ZOLPIMIST- zolpidem tartrate spray, metered
ZOLPIMIST by
Drug Labeling and Warnings
ZOLPIMIST by is a Prescription medication manufactured, distributed, or labeled by Aytu BioPharma, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ZOLPIMIST safely and effectively. See full prescribing information for ZOLPIMIST.
ZOLPIMIST (zolpidem tartrate) Oral Spray CIV
Initial U.S. Approval: 1992WARNING: COMPLEX SLEEP BEHAVIORS
See full prescribing information for complete boxed warning
Complex sleep behaviors including sleep-walking, sleep-driving, and engaging in other activities while not fully awake may occur following use of ZOLPIMIST. Some of these events may result in serious injuries, including death. Discontinue ZOLPIMIST immediately if a patient experiences a complex sleep behavior. (4, 5.1)
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
ZOLPIMIST is indicated for the short-term treatment of insomnia characterized by difficulties with sleep initiation. Zolpidem tartrate has been shown to decrease sleep latency for up to 35 days in controlled clinical studies. (1)
DOSAGE AND ADMINISTRATION
- Adult dose: 10 mg once daily immediately before bedtime. (2.1)
- Elderly/debilitated patients/hepatic impaired: 5 mg once daily immediately before bedtime. (2.2)
- Downward dosage adjustment may be necessary when used with CNS depressants. (2.3)
- Should not be taken with or immediately after a meal. (2.4)
DOSAGE FORMS AND STRENGTHS
Each metered actuation (one spray) of ZOLPIMIST delivers 5 mg of zolpidem tartrate in 100 μL. After an initial priming of 5 actuations, there are 60 metered actuations in each child-resistant container. The total number of available doses is dependent on the number of actuations per dose (1 or 2 actuations) and the frequency of priming. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- CNS-depressant Effects: Impairs alertness and motor coordination, including the risk of morning impairment. If used in combination with other CNS depressants, dose reduction may be needed due to additive effects. Do not use with alcohol. Instruct patients on correct use. (2.3, 5.2)
- Need to Evaluate for Co-Morbid Diagnosis: Reevaluate if insomnia persists after 7 to 10 days of use. (5.3)
- Severe Anaphylactic and Anaphylactoid Reactions: Angioedema and anaphylaxis have been reported. Do not rechallenge if such reactions occur. (5.4)
- Abnormal Thinking and Behavioral changes: Changes including decreased inhibition, bizarre behavior, agitation, and depersonalization have been reported. Immediately evaluate any new onset of behavioral changes. (5.5)
- Depression: Worsening of depression or suicidal thinking may occur. Prescribe the least amount feasible to avoid intentional overdose. (5.7)
- Withdrawal Effects: Symptoms may occur with rapid dose reduction or discontinuation. (5.5, 9.3)
- Elderly/debilitated patients: Use lower dose due to impaired motor, cognitive performance, and increased sensitivity. (2.2, 5.7)
- Patients with hepatic impairment, mild to moderate COPD, impaired drug metabolism or hemodynamic responses, mild to moderate sleep apnea: Use with caution and monitor closely. (5.7)
ADVERSE REACTIONS
Most commonly observed adverse reactions were:
Short-term (<10 nights): Drowsiness, dizziness, and diarrhea
Long-term (28-35 nights): Dizziness and drugged feelings (6.1)To report SUSPECTED ADVERSE REACTIONS, contact Aytu BioScience, Inc. at 1-855-298-8246 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- CNS depressants: Enhanced CNS-depressant effects with combination use. Use with alcohol causes additive psychomotor impairment. (7.1)
- Imipramine: Decreased alertness observed with combination use. (7.1)
- Chlorpromazine: Impaired alertness and psychomotor performance observed with combination use. (7.1)
- Rifampin: Combination use decreases exposure to and effects of zolpidem. (7.2)
- Ketoconazole: Combination use increases exposure to and effect of zolpidem. (7.2)
USE IN SPECIFIC POPULATIONS
- Pregnancy: Based on animal data, zolpidem may cause fetal harm. (8.1)
- Nursing mothers: Infant exposure via breast milk. (8.3)
- Pediatric use: Safety and effectiveness have not been established. Hallucinations (incidence rate 7.4%) and other psychiatric and/or nervous system adverse reactions were observed frequently in a study of pediatric patients with Attention-Deficit/Hyperactivity Disorder. (5.7, 8.4)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 8/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: COMPLEX SLEEP BEHAVIORS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosage in Adults
2.2 Special Populations
2.3 Use with CNS Depressants
2.4 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Complex Sleep Behaviors
5.2 CNS Depressant Effects and Next-Day Impairment
5.3 Need to Evaluate for Co-Morbid Diagnoses
5.4 Severe Anaphylactic and Anaphylactoid Reactions
5.5 Abnormal Thinking and Behavioral Changes
5.6 Withdrawal Effects
5.7 Special Populations
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 CNS-active Drugs
7.2 Drugs that Affect Drug Metabolism via Cytochrome P450
7.3 Other Drugs with No Interaction with Zolpidem
7.4 Drug-laboratory Test Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Labor and Delivery
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.2 Abuse
9.3 Dependence
10 OVERDOSAGE
10.1 Signs and Symptoms
10.2 Recommended Treatment
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Transient Insomnia
14.2 Chronic Insomnia
14.3 Studies Pertinent to Safety Concerns for Sedative-hypnotic Drugs
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: COMPLEX SLEEP BEHAVIORS
Complex sleep behaviors including sleep-walking, sleep-driving, and engaging in other activities while not fully awake may occur following use of ZOLPIMIST. Some of these events may result in serious injuries, including death. Discontinue ZOLPIMIST immediately if a patient experiences a complex sleep behavior [see Contraindications (4) and Warnings and Precautions (5.1)].
-
1 INDICATIONS AND USAGE
ZOLPIMIST (zolpidem tartrate) is indicated for the short-term treatment of insomnia characterized by difficulties with sleep initiation. Zolpidem tartrate has been shown to decrease sleep latency for up to 35 days in controlled clinical studies [see Clinical Studies (14)].
The clinical trials performed in support of efficacy were 4-5 weeks in duration with the final formal assessments of sleep latency performed at the end of treatment.
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosage in Adults
Use the lowest effective dose for the patient. The recommended initial dose is 5 mg for women and either 5 or 10 mg for men, taken only once per night immediately before bedtime with at least 7-8 hours remaining before the planned time of awakening. If the 5 mg dose is not effective, the dose can be increased to 10 mg. In some patients, the higher morning blood levels following use of the 10 mg dose increase the risk of next day impairment of driving and other activities that require full alertness [see Warnings and Precautions (5.1)]. The total dose of ZOLPIMIST should not exceed 10 mg once daily immediately before bedtime.
The recommended initial doses for women and men are different because zolpidem clearance is lower in women.
2.2 Special Populations
Elderly or debilitated patients may be especially sensitive to the effects of zolpidem tartrate. Patients with hepatic insufficiency do not clear the drug as rapidly as normal subjects. The recommended dose of ZOLPIMIST in both of these patient populations is 5 mg once daily immediately before bedtime [see Warnings and Precautions (5.7); Use in Specific Populations(8.5)].
2.3 Use with CNS Depressants
Dosage adjustment may be necessary when ZOLPIMIST is combined with other CNS-depressant drugs because of the potentially additive effects [see Warnings and Precautions (5.2)].
2.4 Administration
ZOLPIMIST is packaged in a child-resistant container. For detailed instructions on how to use ZOLPIMIST, refer to the Patient Instructions for Use (following the Medication Guide). ZOLPIMIST must be primed before it is used for the first time. To prime, patients should be told to point the black spray opening away from their face and other people and spray 5 times. For administration, the child-resistant container should be held upright with the black spray opening pointed directly into the mouth. The patient should fully press down on the pump to make sure a full dose (5 mg) of ZOLPIMIST is sprayed directly into the mouth over the tongue. If a 10 mg dose is prescribed, a second spray should be administered.
If the patient does not use ZOLPIMIST for at least 14 days, it must be primed again with 1 spray. The patient should be referred to the Patient Instructions for Use included at the end of the Medication Guide.
The effect of ZOLPIMIST may be slowed by ingestion with or immediately after a meal.
-
3 DOSAGE FORMS AND STRENGTHS
ZOLPIMIST is available as a clear, colorless, and cherry flavored solution designed to be sprayed directly into the mouth over the tongue. Each metered actuation (one spray) of ZOLPIMIST delivers 5 mg of zolpidem tartrate in 100 μL. Two actuations deliver 10 mg of zolpidem tartrate. After an initial priming of 5 actuations, there are 60 metered actuations in each child-resistant container. The total number of available doses is dependent on the number of actuations per dose (1 or 2 actuations) and the frequency of priming.
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Complex Sleep Behaviors
Complex sleep behaviors including sleep-walking, sleep-driving, and engaging in other activities while not fully awake may occur following the first or any subsequent use of zolpidem. Patients can be seriously injured or injure others during complex sleep behaviors. Such injuries may result in a fatal outcome. Other complex sleep behaviors (e.g., preparing and eating food, making phone calls, or having sex) have also been reported. Patients usually do not remember these events. Post-marketing reports have shown that complex sleep behaviors may occur with zolpidem alone at recommended dosages, with or without the concomitant use of alcohol or other central nervous system (CNS) depressants [see Drug Interactions (7.1)]. Discontinue ZOLPIMIST immediately if a patient experiences a complex sleep behavior.
5.2 CNS Depressant Effects and Next-Day Impairment
Zolpidem tartrate, like other sedative-hypnotic drugs, has CNS-depressant effects. Due to the rapid onset of action, ZOLPIMIST should only be administered immediately prior to going to bed. Patients should be cautioned against engaging in hazardous occupations requiring complete mental alertness or motor coordination such as operating machinery or driving a motor vehicle after ingesting the drug, including potential impairment of the performance of such activities that may occur the day following administration of ZOLPIMIST. Zolpidem tartrate showed additive effects when combined with alcohol and should not be taken with alcohol. Patients should also be cautioned about possible combined effects with other CNS-depressant drugs. Dosage adjustments may be necessary when ZOLPIMIST is administered with such agents because of the potentially additive effects.
Because ZOLPIMIST can cause drowsiness and a decreased level of consciousness, patients, particularly the elderly, are at higher risk of falls.
5.3 Need to Evaluate for Co-Morbid Diagnoses
Because sleep disturbances may be the presenting manifestation of a physical and/or psychiatric disorder, symptomatic treatment of insomnia should be initiated only after a careful evaluation of the patient. The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated. Worsening of insomnia or the emergence of new thinking or behavioral abnormalities may be the consequence of an unrecognized psychiatric or physical disorder. Such findings have emerged during the course of treatment with sedative-hypnotic drugs, including zolpidem.
5.4 Severe Anaphylactic and Anaphylactoid Reactions
Rare cases of angioedema involving the tongue, glottis, or larynx have been reported in patients after taking the first or subsequent doses of sedative-hypnotics, including zolpidem. Some patients have had additional symptoms such as dyspnea, throat closing, or nausea and vomiting that suggest anaphylaxis. Some patients have required medical therapy in the emergency department. If angioedema involves the throat, glottis, or larynx, airway obstruction may occur and be fatal. Patients who develop angioedema after treatment with zolpidem should not be rechallenged with the drug.
5.5 Abnormal Thinking and Behavioral Changes
A variety of abnormal thinking and behavioral changes have been reported to occur in association with the use of sedative-hypnotics. Some of these changes may be characterized by decreased inhibition (e.g., aggressiveness and extroversion that seemed out of character), similar to effects produced by alcohol and other CNS depressants. Visual and auditory hallucinations have been reported as well as behavioral changes such as bizarre behavior, agitation, and depersonalization. In controlled trials, <1% of adults with insomnia who received zolpidem reported hallucinations. In a clinical trial, 7.4% of pediatric patients with insomnia associated with attention-deficit/hyperactivity disorder (ADHD), who received zolpidem, reported hallucinations. [see Use in Specific Populations (8.4)]
It can rarely be determined with certainty whether a particular instance of the abnormal behaviors listed above is drug induced, spontaneous in origin, or a result of an underlying psychiatric or physical disorder. Nonetheless, the emergence of any new behavioral sign or symptom of concern requires careful and immediate evaluation.
5.6 Withdrawal Effects
Following the rapid dose decrease or abrupt discontinuation of sedative-hypnotics, there have been reports of signs and symptoms similar to those associated with withdrawal from other CNS-depressant drugs. [see Drug Abuse and Dependence (9)]
5.7 Special Populations
Use in the elderly and/or debilitated patients: Impaired motor and/or cognitive performance after repeated exposure or unusual sensitivity to sedative-hypnotic drugs is a concern in the treatment of elderly and/or debilitated patients. Therefore, the recommended ZOLPIMIST dosage is 5 mg in such patients to decrease the possibility of side effects. [see Dosage and Administration (2.2)] These patients should be closely monitored.
Use in patients with concomitant illness: Clinical experience with zolpidem tartrate in patients with concomitant systemic illness is limited. Caution is advisable in using ZOLPIMIST in patients with diseases or conditions that could affect metabolism or hemodynamic responses.
Although studies did not reveal respiratory depressant effects at hypnotic doses of zolpidem in normal subjects or in patients with mild to moderate chronic obstructive pulmonary disease (COPD), a reduction in the Total Arousal Index together with a reduction in lowest oxygen saturation and increase in the times of oxygen desaturation below 80% and 90% was observed in patients with mild-to-moderate sleep apnea when treated with zolpidem tartrate (10 mg) when compared to placebo. Since sedative-hypnotics have the capacity to depress respiratory drive, precautions should be taken if ZOLPIMIST is prescribed to patients with compromised respiratory function. Post- marketing reports of respiratory insufficiency, most of which involved patients with pre-existing respiratory impairment, have been received. ZOLPIMIST should be used with caution in patients with sleep apnea syndrome or myasthenia gravis.
Data in end-stage renal failure patients repeatedly treated with zolpidem tartrate did not demonstrate drug accumulation or alterations in pharmacokinetic parameters. No dosage adjustment in renally impaired patients is required; however, these patients should be closely monitored. [see Clinical Pharmacology (12.3)]
A study in subjects with hepatic impairment did reveal prolonged elimination in this group; therefore, treatment should be initiated with 5 mg in patients with hepatic compromise, and they should be closely monitored. [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)]
Use in patients with depression: As with other sedative-hypnotic drugs, ZOLPIMIST should be administered with caution to patients exhibiting signs or symptoms of depression. Suicidal tendencies may be present in such patients and protective measures may be required. Intentional over-dosage is more common in this group of patients; therefore, the least amount of drug that is feasible should be prescribed for the patient at any one time.
Use in pediatric patients: Safety and effectiveness of zolpidem have not been established in pediatric patients. In an 8-week study in pediatric patients (6-17 years of age) with insomnia associated with ADHD, zolpidem did not decrease sleep latency compared to placebo. Hallucinations were reported in 7.4% of the pediatric patients who received zolpidem tartrate; none of the pediatric patients who received placebo reported hallucinations. [see Use in Specific Populations (8.4)]
-
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Complex Sleep Behaviors [see Warnings and Precautions (5.1)]
- CNS-Depressant Effects and Next Day Impairment [see Warnings and Precautions (5.2)]
- Serious anaphylactic and anaphylactoid reactions [see Warnings and Precautions (5.4)]
- Abnormal Thinking and Behavioral Changes [see Warnings and Precautions (5.5)]
- Withdrawal Effects [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating incidence rates.
Associated with discontinuation of treatment: Approximately 4% of 1,701 patients who received zolpidem tartrate at all doses (1.25 to 90 mg) in U.S. premarketing clinical trials discontinued treatment because of an adverse reaction. Reactions most commonly associated with discontinuation from U.S. trials were daytime drowsiness (0.5%), dizziness (0.4%), headache (0.5%), nausea (0.6%), and vomiting (0.5%).
Approximately 4% of 1,959 patients who received zolpidem at all doses (1 to 50 mg) in similar foreign trials discontinued treatment because of an adverse reaction. Reactions most commonly associated with discontinuation from these trials were daytime drowsiness (1.1%), dizziness/vertigo (0.8%), amnesia (0.5%), nausea (0.5%), headache (0.4%), and falls (0.4%).
Data from a clinical study in which selective serotonin reuptake inhibitor (SSRI)-treated patients were given zolpidem revealed that four of the seven discontinuations during double-blind treatment with zolpidem (n=95) were associated with impaired concentration, continuing or aggravated depression, and manic reaction; one patient treated with placebo (n=97) was discontinued after an attempted suicide.
Most commonly observed adverse reactions in controlled trials: During short-term treatment (up to 10 nights) with zolpidem tartrate at doses up to 10 mg, the most commonly observed adverse reactions associated with the use of zolpidem and seen at statistically significant differences from placebo-treated patients were drowsiness (reported by 2% of zolpidem patients), dizziness (1%), and diarrhea (1%). During longer-term treatment (28 to 35 nights) with zolpidem tartrate at doses up to 10 mg, the most commonly observed adverse reactions associated with the use of zolpidem and seen at statistically significant differences from placebo-treated patients were dizziness (5%) and drugged feelings (3%).
Adverse reactions observed at an incidence of ≥1% in controlled trials: The following tables enumerate treatment-emergent adverse reactions frequencies that were observed at an incidence equal to 1% or greater among patients with insomnia who received zolpidem tartrate and at a greater incidence than placebo in U.S. placebo-controlled trials. Events reported by investigators were classified utilizing a modified World Health Organization (WHO) dictionary of preferred terms for the purpose of establishing event frequencies. The prescriber should be aware that these figures cannot be used to predict the incidence of side effects in the course of usual medical practice, in which patient characteristics and other factors differ from those that prevailed in these clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigators involving related drug products and uses, since each group of drug trials is conducted under a different set of conditions. However, the cited figures provide the physician with a basis for estimating the relative contribution of drug and nondrug factors to the incidence of side effects in the population studied.
The following table was derived from results of 11 placebo-controlled short-term U.S. efficacy trials involving zolpidem in doses ranging from 1.25 to 20 mg. The table is limited to data from doses up to and including 10 mg, the highest dose recommended for use.
Incidence of Treatment-Emergent Adverse Reactions in Placebo-Controlled Clinical Trials Lasting up to 10 Nights (Percentage of patients reporting) Body System/Adverse Reaction* Zolpidem
(≤10mg)
(n=685)Placebo
(n=473)*Reactions reported by at least 1% of patients treated with zolpidem tartrate and at a greater frequency than placebo.
Central and Peripheral Nervous System Headache 7 6 Drowsiness 2 - Dizziness 1 - Gastrointestinal System Diarrhea 1 - The following table was derived from results of three placebo-controlled long-term efficacy trials involving zolpidem tartrate. These trials involved patients with chronic insomnia who were treated for 28 to 35 nights with zolpidem tartrate at doses of 5, 10, or 15 mg. The table is limited to data from doses up to and including 10 mg, the highest dose recommended for use. The table includes only adverse reactions occurring at an incidence of at least 1% for zolpidem tartrate patients.
Incidence of Treatment-Emergent Adverse Reactions in Placebo-Controlled Clinical Trials Lasting up to 35 Nights (Percentage of patients reporting) Body System/Adverse Reaction* Zolpidem
(≤10mg)
(n=152)Placebo
(n=161)*Reactions reported by at least 1% of patients treated with zolpidem tartrate and at a greater frequency than placebo.
Autonomic Nervous System Dry mouth 3 1 Body as a Whole Allergy 4 1 Back pain 3 2 Influenza-like symptoms 2 - Chest pain 1 - Cardiovascular System Palpitation 2 - Central and Peripheral Nervous System Drowsiness 8 5 Dizziness 5 1 Lethargy 3 1 Drugged feeling 3 - Lightheadedness 2 1 Depression 2 1 Abnormal dreams 1 - Amnesia 1 - Sleep disorder 1 - Gastrointestinal System Diarrhea 3 2 Abdominal pain 2 2 Constipation 2 1 Respiratory System Sinusitis 4 2 Pharyngitis 3 1 Skin and Appendages Rash 2 1 Dose relationship for adverse reactions: There is evidence from dose comparison trials suggesting a dose relationship for many of the adverse reactions associated with zolpidem tartrate use, particularly for certain CNS and gastrointestinal adverse reactions.
Oral tissue-related adverse reactions in ZOLPIMIST pharmacokinetics studies: The effect of chronic daily administrations of ZOLPIMIST on oral tissue has not been evaluated. In pharmacokinetic studies conducted with ZOLPIMIST in healthy subjects, an oral soft tissue exam was performed, and no signs of oral irritation were noted following administration of single doses of ZOLPIMIST.
Adverse event incidence across the entire preapproval database: Zolpidem tartrate was administered to 3,660 subjects in clinical trials throughout the United States, Canada, and Europe. Treatment-emergent adverse event associated with clinical trial participation were recorded by clinical investigators using terminology of their own choosing. To provide a meaningful estimate of the proportion of individuals experiencing treatment-emergent adverse events, similar types of untoward events were grouped into a smaller number of standardized event categories and classified utilizing a modified WHO dictionary of preferred terms.
The frequencies presented, therefore, represent the proportions of the 3,660 individuals exposed to zolpidem tartrate, at all doses, who experienced an event of the type cited on at least one occasion while receiving zolpidem tartrate. All reported treatment-emergent adverse events are included, except those already listed in the table above of adverse events in placebo-controlled studies, those coding terms that are so general as to be uninformative, and those events where a drug cause was remote. It is important to emphasize that, although the events reported did occur during treatment with zolpidem tartrate, they were not necessarily caused by it.
Adverse events are further classified within body system categories and enumerated in order of decreasing frequency using the following definitions: frequent adverse events are defined as those occurring in greater than 1/100 subjects; infrequent adverse events are those occurring in 1/100 to 1/1,000 patients; rare events are those occurring in less than 1/1,000 patients.
Autonomic nervous system: Infrequent: increased sweating, pallor, postural hypotension, syncope. Rare: abnormal accommodation, altered saliva, flushing, glaucoma, hypotension, impotence, increased saliva, tenesmus.
Body as a whole: Frequent: asthenia. Infrequent: edema, falling, fatigue, fever, malaise, trauma. Rare: allergic reaction, allergy aggravated, anaphylactic shock, face edema, hot flashes, increased ESR, pain, restless legs, rigors, tolerance increased, weight decrease.
Cardiovascular system: Infrequent: cerebrovascular disorder, hypertension, tachycardia. Rare: angina pectoris, arrhythmia, arteritis, circulatory failure, extrasystoles, hypertension aggravated, myocardial infarction, phlebitis, pulmonary embolism, pulmonary edema, varicose veins, ventricular tachycardia.
Central and peripheral nervous system: Frequent: ataxia, confusion, euphoria, headache, insomnia, vertigo. Infrequent: agitation, anxiety, decreased cognition, detached, difficulty concentrating, dysarthria, emotional lability, hallucination, hypoesthesia, illusion, leg cramps, migraine, nervousness, paresthesia, sleeping (after daytime dosing), speech disorder, stupor, tremor. Rare: abnormal gait, abnormal thinking, aggressive reaction, apathy, appetite increased, decreased libido, delusion, dementia, depersonalization, dysphasia, feeling strange, hypokinesia, hypotonia, hysteria, intoxicated feeling, manic reaction, neuralgia, neuritis, neuropathy, neurosis, panic attacks, paresis, personality disorder, somnambulism, suicide attempts, tetany, yawning.
Gastrointestinal system: Frequent: dyspepsia, hiccup, nausea. Infrequent: anorexia, constipation, dysphagia, flatulence, gastroenteritis, vomiting. Rare: enteritis, eructation, esophagospasm, gastritis, hemorrhoids, intestinal obstruction, rectal hemorrhage, tooth caries.
Hematologic and lymphatic system: Rare: anemia, hyperhemoglobinemia, leukopenia, lymphadenopathy, macrocytic anemia, purpura, thrombosis.
Immunologic system: Infrequent: infection. Rare: abscess, herpes simplex, herpes zoster, otitis externa, otitis media.
Liver and biliary system: Infrequent: abnormal hepatic function, increased SGPT. Rare: bilirubinemia, increased SGOT.
Metabolic and nutritional: Infrequent: hyperglycemia, thirst. Rare: gout, hypercholesteremia, hyperlipidemia, increased alkaline phosphatase, increased BUN, periorbital edema.
Musculoskeletal system: Frequent: arthralgia, myalgia. Infrequent: arthritis. Rare: arthrosis, muscle weakness, sciatica, tendonitis.
Reproductive system: Infrequent: menstrual disorder, vaginitis. Rare: breast fibroadenosis, breast neoplasm, breast pain.
Respiratory system: Frequent: upper respiratory infection, lower respiratory infection. Infrequent: bronchitis, coughing, dyspnea, rhinitis. Rare: bronchospasm, respiratory depression, epistaxis, hypoxia, laryngitis, pneumonia.
Skin and appendages: Infrequent: pruritus. Rare: acne, bullous eruption, dermatitis, furunculosis, injection-site inflammation, photosensitivity reaction, urticaria.
Special senses: Frequent: diplopia, vision abnormal. Infrequent: eye irritation, eye pain, scleritis, taste perversion, tinnitus. Rare: conjunctivitis, corneal ulceration, lacrimation abnormal, parosmia, photopsia.
Urogenital system: Frequent: urinary tract infection. Infrequent: cystitis, urinary incontinence. Rare: acute renal failure, dysuria, micturition frequency, nocturia, polyuria, pyelonephritis, renal pain, urinary retention.
-
7 DRUG INTERACTIONS
7.1 CNS-active Drugs
Since the systematic evaluations of zolpidem in combination with other CNS-active drugs have been limited, careful consideration should be given to the pharmacology of any CNS-active drug to be used with ZOLPIMIST. Any drug with CNS-depressant effects could potentially enhance the CNS-depressant effects of zolpidem.
Zolpidem tartrate was evaluated in healthy subjects in single-dose interaction studies for several CNS drugs. Imipramine in combination with zolpidem produced no pharmacokinetic interaction other than a 20% decrease in peak levels of imipramine, but there was an additive effect of decreased alertness. Similarly, chlorpromazine in combination with zolpidem tartrate produced no pharmacokinetic interaction, but there was an additive effect of decreased alertness and psychomotor performance. A study involving haloperidol and zolpidem revealed no effect of haloperidol on the pharmacokinetics or pharmacodynamics of zolpidem. The lack of a drug interaction following single-dose administration does not predict a lack following chronic administration.
An additive effect on psychomotor performance between alcohol and zolpidem was demonstrated [see Warnings and Precautions (5.1, 5.2)].
A single-dose interaction study with zolpidem 10 mg and fluoxetine 20 mg at steady-state levels in male volunteers did not demonstrate any clinically significant pharmacokinetic or pharmacodynamic interactions. When multiple doses of zolpidem and fluoxetine at steady-state concentrations were evaluated in healthy females, the only significant change was a 17% increase in the zolpidem half-life (t1/2). There was no evidence of an additive effect in psychomotor performance.
Following five consecutive nightly doses of zolpidem 10 mg in the presence of sertraline 50 mg (17 consecutive daily doses at 7:00 am in healthy female volunteers), zolpidem maximum concentration (Cmax) was significantly higher (43%) and time to maximum concentration (Tmax) was significantly decreased (53%). Pharmacokinetics of sertraline and N-desmethylsertraline were unaffected by zolpidem.
7.2 Drugs that Affect Drug Metabolism via Cytochrome P450
Some compounds known to inhibit CYP3A may increase exposure to zolpidem. The effect of inhibitors of other P450 enzymes has not been carefully evaluated. A randomized, double-blind, crossover interaction study in ten healthy volunteers between itraconazole (200 mg once daily for 4 days) and a single dose of zolpidem (10 mg) given 5 hours after the last dose of itraconazole resulted in a 34% increase in AUC0–∞ of zolpidem. There were no significant pharmacodynamic effects of zolpidem on subjective drowsiness, postural sway, or psychomotor performance.
A randomized, placebo-controlled, crossover interaction study in eight healthy female subjects between five consecutive daily doses of rifampin (600 mg) and a single dose of zolpidem (20 mg) given 17 hours after the last dose of rifampin showed significant reductions of the AUC (– 73%), Cmax (–58%), and t1/2 (–36%) of zolpidem, together with significant reductions in the pharmacodynamic effects of zolpidem.
A randomized double-blind crossover interaction study in twelve healthy subjects showed that co-administration of single 5 mg dose of zolpidem tartrate with ketoconazole, a potent CYP3A4 inhibitor, given as 200 mg twice daily for 2 days increased Cmax of zolpidem by a factor of 1.3 and increased the total AUC of zolpidem by a factor of 1.7 compared to zolpidem alone and prolonged the t1/2 by approximately 30% along with an increase in the pharmacodynamic effects of zolpidem. Caution should be used when ketoconazole is given with zolpidem and consideration should be given to using a lower dose of zolpidem when ketoconazole and zolpidem are given together. Patients should be advised that use of ZOLPIMIST with ketoconazole may enhance the sedative effects.
7.3 Other Drugs with No Interaction with Zolpidem
A study involving cimetidine/zolpidem and ranitidine/zolpidem combinations revealed no effect of either drug on the pharmacokinetics or pharmacodynamics of zolpidem.
Zolpidem had no effect on digoxin pharmacokinetics and did not affect prothrombin time when given with warfarin in normal subjects.
7.4 Drug-laboratory Test Interactions
Zolpidem is not known to interfere with commonly employed clinical laboratory tests. In addition, clinical data indicate that zolpidem does not cross-react with benzodiazepines, opiates, barbiturates, cocaine, cannabinoids, or amphetamines in two standard urine drug screens.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
There are no adequate and well-controlled studies of ZOLPIMIST in pregnant women.
Studies in children to assess the effects of prenatal exposure to zolpidem have not been conducted; however, cases of severe neonatal respiratory depression have been reported when zolpidem was used at the end of pregnancy, especially when taken with other CNS depressants. Children born to mothers taking sedative-hypnotic drugs may be at risk for withdrawal symptoms during the postnatal period. Neonatal flaccidity has also been reported in infants born to mothers who received sedative-hypnotic drugs during pregnancy. ZOLPIMIST should be used during pregnancy only if the potential benefit outweighs the potential risk to the fetus.
Administration of zolpidem to pregnant rats and rabbits resulted in adverse effects on offspring development at doses greater than the zolpidem tartrate maximum recommended human dose (MRHD) of 10 mg/day (approximately 8 mg/day zolpidem base); however, teratogenicity was not observed.
When zolpidem was administered at oral doses of 4, 20, and 100 mg base/kg/day to pregnant rats during the period of organogenesis, dose-related decreases in fetal skull ossification occurred at all but the lowest dose, which is approximately 5 times the MRHD on a mg/m2 basis. In rabbits treated during organogenesis with zolpidem at oral doses of 1, 4, and 16 mg base/kg/day, increased embryo-fetal death and incomplete fetal skeletal ossification occurred at the highest dose tested. The no-effect dose for embryo-fetal toxicity in rabbits is approximately 10 times the MRHD on a mg/m2 basis. Administration of zolpidem to rats at oral doses of 4, 20, and 100 mg base/kg/day during the latter part of pregnancy and throughout lactation produced decreased offspring growth and survival at all but the lowest dose, which is approximately 5 times the MRHD on a mg/m2 basis.
8.2 Labor and Delivery
ZOLPIMIST has no established use in labor and delivery. [see Pregnancy (8.1)]
8.3 Nursing Mothers
Zolpidem is excreted in human milk. Studies in lactating mothers indicate that the t1/2 of zolpidem is similar to that in non-lactating women (2.6 ± 0.3 hours). Between 0.004% and 0.019% of the total administered dose is excreted into milk. The effect of zolpidem on the nursing infant is not known.
8.4 Pediatric Use
ZOLPIMIST is not recommended for use in children. Safety and effectiveness of zolpidem in pediatric patients below the age of 18 years have not been established.
In an 8-week controlled study of pediatric patients (6-17 years of age) with insomnia associated with attention-deficit/hyperactivity-disorder (ADHD) an oral solution of zolpidem tartrate dosed at 0.25 mg/kg at bedtime did not decrease sleep latency compared to placebo. Psychiatric and nervous system disorders comprised the most frequent (>5%) treatment emergent adverse reactions observed with zolpidem versus placebo and included dizziness (23.5% vs. 1.5%), headache (12.5% vs. 9.2%), and hallucinations were reported in 7% of the pediatric patients who received zolpidem; none of the pediatric patients who received placebo reported hallucinations. [see Warnings and Precautions (5.7)] Ten patients on zolpidem (7.4%) discontinued treatment due to an adverse reaction.
8.5 Geriatric Use
A total of 154 patients in U.S. controlled clinical trials and 897 patients in non-U.S. clinical trials who received oral zolpidem were ≥60 years of age. For a pool of U.S. patients receiving zolpidem tartrate at doses of ≤10 mg or placebo, there were three adverse reactions occurring at an incidence of at least 3% for zolpidem and for which the zolpidem incidence was at least twice the placebo incidence (i.e., they could be considered drug related).
Adverse Reaction Zolpidem Placebo Dizziness 3% 0% Drowsiness 5% 2% Diarrhea 3% 1% A total of 30/1,959 (1.5%) non-U.S. patients receiving zolpidem reported falls, including 28/30 (93%) who were ≥70 years of age. Of these 28 patients, 23 (82%) were receiving zolpidem doses >10 mg. A total of 24/1,959 (1.2%) non-U.S. patients receiving zolpidem reported confusion, including 18/24 (75%) who were ≥70 years of age. Of these 18 patients, 14 (78%) were receiving zolpidem doses >10 mg.
The dose of ZOLPIMIST in elderly patients is 5 mg to minimize the adverse effects related to impaired motor and/or cognitive performance and unusual sensitivity to sedative-hypnotic drugs. [see Warnings and Precautions (5.7)]
-
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Zolpidem tartrate is classified as a Schedule IV controlled substance by federal regulation.
9.2 Abuse
Abuse and addiction are separate and distinct from physical dependence and tolerance. Abuse is characterized by misuse of the drug for non-medical purposes, often in combination with other psychoactive substances. Tolerance is a state of adaptation in which exposure to a drug induces changes that result in a diminution of one or more of the drug effects over time. Tolerance may occur to both desired and undesired effects of drugs and may develop at different rates for different effects.
Addiction is a primary, chronic, neurobiological disease with genetic, psychosocial, and environmental factors influencing its development and manifestations. It is characterized by behaviors that include one or more of the following: impaired control over drug use, compulsive use, continued use despite harm, and craving. Drug addiction is a treatable disease, using a multidisciplinary approach, but relapse is common.
Studies of abuse potential in former drug abusers found that the effects of single doses of zolpidem tartrate 40 mg were similar, but not identical, to diazepam 20 mg, while zolpidem tartrate 10 mg was difficult to distinguish from placebo.
Because persons with a history of addiction to, or abuse of, drugs or alcohol are at increased risk for misuse, abuse and addiction of zolpidem, they should be monitored carefully when receiving zolpidem or any other hypnotic.
9.3 Dependence
Physical dependence is a state of adaptation that is manifested by a specific withdrawal syndrome that can be produced by abrupt cessation, rapid dose reduction, decreasing blood level of the drug, and/or administration of an antagonist.
Sedative-hypnotics have produced withdrawal signs and symptoms following abrupt discontinuation. These reported symptoms range from mild dysphoria and insomnia to a withdrawal syndrome that may include abdominal and muscle cramps, vomiting, sweating, tremors, and convulsions. The following adverse reactions which are considered to meet the DSM-III-R criteria for uncomplicated sedative-hypnotic withdrawal were reported during U.S. clinical trials following placebo substitution occurring within 48 hours following last zolpidem treatment: fatigue, nausea, flushing, lightheadedness, uncontrolled crying, emesis, stomach cramps, panic attack, nervousness, and abdominal discomfort. These reported adverse reactions occurred at an incidence of 1% or less. However, available data cannot provide a reliable estimate of the incidence, if any, of dependence during treatment at recommended doses. Post-marketing reports of abuse, dependence, and withdrawal have been received.
-
10 OVERDOSAGE
10.1 Signs and Symptoms
In post-marketing experience of overdose with zolpidem tartrate alone, or in combination with CNS-depressant agents, impairment of consciousness ranging from somnolence to coma, cardiovascular and/or respiratory compromise, and fatal outcomes have been reported.
10.2 Recommended Treatment
General symptomatic and supportive measures should be used along with immediate gastric lavage where appropriate. Intravenous fluids should be administered as needed. Zolpidem's sedative-hypnotic effect was shown to be reduced by flumazenil and therefore may be useful; however, flumazenil administration may contribute to the appearance of neurological symptoms (convulsions). As in all cases of drug overdose, respiration, pulse, blood pressure, and other appropriate signs should be monitored, and general supportive measures employed. Hypotension and CNS depression should be monitored and treated by appropriate medical intervention. Sedating drugs should be withheld following zolpidem overdosage, even if excitation occurs. The value of dialysis in the treatment of overdosage has not been determined, although hemodialysis studies in patients with renal failure receiving therapeutic doses have demonstrated that zolpidem is not dialyzable.
As with the management of all overdosage, the possibility of multiple drug ingestion should be considered. The physician may wish to consider contacting a poison control center for up-to-date information on the management of hypnotic drug product overdosage.
-
11 DESCRIPTION
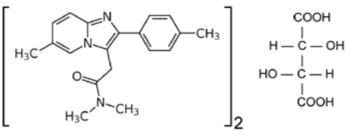
ZOLPIMIST (zolpidem tartrate) is a gamma-aminobutyric acid (GABA) A agonist of the imidazopyridine class. Chemically, zolpidem is N,N,6-trimethyl-2-p-tolylimidazo[1,2-a] pyridine-3-acetamide l-(+)-tartrate (2:1). It has the following structure:
Zolpidem tartrate is a white to off-white crystalline powder that is sparingly soluble in water, alcohol, and propylene glycol. It has a molecular weight of 764.89.
ZOLPIMIST is available as an oral solution designed to be sprayed directly into the mouth over the tongue. Each metered actuation of ZOLPIMIST delivers 5 mg of zolpidem tartrate in 100 μL. Two actuations deliver 10 mg of zolpidem tartrate. ZOLPIMIST includes the following inactive ingredients: artificial cherry flavor, benzoic acid, citric acid monohydrate, hydrochloric acid, neotame, propylene glycol, and purified water.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Zolpidem, the active moiety of zolpidem tartrate, is a hypnotic agent with a chemical structure unrelated to benzodiazepines, barbiturates, or other drugs with known hypnotic properties. It interacts with a GABA-BZ receptor complex and shares some of the pharmacological properties of the benzodiazepines. In contrast to the benzodiazepines which non-selectively bind to and activate all BZ receptor subtypes, zolpidem in vitro binds the BZ1 receptor preferentially with a high affinity ratio of the α1/α5 subunits. This selective binding of zolpidem on the BZ1 receptor is not absolute, but it may explain the relative absence of myorelaxant and anticonvulsant effects in animal studies as well as the preservation of deep sleep (stages 3 and 4) in human studies of zolpidem at hypnotic doses.
12.3 Pharmacokinetics
ZOLPIMIST (zolpidem tartrate) Oral Spray is bioequivalent to Ambien® tablets (Sanofi-Aventis). The pharmacokinetic profile of ZOLPIMIST is characterized by rapid absorption from the oral mucosa and gastrointestinal tract, and a short elimination t1/2 in healthy subjects.
In a single-dose crossover study in 10 healthy young (18-40 years of age) male subjects administered 2.5, 5, and 10 mg ZOLPIMIST, the results demonstrated a linear relationship to dose for mean Cmax and AUC0-∞ over the range of doses administered in the study.
In a single-dose crossover study in 43 healthy young (18-45 years of age) subjects administered 5 and 10 mg ZOLPIMIST, the means for Cmax were 114 (range: 19 to 197) and 210 ng/mL (range: 77 to 401), respectively, occurring at a mean time to maximum concentration (Tmax) of approximately 0.9 hours for both. The mean zolpidem t1/2 was 2.7 (range: 1.7 to 5.0) and 3.0 hours (range: 1.7 to 8.4), for 5 and 10 mg ZOLPIMIST, respectively. In the same study, the means for Cmax were 123 (range: 53 to 221) and 219 ng/mL (range: 101 to 446) for 5 and 10 mg Ambien® tablets, respectively, occurring at a mean Tmax of 0.9 and 1.0 hours, respectively. The mean zolpidem t1/2 was 2.8 (range: 1.5 to 6.0) and 3.1 hours (range: 1.1 to 8.6) for the 5 and 10 mg Ambien® tablets, respectively.
Zolpidem is converted to inactive metabolites that are eliminated primarily by renal excretion. Total protein binding for zolpidem was found to be 92.5 ± 0.1% and remained constant, independent of concentration between 40 and 790 ng/mL. Zolpidem did not accumulate in young adults following nightly dosing with 20 mg zolpidem tartrate for 2 weeks.
A food-effect crossover study in 14 healthy young (18-45 years of age) male subjects compared the pharmacokinetics of ZOLPIMIST 10 mg when administered while fasting at least 8 hours or 5 minutes after eating a standard high-fat meal. Results demonstrated that with food, mean AUC0-∞ and Cmax were decreased by 27% and 58%, respectively, while mean Tmax was prolonged by 225% (from 0.8 to 2.6 hours). These results suggest that, for faster sleep onset, as with all zolpidem products, ZOLPIMIST should not be administered with or immediately after a meal.
Special Populations:
Elderly: In the elderly, the dose for zolpidem tartrate should be 5 mg [see Warnings and Precautions (5) and Dosage and Administration (2)]. This recommendation is based on several studies in which the mean Cmax, t1/2, and AUC were significantly increased when compared to results in young adults administered zolpidem tartrate. In a pharmacokinetic study of 24 elderly (≥65 years of age) subjects administered 5 mg ZOLPIMIST, the means for Cmax and AUC were 134 ng/mL and 493 ng∙hr/mL respectively, following administration of a single 5 mg oral dose of ZOLPIMIST. Zolpidem tartrate did not accumulate in elderly subjects following nightly oral dosing of 10 mg for 1 week.
Hepatic impairment: The pharmacokinetics of zolpidem in eight patients with chronic hepatic insufficiency were compared to results in healthy subjects. Following a single 20 mg oral zolpidem tartrate dose, mean Cmax and AUC were found to be two times (250 vs 499 ng/mL) and five times (788 vs 4,203 ng∙hr/mL) higher, respectively, in hepatically compromised patients. Tmax did not change. The mean t1/2 in cirrhotic patients of 9.9 hours (range: 4.1 to 25.8 hours) was greater than that observed in normal subjects of 2.2 hours (range: 1.6 to 2.4 hours). Dosing should be modified accordingly in patients with hepatic insufficiency. [see Dosage and Administration (2.2)]
Renal impairment: The pharmacokinetics of zolpidem were studied in 11 patients with end-stage renal failure (mean Cl Cr=6.5 ± 1.5 mL/min) undergoing hemodialysis three times a week, who were dosed with zolpidem tartrate 10 mg orally each day for 14 or 21 days. No statistically significant differences were observed for Cmax, Tmax, t1/2, and AUC between the first and last day of drug administration when baseline concentration adjustments were made. Zolpidem was not hemodialyzable. No accumulation of unchanged drug appeared after 14 or 21 days. Zolpidem pharmacokinetics were not significantly different in renally impaired patients. No dosage adjustment is necessary in patients with compromised renal function.
No dosage adjustment is necessary in patients with compromised renal function. However, as a general precaution, these patients should be closely monitored.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis: Zolpidem was administered to mice and rats for 2 years at oral doses of 4, 18, and 80 mg base/kg/day. In mice, these doses are approximately 2.5, 10, and 50 times the maximum recommended human dose (MRHD) of 10 mg/day (8 mg zolpidem base) on mg/m2 basis. In rats, these doses are approximately 5, 20, and 100 times the MRHD on a mg/m2 basis. No evidence of carcinogenic potential was observed in mice. In rats, renal tumors (lipoma, liposarcoma) were seen at the mid- and high doses.
Mutagenesis: Zolpidem was negative in in vitro (bacterial reverse mutation, mouse lymphoma, and chromosomal aberration) and in vivo (mouse micronucleus) genetic toxicology assays.
Impairment of fertility: Oral administration of zolpidem (doses of 4, 20, and 100 mg base/kg/day) to rats prior to and during mating, and continuing in females through postpartum day 25, resulted in irregular estrus cycles and prolonged precoital intervals at the highest dose tested. The no-effect dose for these findings is approximately 24 times the MRHD on a mg/m2 basis. There was no impairment of fertility at any dose tested.
-
14 CLINICAL STUDIES
14.1 Transient Insomnia
Normal adults experiencing transient insomnia (n=462) during the first night in a sleep laboratory were evaluated in a double-blind, parallel group, single-night trial comparing two doses of zolpidem (7.5 and 10 mg) and placebo. Both zolpidem doses were superior to placebo on objective (polysomnographic) measures of sleep latency, sleep duration, and number of awakenings.
Normal elderly adults (mean age 68) experiencing transient insomnia (n=35) during the first two nights in a sleep laboratory were evaluated in a double-blind, crossover, 2-night trial comparing four doses of zolpidem (5, 10, 15, and 20 mg) and placebo. All zolpidem doses were superior to placebo on the two primary PSG parameters (sleep latency and efficiency) and all four subjective outcome measures (sleep duration, sleep latency, number of awakenings, and sleep quality).
14.2 Chronic Insomnia
Zolpidem was evaluated in two controlled studies for the treatment of patients with chronic insomnia (most closely resembling primary insomnia, as defined in the APA Diagnostic and Statistical Manual of Mental Disorders, DSM-IV™). Adult outpatients with chronic insomnia (n=75) were evaluated in a double-blind, parallel group, 5-week trial comparing two doses of zolpidem tartrate and placebo. On objective (polysomnographic) measures of sleep latency and sleep efficiency, zolpidem 10 mg was superior to placebo on sleep latency for the first 4 weeks and on sleep efficiency for weeks 2 and 4. Zolpidem was comparable to placebo on number of awakenings at both doses studied.
Adult outpatients (n=141) with chronic insomnia were also evaluated, in a double-blind, parallel group, 4-week trial comparing two doses of zolpidem and placebo. Zolpidem 10 mg was superior to placebo on a subjective measure of sleep latency for all 4 weeks, and on subjective measures of total sleep time, number of awakenings, and sleep quality for the first treatment week.
Increased wakefulness during the last third of the night as measured by polysomnography has not been observed in clinical trials with zolpidem.
14.3 Studies Pertinent to Safety Concerns for Sedative-hypnotic Drugs
Next-day residual effects: Next-day residual effects of zolpidem tartrate were evaluated in seven studies involving normal subjects. In three studies in adults (including one study in a phase advance model of transient insomnia) and in one study in elderly subjects, a small but statistically significant decrease in performance was observed in the Digit Symbol Substitution Test (DSST) when compared to placebo. Studies of zolpidem tartrate in non-elderly patients with insomnia did not detect evidence of next-day residual effects using the DSST, the Multiple Sleep Latency Test (MSLT), and patient ratings of alertness.
Rebound effects: There was no objective (polysomnographic) evidence of rebound insomnia at recommended doses seen in studies evaluating sleep on the nights following discontinuation of zolpidem tartrate. There was subjective evidence of impaired sleep in the elderly on the first post-treatment night at doses above the recommended elderly dose of 5 mg.
Memory impairment: Controlled studies in adults utilizing objective measures of memory yielded no consistent evidence of next-day memory impairment following the administration of zolpidem tartrate. However, in one study involving zolpidem doses of 10 and 20 mg, there was a significant decrease in next-morning recall of information presented to subjects during peak drug effect (90 minutes post-dose) (i.e., these subjects experienced anterograde amnesia). There was also subjective evidence from adverse event data for anterograde amnesia occurring in association with the administration of zolpidem tartrate, predominantly at doses above 10 mg.
Effects on sleep stages: In studies that measured the percentage of sleep time spent in each sleep stage, zolpidem tartrate has generally been shown to preserve sleep stages. Sleep time spent in stages 3 and 4 (deep sleep) was found comparable to placebo with only inconsistent, minor changes in REM (paradoxical) sleep at the recommended dose.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
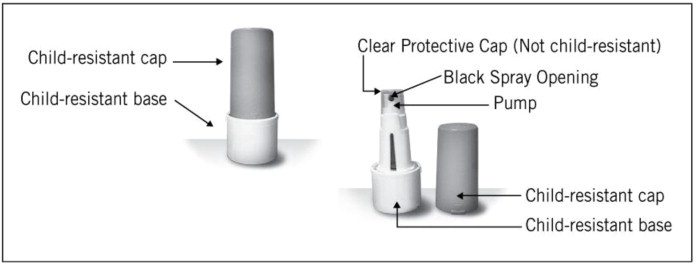
ZOLPIMIST is available in a child-resistant container. Each container includes a child-resistant cap and base with a metered-dose pump assembly and clear cover cap. There are two different fill volumes. One container contains 4.5 ml (4.8 g) of product formulation and the other 7.7 ml (8.22 g). One and two actuations of ZOLPIMIST from either container are equal to 5 and 10 mg of zolpidem tartrate, respectively. There are 30 metered actuations per container after 5 initial priming actuations in the 4.5 ml container and 60 metered actuations in the 7.7 ml container. ZOLPIMIST is supplied as:
NDC Number
69654-0510-30 - Carton includes a child-resistant container with 4.5 mL (4.8 g) of product formulation; 30 metered actuations per container.69654-0510-60 - Carton includes a child-resistant container with 7.7 mL (8.22 g) of product formulation; 60 metered actuations per container.
Store upright at 25 °C (77 °F) with excursions permitted to 15-30 °C (59-86 °F) (USP Controlled Room Temperature). Do not freeze. Avoid prolonged product exposure to temperatures above 30 °C (86 °F). The child-resistant container should be discarded when the labeled number of actuations (30 or 60 sprays for the 4.5 ml and 7.7 ml bottles respectively) have been used.
KEEP OUT OF REACH OF CHILDREN.
-
17 PATIENT COUNSELING INFORMATION
Prescribers or other healthcare professionals should inform patients, their families, and their caregivers about the benefits and risks associated with treatment with sedative-hypnotics, should counsel them in its appropriate use, and should instruct them to read the accompanying Medication Guide and Patient Instructions for Use [see Medication Guide and Patient Instructions for Use].
Complex Sleep Behaviors
Instruct patients and their families that ZOLPIMIST may cause complex sleep behaviors, including sleep-walking, sleep-driving, preparing and eating food, making phone calls, or having sex while not being fully awake. Serious injuries and death have occurred during complex sleep behavior episodes. Tell patients to discontinue ZOLPIMIST and notify their healthcare provider immediately if they develop any of these symptoms [see Boxed Warning, Warnings and Precautions (5.1)].
CNS-Depressant Effects and Next-Day Impairment
Inform patients that ZOLPIMIST should be administered immediately prior to going to bed. Caution patients against engaging in hazardous occupations requiring complete mental alertness or motor coordination such as driving. Inform patients that potential impairment of the performance of such activities may occur the day following administration of ZOLPIMST. Advise patients that increased drowsiness and decreased consciousness may increase the risk of falls in some patients [see Warnings and Precautions (5.2)].
Severe Anaphylactic and Anaphylactoid Reactions
Inform patients that severe anaphylactic and anaphylactoid reactions have occurred with zolpidem. Describe the signs/symptoms of these reactions and advise patients to seek medical attention immediately if any of them occur. [see Warnings and Precautions (5.4)]
Administration Instructions
See the Dosage and Administration section [see Administration (2.4)]. ZOLPIMIST is packaged in a child-resistant container. Patients should be referred to the Patient Instructions for Use (following the Medication Guide) for detailed instructions on how to use ZOLPIMIST. Patients should be counseled to take ZOLPIMIST right before they get into bed and only when they are able to stay in bed a full night (7-8 hours) before being active again. ZOLPIMIST should not be taken with or immediately after a meal. Advise patients NOT to take ZOLPIMIST when drinking alcohol.
-
MEDICATION GUIDE
MEDICATION GUIDE
ZOLPIMIST Oral Spray (C-IV)
(zolpidem tartrate) Spray, Metered for Oral Use
Read the Medication Guide that comes with ZOLPIMIST before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your doctor about your medical condition or treatment.
What is the most important information I should know about ZOLPIMIST?
Complex sleep behaviors that have caused serious injury and death. After taking ZOLPIMIST, you may get up out of bed while not being fully awake and do an activity that you do not know you are doing (complex sleep behaviors). The next morning, you may not remember that you did anything during the night. These activities may happen whether or not you drink alcohol or take other medicines that make you sleepy with ZOLPIMIST. Reported activities include:
- driving a car (“sleep-driving”)
- making and eating food
- talking on the phone
- having sex
- sleep-walking
Stop taking Zolpimist and call your doctor right away if you find out that you have done any of the above activities after taking ZOLPIMIST.
Important:
1. Take ZOLPIMIST exactly as prescribed
- Do not take more ZOLPIMIST than prescribed.
- Take ZOLPIMIST right before you get in bed, not sooner.
2. Do not take ZOLPIMIST if you:
- have ever experienced a complex sleep behavior (such as driving a car, making and eating food, talking on the phone or having sex while not fully awake) after taking ZOLPIMIST
- drink alcohol
- take other medicines that can make you sleepy. Talk to your doctor about all of your medicines. Your doctor will tell you if you can take ZOLPIMIST with your other medicines.
- cannot get a full night sleep
What is ZOLPIMIST?
ZOLPIMIST is a sedative-hypnotic (sleep) medicine. ZOLPIMIST is used in adults for the short-term treatment of a sleep problem called insomnia. Symptoms of insomnia include:
- trouble falling asleep
ZOLPIMIST is not for children.
ZOLPIMIST is a federally controlled substance (C-IV) because it can be abused and lead to dependence. Keep ZOLPIMIST in a safe place to prevent misuse and abuse. Selling or giving away ZOLPIMIST may harm others and is against the law. Tell your doctor if you have ever abused or have been dependent on alcohol, prescription medicines, or street drugs.
Who should not take ZOLPIMIST? Do not take ZOLPIMIST if you have had an allergic reaction to zolpidem (Ambien, Ambien CR, ZOLPIMIST). Some signs of allergic reaction may be swelling of the face, a feeling of the throat closing, or difficulty breathing shortly after taking Zolpidem.
See the end of this Medication Guide for a complete list of ingredients in ZOLPIMIST.
ZOLPIMIST may not be right for you. Before starting ZOLPIMIST, tell your doctor about all of your health conditions, including if you:
- have a history of depression, mental illness, or suicidal thoughts
- have a history of drug or alcohol abuse or addiction
- have kidney or liver disease
- have a lung disease or breathing problems
- are pregnant, planning to become pregnant, or breastfeeding
Tell your doctor about all the medicines you take including prescription and nonprescription medicines, vitamins, and herbal supplements. Medicines can interact with each other, sometimes causing serious side effects. Do not take ZOLPIMIST with other medicines that can make you sleepy.
Know the medicines you take. Keep a list of your medicines with you to show your doctor and pharmacist each time you get a new medicine.
How should I take ZOLPIMIST?
- Take ZOLPIMIST exactly as prescribed. Do not take more ZOLPIMIST than prescribed for you.
- Take ZOLPIMIST right before you get into bed.
- Do not take ZOLPIMIST unless you are able to stay in bed a full night (7-8 hours) before you must be active again.
- For faster sleep onset, ZOLPIMIST should NOT be taken with or immediately after a meal.
- Call your doctor if your insomnia worsens or is not better within 7 to 10 days. This may mean that there is another condition causing your sleep problem.
- If you take too much ZOLPIMIST or overdose, call your doctor or poison control center right away, or get emergency treatment.
What are the possible side effects of ZOLPIMIST?
Serious side effects of ZOLPIMIST include:
- getting out of bed while not being fully awake and doing an activity that you do not know you are doing. (See “What is the most important information I should know about ZOLPIMIST?”)
- abnormal thoughts and behavior. Symptoms include more outgoing or aggressive behavior than normal, confusion, agitation, hallucinations, worsening of depression, and suicidal thoughts or actions.
- memory loss
- anxiety
- severe allergic reactions. Symptoms include swelling of the tongue or throat, trouble breathing, and nausea and vomiting. Get emergency medical help if you get these symptoms after taking ZOLPIMIST.
Call your doctor right away if you have any of the above side effects or any other side effects that worry you while using ZOLPIMIST.
The most common side effects of ZOLPIMIST are:
- drowsiness
- dizziness
- diarrhea
- “drugged feelings”
- You may still feel drowsy the next day after taking ZOLPIMIST. Do not drive or do other dangerous activities after taking ZOLPIMIST until you feel fully awake.
After you stop taking a sleep medicine, you may have symptoms for 1 or 2 days such as: tiredness, trouble sleeping, nausea, flushing, lightheadedness, uncontrolled crying, vomiting, stomach cramps, panic attack, nervousness, and stomach area pain.
These are not all the side effects of ZOLPIMIST. Ask your doctor or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
- Store ZOLPIMIST in an upright position at 59 °F to 86 °F (15 °C to 30 °C).
- Do not freeze.
- Avoid prolonged product exposure above 86 °F (30 °C).
- The child-resistant container should be thrown away when the 60 sprays have been used.
Keep ZOLPIMIST and all medicines out of reach of children.
General Information about ZOLPIMIST
- Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide.
- Do not use ZOLPIMIST for a condition for which it was not prescribed
- Do not share ZOLPIMIST with other people, even if you think they may have the same symptoms that you have. It may harm them, and it is against the law.
This Medication Guide summarizes the most important information about ZOLPIMIST. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about ZOLPIMIST that is written for healthcare professionals. For more information about ZOLPIMIST, call 1-855-298-8246.
What are the ingredients in ZOLPIMIST?
Active Ingredient: Zolpidem tartrate
Inactive Ingredients: artificial cherry flavor, benzoic acid, citric acid monohydrate, hydrochloric acid, neotame, propylene glycol, and purified water.
Rx Only
Revised August 2019
This Medication Guide has been approved by U.S. Food and Drug Administration.
Aytu BioScience, Inc.
Englewood, CO 80112
-
INSTRUCTIONS FOR USE
Patient Instructions for Use
ZOLPIMIST (zolpidem tartrate)
Oral Spray, Metered for Oral UseBe sure to carefully read, understand, and follow these instructions so that you use ZOLPIMIST the right way. Ask your healthcare provider or pharmacist if you have any questions about how to use ZOLPIMIST.
Priming:
Before you use ZOLPIMIST for the first time or if you have not used ZOLPIMIST for 14 days, you will need to prime the pump (Steps 1-6). Otherwise go directly to Step 7.
To prime the pump:
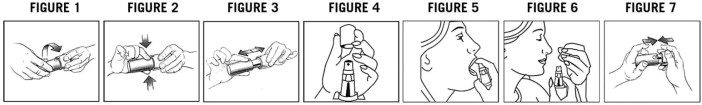
1. Line up the arrows on the child-resistant cap and base (see Figure 1).
2. Squeeze the cap at arrows (see Figure 2).
3. Pull the cap and base to separate (see Figure 3).
4. Remove the clear protective cap from the pump (see Figure 4).
5. Hold the container upright. Point the black spray opening in a safe direction away from your face and other people. Fully press down on the pump with your forefinger. Release the pump and let the pump return to the starting position.
6. Follow step 5 and press down on the pump 4 more times. You should see a fine spray. ZOLPIMIST is now ready to use. Now go directly to Step 11.
Taking a dose of ZOLPIMIST:
- If you are using ZOLPIMIST for the first time or you have not used ZOLPIMIST for 14 days, you will need to prime the pump (Steps 1-6). Otherwise, there is no need to prime the pump.
- Take ZOLPIMIST exactly as prescribed. Do not take more ZOLPIMIST than prescribed for you. Your healthcare provider will tell you whether to take 1 or 2 sprays of ZOLPIMIST.
- Take ZOLPIMIST right before you get into bed.
- Do not take ZOLPIMIST unless you are able to stay in bed a full night (7-8 hours) before you must be active again.
7. Line up the arrows on the child-resistant cap and base (see Figure 1).
8. Squeeze the cap at arrows (see Figure 2).
9. Pull the cap and base to separate (see Figure 3).
10. Remove the clear protective cap from the pump (see Figure 4).
11. Hold the container upright with the black spray opening pointed directly into your mouth. Fully press down on the pump to make sure that a full dose of ZOLPIMIST is sprayed directly into your open mouth over your tongue (see Figure 5).
12. Let the pump return to the starting position. If your healthcare provider prescribed only one spray of ZOLPIMIST (5 mg dose), go directly to Step 14.
13. If your healthcare provider prescribes a second spray of ZOLPIMIST (10 mg dose), repeat Step 11.
14. Put the clear protective cap back over the pump at the top of the child-resistant base after each use (see Figure 6).
15. Snap the child-resistant cap back onto the base and rotate the child-resistant cap and the child-resistant base so that the arrows are not lined up (see Figure 7).
16. The child-resistant container should be thrown away when the 60 sprays have been used.
There are no special requirements for cleaning and maintaining ZOLPIMIST. Professional assistance regarding questions about product performance or use can be obtained by calling 1-855-298-8246.
See Medication Guide section “How should I store ZOLPIMIST?” for instructions about how to store ZOLPIMIST.
Manufactured for
Aytu BioScience, Inc.
373 Inverness Parkway, Suite 206
Englewood, CO 80112by
Rechon Life Sciences AB
SE-216 10 Limhamn, Sweden -
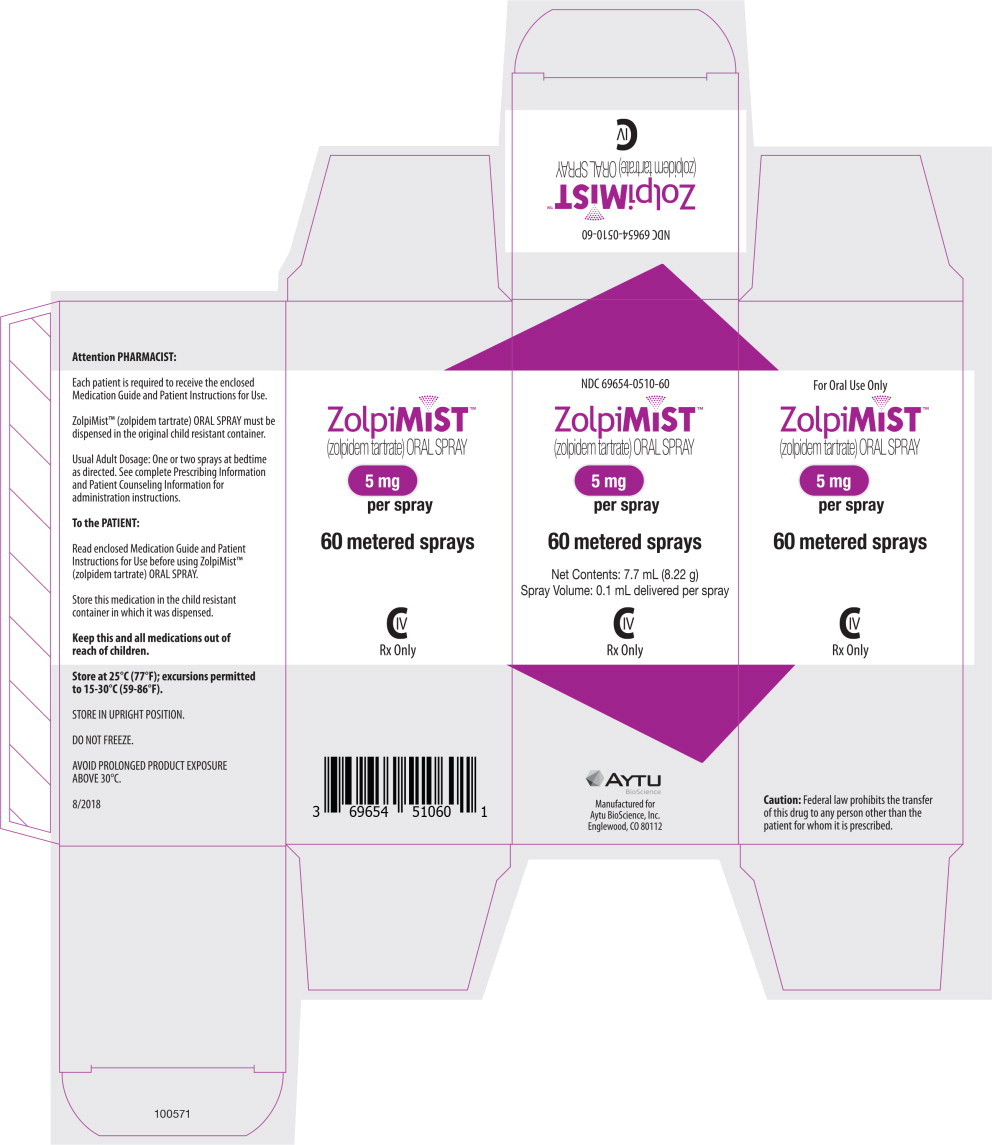
Principal Display Panel - Zolpimist 30 Carton Label
NDC: 69654-0510-30
ZolpiMIST™
(zolpidem tartrate) ORAL SPRAY
5 mg
per spray
30 metered sprays
Net Contents: 4.5 mL (4.8 g)
Spray Volume: 0.1 mL delivered per spray
CIV
Rx Only
-
Principal Display Panel - Zolpimist 60 Carton Label
NDC: 69654-0510-60
ZolpiMIST™
(zolpidem tartrate) ORAL SPRAY
5 mg
per spray
60 metered sprays
Net Contents: 7.7 mL (8.22 g)
Spray Volume: 0.1 mL delivered per spray
CIV
Rx Only
-
INGREDIENTS AND APPEARANCE
ZOLPIMIST
zolpidem tartrate spray, meteredProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69654-510 Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Zolpidem Tartrate (UNII: WY6W63843K) (Zolpidem - UNII:7K383OQI23) Zolpidem Tartrate 5 mg Inactive Ingredients Ingredient Name Strength cherry (UNII: BUC5I9595W) benzoic acid (UNII: 8SKN0B0MIM) citric acid monohydrate (UNII: 2968PHW8QP) hydrochloric acid (UNII: QTT17582CB) neotame (UNII: VJ597D52EX) propylene glycol (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color Score Shape Size Flavor CHERRY (CHERRY) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69654-510-30 1 in 1 CARTON 02/26/2019 1 30 in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 2 NDC: 69654-510-60 1 in 1 CARTON 02/26/2019 2 60 in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022196 02/26/2019 Labeler - Aytu BioScience, Inc. (079847712)
Trademark Results [ZOLPIMIST]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ZOLPIMIST 88016434 5684905 Live/Registered |
Aytu BioScience, Inc. 2018-06-26 |
 ZOLPIMIST 88013915 5689515 Live/Registered |
AYTU BIOSCIENCE, INC. 2018-06-25 |
 ZOLPIMIST 77176611 3958019 Dead/Cancelled |
AMHERST PHARMACEUTICALS 2007-05-09 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.