH 46.26 Part 3 1 POUCH 55.65 Part 1 of 3 DOSE 1
polyethylene glycol 3350, sodium sulfate, sodium chloride, potassium chloride powder, for solutionProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) (POLYETHYLENE GLYCOL 3350 - UNII:G2M7P15E5P) POLYETHYLENE GLYCOL 3350 100 g SODIUM SULFATE (UNII: 0YPR65R21J) (SODIUM SULFATE ANHYDROUS - UNII:36KCS0R750) SODIUM SULFATE 9 g SODIUM CHLORIDE (UNII: 451W47IQ8X) (CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 2 g POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152) POTASSIUM CHLORIDE 1 g Inactive Ingredients Ingredient Name Strength SUCRALOSE (UNII: 96K6UQ3ZD4) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) MANGO (UNII: I629I3NR86) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 115.96 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209381 05/04/2018 Part 2 of 3 DOSE 2 POUCH A
polyethylene glycol 3350, sodium chloride, potassium chloride powder, for solutionProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) (POLYETHYLENE GLYCOL 3350 - UNII:G2M7P15E5P) POLYETHYLENE GLYCOL 3350 40 g SODIUM CHLORIDE (UNII: 451W47IQ8X) (CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 3.2 g POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152) POTASSIUM CHLORIDE 1.2 g Inactive Ingredients Ingredient Name Strength ASPARTAME (UNII: Z0H242BBR1) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 46.26 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209381 05/04/2018 Part 3 of 3 DOSE 2 POUCH B
sodium ascorbate, ascorbic acid powder, for solutionProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM ASCORBATE (UNII: S033EH8359) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 48.11 g ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 7.54 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 55.65 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209381 05/04/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209381 05/04/2018 Labeler - Salix Pharmaceuticals, Inc (793108036) Establishment Name Address ID/FEI Business Operations Norgine Limited 239828197 MANUFACTURE(65649-400)
Trademark Results [Plenvu]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 PLENVU 79187028 5116648 Live/Registered |
Velinor AG 2016-03-02 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.