MEDERMA ADVANCED SCAR- allantoin gel
Mederma Advanced Scar by
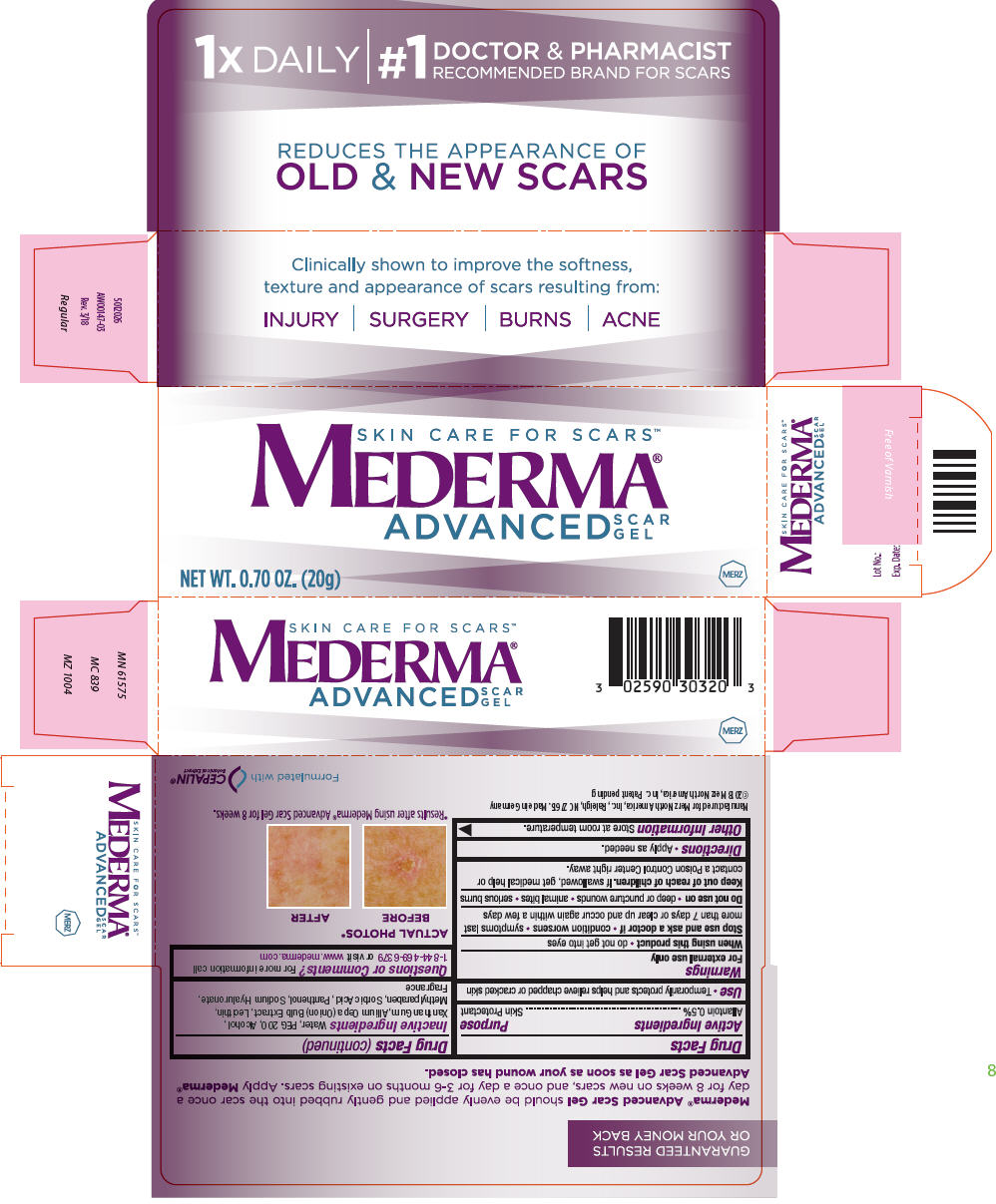
Drug Labeling and Warnings
Mederma Advanced Scar by is a Otc medication manufactured, distributed, or labeled by Merz Pharmaceuticals, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
- Use
- Warnings
- Directions
- Other information
- Inactive Ingredients
- Questions or Comments?
- PRINCIPAL DISPLAY PANEL - 20 g Tube Box
-
INGREDIENTS AND APPEARANCE
MEDERMA ADVANCED SCAR
allantoin gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0259-1105 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Allantoin (UNII: 344S277G0Z) (Allantoin - UNII:344S277G0Z) Allantoin 5 mg in 1 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Polyethylene Glycol 200 (UNII: R95B8J264J) Alcohol (UNII: 3K9958V90M) Xanthan Gum (UNII: TTV12P4NEE) Onion (UNII: 492225Q21H) Egg Phospholipids (UNII: 1Z74184RGV) Methylparaben (UNII: A2I8C7HI9T) Sorbic Acid (UNII: X045WJ989B) Panthenol (UNII: WV9CM0O67Z) Hyaluronate Sodium (UNII: YSE9PPT4TH) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0259-1105-20 1 in 1 BOX 11/01/2013 1 20 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC: 0259-1105-50 1 in 1 BOX 11/01/2013 2 50 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC: 0259-1105-01 1 g in 1 PACKET; Type 0: Not a Combination Product 11/01/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part347 11/01/2013 Labeler - Merz Pharmaceuticals, LLC (126209282)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.