TEMOZOLOMIDE by Areva Pharmaceuticals / NerPharMa SRL TEMOZOLOMIDE capsule
TEMOZOLOMIDE by
Drug Labeling and Warnings
TEMOZOLOMIDE by is a Prescription medication manufactured, distributed, or labeled by Areva Pharmaceuticals, NerPharMa SRL. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use TEMOZOLOMIDE CAPSULES safely and effectively. See full prescribing information for TEMOZOLOMIDE CAPSULES.
TEMOZOLOMIDE CAPSULES for oral use
Initial U.S. Approval: 1999RECENT MAJOR CHANGES
Warnings and Precautions (5.5) 02/2020
INDICATIONS AND USAGE
Temozolomide Capsules are an alkylating drug indicated for the treatment of adult patients with: (1)
- Newly diagnosed glioblastoma concomitantly with radiotherapy and then as maintenance treatment (1.1)
- Refractory anaplastic astrocytoma patients who have experienced disease progression on a drug regimen containing nitrosourea and procarbazine. (1.2)
DOSAGE AND ADMINISTRATION
- Newly Diagnosed Glioblastoma:
- 75 mg/m 2 once daily for 42 days concomitant with focal radiotherapy followed by initial maintenance dose of 150 mg/m 2 once daily for Days 1 to 5 of each 28-day cycle for 6 cycles. May increase maintenance dose to 200 mg/m 2 for cycles 2 – 6 based on toxicity. (2.1)
- Provide Pneumocystis Pneumonia (PCP) prophylaxis during concomitant phase and continue in patients who develop lymphopenia until resolution to grade 1 or less. (2.1)
- Refractory Anaplastic Astrocytoma: Initial dose of 150 mg/m 2 once daily on Days 1 to 5 of each 28-day cycle. (2.2)
DOSAGE FORMS AND STRENGTHS
- Capsules: 5 mg, 20 mg, 100 mg, 140 mg, 180 mg, and 250 mg (3)
CONTRAINDICATIONS
- History of hypersensitivity to temozolomide or any other ingredients in Temozolomide Capsules and dacarbazine. (4.1)
WARNINGS AND PRECAUTIONS
- Myelosuppression: Monitor absolute neutrophil count (ANC) and platelet count prior to each cycle and during treatment. Geriatric patients and women have a higher risk of developing myelosuppression. (5.1)
- Myelodysplastic Syndrome and Secondary Malignancies, including myeloid leukemia, have been observed. (5.2)
- Pneumocystis Pneumonia (PCP): Closely monitor all patients, particularly those receiving steroids, for the development of lymphopenia and PCP. (5.3)
- Hepatotoxicity: Fatal and severe hepatotoxicity have been reported. Perform liver tests at baseline, midway through the first cycle, prior to each subsequent cycle, and approximately 2 to 4 weeks after the last dose of temozolomide.
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. Advise male patients with pregnant partners or female partners of reproductive potential to use condoms. (5.5, 8.1, 8.3)
ADVERSE REACTIONS
- The most common adverse reactions (greater than or equal to 20% incidence) are: alopecia, fatigue, nausea, vomiting, headache, constipation, anorexia, and convulsions. (6.1)
- The most common Grade 3 to 4 hematologic laboratory abnormalities (greater than or equal to 10% incidence) in patients with anaplastic astrocytoma are: decreased lymphocytes, decreased platelets, decreased neutrophils, and decreased leukocytes. (6.1)
(6)
(6)
To report SUSPECTED ADVERSE REACTIONS, contact Areva at 1-855-853-4760 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. (6)
USE IN SPECIFIC POPULATIONS
See 17 for FDA-approved patient labeling.
Revised: 3/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1. INDICATIONS AND USAGE
1.1 Newly Diagnosed Glioblastoma
1.2 Refractory Anaplastic Astrocytoma
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage and Dosage Modifications for Newly Diagnosed Glioblastoma
2.2 Recommended Dosage and Dosage Modifications for Refractory Anaplastic Astrocytoma
2.3 Preparation and Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Myelosuppression
5.2 Myelodysplastic Syndrome and Secondary Malignancies
5.3 Pneumocystis Pneumonia
5.4 Hepatotoxicity
5.5 Embryo-Fetal Toxicity
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Newly Diagnosed Glioblastoma
14.2 Refractory Anaplastic Astrocytoma
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1. INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage and Dosage Modifications for Newly Diagnosed Glioblastoma
Administer Temozolomide Capsules once daily for 42 consecutive days during the concomitant phase with focal radiotherapy and then once daily on Days 1 to 5 of each 28-day cycle for 6 cycles during the maintenance phase.
Provide Pneumocystis pneumonia (PCP) prophylaxis during the concomitant phase and continue in patients who develop lymphocytopenia until resolution to grade 1 or less [see Warnings and Precautions (5.3)].
Concomitant Phase:
The recommended dosage of Temozolomide is 75 mg/m 2 once daily for 42 days (up to 49 days) concomitant with focal radiotherapy (60 Gy administered in 30 fractions). Focal radiotherapy includes the tumor bed or resection site with a 2- to 3-cm margin.
Obtain a complete blood count weekly. No dose reductions are recommended during the concomitant phase. The recommended dosage modifications during the concomitant phase are provided in Table 1.
TABLE 1: Temozolomide Dosage Modifications During Concomitant Phase
Adverse Reaction
Interruption
Discontinuation
Absolute Neutrophil Count
Withhold Temozolomide if ANC is greater than or equal to 0.5 x 10 9/L and less than 1.5 x 10 9/L.
Resume Temozolomide when ANC is greater than or equal to 1.5 x 10 9/L.
Discontinue Temozolomide if platelet count is less than 0.5 x 10 9/L.
Platelet Count
Withhold Temozolomide if platelet count is greater than or equal to 10 x 10 9/L and less than 100 x 10 9/L.
Resume Temozolomide when platelet count is greater than or equal to 100 x 10 9/L.
Discontinue Temozolomide if platelet count is less than 10 x 10 9/L.
Non-hematological Adverse Reaction (except for alopecia, nausea, vomiting)
Withhold Temozolomide if Grade 2 adverse reaction occurs.
Resume Temozolomide when resolution to Grade 1 or less.
Discontinue Temozolomide if Grade 3 or 4 adverse reaction occurs.
Maintenance Phase
Beginning 4 weeks after Concomitant Phase completion, administer Temozolomide once daily on Days 1 to 5 of each 28-day cycle for 6 cycles. The recommended dosage of Temozolomide is as follows:
- Cycle 1: 150 mg/m 2 per day
- Cycles 2 to 6: may increase to 200 mg/m 2 per day if the following conditions are met before starting cycle 2. If the dose was not escalated at the onset of Cycle 2, do not increase the dose for Cycles 3 to 6.
- Non-hematologic toxicity is Grade 2 or less (except for alopecia, nausea, vomiting)
- ANC is greater than or equal to 1.5 x 10 9/L and
- Platelet count is greater than or equal to 100 x 10 9/L.
Obtain a complete blood count on Day 22 and then weekly until the ANC is above 1.5 x 10 9/L and the platelet count is above 100 x 10 9/L. Do not start the next cycle until the ANC and platelet count exceed these levels.
The recommended dosage modifications during the maintenance phase are provided in Table 2. If Temozolomide is withheld, reduce the dose for the next cycle by 50 mg/m 2 per day. Permanently discontinue Temozolomide in patients who are unable to tolerate a dose of 100 mg/m 2 per day.
TABLE 2: Temozolomide Dosage Modifications During Maintenance Treatment
Toxicity
Interruption and Dose Reduction
Discontinuation
Absolute Neutrophil Count
Withhold Temozolomide if ANC less than 1 x 10 9/L.
When ANC is above 1.5 x 10 9/L, resume Temozolomide at reduced dose for the next cycle.
Unable to tolerate a dose of 100 mg/m 2 per day.
Platelet Count
Withhold Temozolomide if platelet less than 50 x 10 9/L.
When platelet count is above 100 x 10 9/L, resume Temozolomide at reduced dose for the next cycle.
Unable to tolerate a dose of 100 mg/m 2 per day.
Non-hematological Adverse Reaction (except for alopecia, nausea, vomiting)
Withhold Temozolomide if Grade 3 adverse reaction.
When resolved to Grade 1 or less, resume Temozolomide at reduced dose for the next cycle.
Recurrent Grade 3 after dose reduction.
Grade 4
Unable to tolerate a dose of 100 mg/m 2 per day.
2.2 Recommended Dosage and Dosage Modifications for Refractory Anaplastic Astrocytoma
The recommended initial dosage of Temozolomide is 150 mg/m 2 once daily on Days 1 to 5 of each 28-day cycle. Increase the Temozolomide dose to 200 mg/m 2 per day if the following conditions are met at the nadir and on Day 1 of the next cycle:
- ANC is greater than or equal to 1.5 x 10 9/L and
- Platelet count is greater than or equal to 100 x 10 9/L.
Continue Temozolomide until disease progression or unacceptable toxicity. In the clinical trial, treatment could be continued for a maximum of 2 years, but the optimum duration of therapy is not known.
Obtain a complete blood count on Day 22 and then weekly until the ANC is above 1.5 x 10 9/L and the platelet count is above 100 x 10 9/L. Do not start the next cycle until the ANC and platelet count exceed these levels.
If the ANC is less than 1 x 10 9/L or the platelet count is less than 50 x 10 9/L during any cycle, reduce the Temozolomide dose for the next cycle by 50 mg/m 2 per day. Permanently discontinue Temozolomide in patients who are unable to tolerate a dose of 100 mg/m 2 per day.
2.3 Preparation and Administration
Temozolomide is a cytotoxic drug. Follow applicable special handling and disposal procedures.
Temozolomide capsules
Administer Temozolomide consistently with respect to food (fasting vs. nonfasting) [see Clinical Pharmacology (12.3)]. To reduce nausea and vomiting, take Temozolomide on an empty stomach or at bedtime and consider antiemetic therapy prior to and/or following Temozolomide administration.
Swallow Temozolomide capsules whole. Do not open or chew capsules.
If capsules are accidentally opened or damaged, take precautions to avoid inhalation or contact with the skin or mucous membranes. In case of powder contact, the hands should be washed.
-
3 DOSAGE FORMS AND STRENGTHS
Capsules:
- 5 mg capsules have opaque white bodies with green caps. The capsule body is imprinted with the dosage strength.
- 20 mg capsules have opaque white bodies with yellow caps. The capsule body is imprinted with the dosage strength.
- 100 mg capsules have opaque white bodies with pink caps. The capsule body is imprinted with the dosage strength.
- 140 mg capsules have opaque white bodies with blue caps. The capsule body is imprinted with the dosage strength.
- 180 mg capsules have opaque white bodies with orange caps. The capsule body is imprinted with the dosage strength.
- 250 mg capsules have opaque white bodies with white caps. The capsule body is imprinted with the dosage strength.
-
4 CONTRAINDICATIONS
Temozolomide is contraindicated in patients with a history of hypersensitivity reactions to:
- temozolomide or any other ingredients in Temozolomide capsules; and
- decarbazine, since both temozolomide and decarbazine are metabolized to the same active metabolite 5-(3-methyltriazen-1-yl)-imidazole-4-carboxamide.
Reactions to Temozolomide have included anaphylaxis [see Adverse Reactions (6.2)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Myelosuppression
Myelosuppression, including pancytopenia, leukopenia and anemia, some with fatal outcomes, have occurred with Temozolomide [see Adverse Reactions (6.1, 6.2)]. Geriatric patients and women have been shown in clinical trials to have a higher risk of developing myelosuppression.
Prior to dosing, patients must have an ANC of 1.5 x 10 9/L or greater and a platelet count of 100 x 10 9/L or greater.
For the concomitant phase with radiotherapy, obtain a complete blood count prior to initiation of treatment and weekly during treatment [see Dosage and Administration (2.1)].
For the 28-day treatment cycles, obtain a complete blood count prior to treatment on Day 1 and on Day 22 of each cycle. Perform complete blood counts weekly until recovery if the ANC falls below 1.5 x 10 9/L and the platelet count falls below 100 x 10 9/L [see Dosage and Administration (2.1, 2.2)].
5.2 Myelodysplastic Syndrome and Secondary Malignancies
Cases of myelodysplastic syndrome and secondary malignancies, including myeloid leukemia, have been observed following Temozolomide administration.
5.3 Pneumocystis Pneumonia
Pneumocystis pneumonia (PCP) can occur in patients receiving Temozolomide. The risk of PCP is increased in patients receiving steroids or with longer treatment regimens.
For patients with newly diagnosed glioblastoma, provide PCP prophylaxis for all patients during the concomitant phase. Continue in patients who experience lymphopenia until resolution to grade 1 or less [see Dosage and Administration (2.1)].
Monitor all patients receiving Temozolomide for the development of lymphopenia and PCP.
5.4 Hepatotoxicity
Fatal and severe hepatotoxicity have been reported in patients receiving Temozolomide. Perform liver tests at baseline, midway through the first cycle, prior to each subsequent cycle, and approximately two to four weeks after the last dose of Temozolomide.
5.5 Embryo-Fetal Toxicity
Based on findings from animal studies and its mechanism of action, Temozolomide can cause fetal harm when administered to a pregnant woman. Adverse developmental outcomes have been reported in both pregnant patients and pregnant partners of male patients. Oral administration of Temozolomide to rats and rabbits during the period of organogenesis resulted in embryolethality and polymalformations at doses less than the maximum human dose based on body surface area.
Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with Temozolomide and for at least 6 months after the final dose. Because of potential risk of genotoxic effects on sperm, advise male patients with female partners of reproductive potential to use condoms during treatment with Temozolomide and for at least 3 months after the final dose. Advise male patients not to donate semen during treatment with Temozolomide and for at least 3 months after the final dose [see Use in Specific Populations (8.1, 8.3)].
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Myelosuppression [see Warnings and Precautions (5.1)].
- Myelodysplastic Syndrome and Secondary Malignancies [see Warnings and Precautions (5.2)].
- Pneumocystis Pneumonia [see Warnings and Precautions (5.3)].
- Hepatotoxicity [see Warnings and Precautions (5.4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Newly Diagnosed Glioblastoma
The safety of Temozolomide was evaluated in Study MK-7365-051 [see Clinical Studies (14.1)].
Forty-nine percent (49%) of patients treated with Temozolomide reported one or more severe or life-threatening reactions, most commonly fatigue (13%), convulsions (6%), headache (5%), and thrombocytopenia (5%).
The most common adverse reactions (greater than or equal to 20%) across the cumulative Temozolomide experience were alopecia, fatigue, nausea, and vomiting. Table 3 summarizes the adverse reactions in Newly Diagnosed Glioblastoma Trial. Overall, the pattern of reactions during the maintenance phase was consistent with the known safety profile of Temozolomide.
TABLE 3: Adverse Reactions (5% or Greater) in Patients Receiving Temozolomide in
Newly Diagnosed Glioblastoma Trial
Adverse Reactions
Concomitant Phase
Maintenance Phase
Radiation Therapy and Temozolomide
N=288*
Radiation Therapy Alone
N=285
Temozolomide
N=224
All Grades
(%)
Grades greater than or equal to 3 (%)
(%)
All Grades
(%)
Grades greater than or equal to 3 (%)
All Grades
(%)
Grades greater than or equal to
3 (%)
Skin and Subcutaneous Tissue
Alopecia
69
63
55
Rash
19
1
15
13
1
Dry Skin
2
2
5
<1
Pruritus
4
1
5
Erythema
5
5
1
General
Fatigue
54
7
49
5
61
9
Anorexia
19
1
9
<1
27
1
Headache
19
2
17
4
23
4
Weakness
3
2
3
1
7
2
Dizziness
4
1
4
5
Gastrointestinal System
Nausea
36
1
16
<1
49
1
Vomiting
20
<1
6
<1
29
2
Constipation
18
1
6
22
Diarrhea
6
3
10
1
Stomatitis
7
5
<1
9
1
Abdominal Pain
2
<1
1
5
<1
Eye
Vision Blurred
9
1
9
1
8
Injury
Radiation Injury NOS
7
4
<1
2
Central and Peripheral Nervous System
Convulsions
6
3
7
3
11
3
Memory Impairment
3
<1
4
<1
7
1
Confusion
4
1
4
2
5
2
Special Senses Other
Taste Perversion
6
2
5
Respiratory System
Coughing
5
1
1
8
<1
Dyspnea
4
2
3
1
5
<1
Psychiatric
Insomnia
5
3
<1
4
Immune System
Allergic Reaction
5
2
<1
3
Platelet, Bleeding and Clotting
Thrombocytopenia
4
3
1
8
4
Musculoskeletal System
Arthralgia
2
<1
1
6
*One patient who was randomized to radiation therapy only arm received radiation therapy and Temozolomide.
NOS=not otherwise specified.
Note: Grade 5 (fatal) adverse reactions are included in the Grade greater than or equal to 3 column.
When laboratory abnormalities and adverse reactions were combined, Grade 3 or Grade 4 neutrophil abnormalities including neutropenic reactions were observed in 8% of the patients, and Grade 3 or Grade 4 platelet abnormalities including thrombocytopenic reactions, were observed in 14% of the patients.
Refractory Anaplastic Astrocytoma
The safety of Temozolomide was evaluated in Study MK-7365-006 [see Clinical Studies (14.2)].
Myelosuppression (thrombocytopenia and neutropenia) was the dose-limiting adverse reaction. It usually occurred within the first few cycles of therapy and was not cumulative.
Myelosuppression occurred late in the treatment cycle and returned to normal, on average, within 14 days of nadir counts. The median nadirs occurred at 26 days for platelets (range: 21-40 days) and 28 days for neutrophils (range: 1-44 days). Only 14% (22/158) of patients had a neutrophil nadir and 20% (32/158) of patients had a platelet nadir, which may have delayed the start of the next cycle. Less than 10% of patients required hospitalization, blood transfusion, or discontinuation of therapy due to myelosuppression.
The most common adverse reactions (greater than or equal to 20%) were nausea, vomiting, headache, fatigue, constipation, and convulsions.
Tables 4 and 5 summarize the adverse reactions and hematological laboratory abnormalities in Refractory Anaplastic Astrocytoma Trial.
TABLE 4: Adverse Reactions (5% or Greater) in Patients Receiving Temozolomide in Refractory Anaplastic Astrocytoma Trial
Adverse Reactions
Temozolomide
N=158
All Reactions
(%)
Grade 3-4
(%)
Gastrointestinal System
Nausea
53
10
Vomiting
42
6
Constipation
33
1
Diarrhea
16
2
Abdominal Pain
9
1
Anorexia
9
1
General
Headache
41
6
Fatigue
34
4
Asthenia
13
6
Fever
13
2
Back Pain
8
3
Central and Peripheral Nervous System
Convulsions
23
5
Hemiparesis
18
6
Dizziness
12
1
Coordination abnormal
11
1
Amnesia
10
4
Insomnia
10
Paresthesia
9
1
Somnolence
9
3
Paresis
8
3
Urinary incontinence
8
2
Ataxia
8
2
Dysphasia
7
1
Convulsions local
6
Gait abnormal
6
1
Confusion
5
Cardiovascular
Edema peripheral
11
1
Resistance Mechanism
Infection viral
11
Endocrine
Adrenal hypercorticism
8
Respiratory System
Upper respiratory tract infection
8
Pharyngitis
8
Sinusitis
6
Coughing
5
Skin and Appendages
Rash
8
Pruritus
8
1
Urinary System
Urinary tract infection
8
Micturition increased frequency
6
Psychiatric Disorders
Anxiety
7
1
Depression
6
Reproductive Disorders
Breast pain, female
6
Metabolic
Weight increase
5
Musculoskeletal System
Myalgia
5
Vision
Diplopia
5
Vision abnormal*
5
*This item includes blurred vision; visual deficit; vision changes; and vision troubles.
TABLE 5: Grade 3 to 4 Adverse Hematologic Laboratory Abnormalities in Refractory Anaplastic Astrocytoma Trial
Temozolomide *†
Decreased lymphocytes
55%
Decreased platelets
19%
Decreased neutrophils
14%
Decreased leukocytes
11%
Decreased hemoglobin
4%
* Change from Grade 0 to 2 at baseline to Grade 3 or 4 during treatment
† Dominator range = 142, 158
Hematological Toxicities for Advanced Gliomas
In clinical trial experience with 110 to 111 females and 169 to 174 males (depending on measurements), females experienced higher rates of Grade 4 neutropenia (ANC less than 0.5 x 10 9/L) and thrombocytopenia (less than 20 x 10 9/L) than males in the first cycle of therapy (12% vs. 5% and 9% vs. 3%, respectively).
In the entire safety database for which hematologic data exist (N=932), 7% (4/61) and 9.5% (6/63) of patients over age 70 experienced Grade 4 neutropenia or thrombocytopenia in the first cycle, respectively. For patients less than or equal to age 70, 7% (62/871) and 5.5% (48/879) experienced Grade 4 neutropenia or thrombocytopenia in the first cycle, respectively. Pancytopenia, leukopenia, and anemia also occurred.
Adverse reactions with Temozolomide for injection
Adverse reactions that were reported in 35 patients who received Temozolomide for injection that were not reported in patients who received Temozolomide capsules were pain, irritation, pruritus, warmth, swelling, and erythema at infusion site; petechiae and hematoma.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Temozolomide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to the drug exposure.
Dermatologic: Toxic epidermal necrolysis and Stevens-Johnson syndrome
Immune System: Hypersensitivity reactions, including anaphylaxis. Erythema multiforme, which resolved after discontinuation of Temozolomide and, in some cases, recurred upon rechallenge.
Hematopoietic: Prolonged pancytopenia, which may result in aplastic anemia and fatal outcomes.
Hepatobiliary: Fatal and severe hepatotoxicity, elevation of liver enzymes, hyperbilirubinemia, cholestasis, and hepatitis.
Infections: Serious opportunistic infections, including some cases with fatal outcomes, can occur with bacterial, viral (primary and reactivated), fungal, and protozoan organisms.
Pulmonary: Interstitial pneumonitis, pneumonitis, alveolitis, and pulmonary fibrosis.
Endocrine: Diabetes insipidus
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on its mechanism of action [see Clinical Pharmacology (12.1)] and findings from animal studies, Temozolomide can cause fetal harm when administered to a pregnant woman. Available postmarketing reports describe cases of spontaneous abortions and congenital malformations, including polymalformations with central nervous system, facial, cardiac, skeletal, and genitourinary system anomalies with exposure to Temozolomide during pregnancy. These cases report similar adverse developmental outcomes to those observed in animal studies. Administration of Temozolomide to rats and rabbits during the period of organogenesis caused numerous external, internal, and skeletal malformations at doses less than the maximum human dose based on body surface area (see Data). Advise pregnant women of potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Five consecutive days of oral administration of Temozolomide at doses of 75 and 150 mg/m 2 (0.38 and 0.75 times the human dose of 200 mg/m 2) in rats and rabbits, respectively, during the period of organogenesis (Gestation Days 8-12) caused numerous malformations of the external and internal organs and skeleton in both species. In rabbits, Temozolomide at the 150 mg/m 2 dose (0.75 times the human dose of 200 mg/m 2) caused embryolethality as indicated by increased resorptions.
8.2 Lactation
There are no data on the presence of Temozolomide or its metabolites in human milk, the effects on a breastfed child, or the effects on milk production. Because of the potential for serious adverse reactions, including myelosuppression from Temozolomide in the breastfed children, advise women not to breastfeed during treatment with Temozolomide and for at least 1 week after the final dose.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to initiating Temozolomide [see Use in Specific Population (8.1)].
Contraception
Females
Temozolomide can cause embryo-fetal harm when administered to a pregnant woman [see Use in Specific Population (8.1)]. Advise females of reproductive potential to use effective contraception during treatment with Temozolomide and for at least 6 months after the last dose.
Males
Because of the potential for embryofetal toxicity and genotoxic effects on sperm cells, advise male patients with pregnant partners or female partners of reproductive potential to use condoms during treatment with Temozolomide and for at least 3 months after the final dose [see Use in Specific Population (8.1), Nonclinical Toxicology (13.1)]. Advise male patients not to donate semen during treatment with Temozolomide and or at least 3 months after the final dose.
Infertility
Temozolomide may impair male fertility [see Nonclinical Toxicology (13.1)]. Limited data from male patients show changes in sperm parameters during treatment with Temozolomide; however, no information is available on the duration or reversibility of these changes.
8.4 Pediatric Use
Safety and effectiveness of Temozolomide have not been established in pediatric patients. Safety and effectiveness of Temozolomide capsules were assessed, but not established, in 2 open-label studies in pediatric patients aged 3 to 18 years. In one study, 29 patients with recurrent brain stem glioma and 34 patients with recurrent high-grade astrocytoma were enrolled. In a second study conducted by the Children’s Oncology Group (COG), 122 patients were enrolled, including patients with medulloblastoma/PNET (29), high grade astrocytoma (23), low grade astrocytoma (22), brain stem glioma (16), ependymoma (14), other CNS tumors (9), and non-CNS tumors (9). The adverse reaction profile in pediatric patients was similar to adults.
8.5 Geriatric Use
In the Newly Diagnosed Glioblastoma trial, Study MK-7365-051, 15% of patients were 65 years and older. This study did not include sufficient numbers of patients aged 65 years and older to determine differences in effectiveness from younger patients. No overall differences in safety were observed between patients greater than or equal to 65 years and younger patients.
In the Refractory Anaplastic Astrocytoma trial, MK-7365-0006, 4% of patients were 70 years and older. This study did not include sufficient numbers of patients aged 70 years and older to determine differences in effectiveness from younger patients. Patients 70 years and older had a higher incidence of Grade 4 neutropenia (25%) and Grade 4 thrombocytopenia (20%) in the first cycle of therapy than patients less than 70 years of age [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
8.6 Renal Impairment
No dosage adjustment is recommended for patients with creatinine clearance (CLcr) of 36 to 130 mL/min/m 2[see Clinical Pharmacology (12.3)]. The recommended dose of Temozolomide has not been established for patients with severe renal impairment (CLcr less than 36 mL/min/m 2) or for patients with end-stage disease on dialysis.
8.7 Hepatic Impairment
No dosage adjustment is recommended for patients with mild to moderate hepatic impairment (Child Pugh class A and B) [see Clinical Pharmacology (12.3)]. The recommended dose of Temozolomide has not been established for patients with severe hepatic impairment (Child Pugh class C).
-
10 OVERDOSAGE
Dose-limiting toxicity was myelosuppression and was reported with any dose but is expected to be more severe at higher doses. An overdose of 2000 mg per day for 5 days was taken by one patient and the adverse reactions reported were pancytopenia, pyrexia, multi-organ failure, and death. There are reports of patients who have taken more than 5 days of treatment (up to 64 days), with adverse reactions reported including myelosuppression, which in some cases was severe and prolonged, and infections and resulted in death. In the event of an overdose, monitor complete blood count and provide supportive measures as necessary.
-
11 DESCRIPTION
Temozolomide is an alkylating drug. The chemical name of temozolomide is 3,4-dihydro-3methyl-4-oxoimidazo[5,1-d]- as-tetrazine-8-carboxamide. The structural formula is:
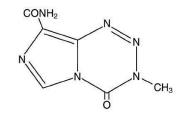
The material is a white to light tan/light pink powder with a molecular formula of C 6 H6 N6 O 2 and a molecular weight of 194.15. The molecule is stable at acidic pH (less than 5) and labile at pH greater than 7; hence Temozolomide can be administered orally. The prodrug, temozolomide, is rapidly hydrolyzed to the active 5-(3-methyltriazen-1-yl) imidazole-4-carboxamide (MTIC) at neutral and alkaline pH values, with hydrolysis taking place even faster at alkaline pH.
Temozolomide capsules:
Temozolomide capsules for oral use contains either 5 mg, 20 mg, 100 mg, 140 mg, 180 mg, or 250 mg of temozolomide.
The inactive ingredients for Temozolomide capsules are as follows:
Temozolomide capsules 5 mg: lactose anhydrous (132.8 mg), colloidal silicon dioxide (0.2 mg), sodium starch glycolate (7.5 mg), tartaric acid (1.5 mg), and stearic acid (3 mg).
Temozolomide capsules 20 mg: lactose anhydrous (182.2 mg), colloidal silicon dioxide (0.2 mg), sodium starch glycolate (11 mg), tartaric acid (2.2 mg), and stearic acid (4.4 mg).
Temozolomide capsules 100 mg: lactose anhydrous (175.7 mg), colloidal silicon dioxide (0.3 mg), sodium starch glycolate (15 mg), tartaric acid (3 mg), and stearic acid (6 mg).
Temozolomide capsules 140 mg: lactose anhydrous (246 mg), colloidal silicon dioxide (0.4 mg), sodium starch glycolate (21 mg), tartaric acid (4.2 mg), and stearic acid (8.4 mg).
Temozolomide capsules 180 mg: lactose anhydrous (316.3 mg), colloidal silicon dioxide (0.5 mg), sodium starch glycolate (27 mg), tartaric acid (5.2 mg), and stearic acid (10.8 mg).
Temozolomide capsules 250 mg: lactose anhydrous (154.3 mg), colloidal silicon dioxide (0.7 mg), sodium starch glycolate (22.5 mg), tartaric acid (9 mg), and stearic acid (13.5 mg).
The body of the capsules is made of gelatin, and is opaque white. The cap is also made of gelatin, and the colors vary based on the dosage strength. The capsule body is imprinted with pharmaceutical branding ink, which contains shellac, propylene glycol, strong ammonia solution, potassium hydroxide, and black iron oxide.
Temozolomide capsules 5 mg: The green cap contains gelatin, titanium dioxide, iron oxide yellow and FD&C Blue #2.
Temozolomide capsules 20 mg: The yellow cap contains gelatin, titanium dioxide, and iron oxide yellow.
Temozolomide capsules 100 mg: The pink cap contains gelatin, titanium dioxide and iron oxide red.
Temozolomide capsules 140 mg: The blue cap contains gelatin, titanium dioxide, and FD&C Blue #2.
Temozolomide capsules 180 mg: The orange cap contains gelatin, iron oxide red and titanium dioxide.
Temozolomide capsules 250 mg: The white cap contains gelatin and titanium dioxide.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Temozolomide is not directly active but undergoes rapid nonenzymatic conversion at physiologic pH to the reactive compound 5-(3-methyltriazen-1-yl)-imidazole-4-carboxamide (MTIC). The cytotoxicity of MTIC is thought to be primarily due to alkylation of DNA. Alkylation (methylation) occurs mainly at the O 6 and N 7 positions of guanine.
12.3 Pharmacokinetics
Following a single oral dose of 150 mg/m 2, the mean C max value for Temozolomide was 7.5 mcg/mL and for MTIC was 282 ng/mL.
The mean AUC value for Temozolomide was 23.4 mcghr/mL and for MTIC was 864 nghr/mL.
Following a single 90-minute intravenous infusion of 150 mg/m 2, the mean C max value for Temozolomide was 7.3 mcg/mL and for MTIC was 276 ng/mL. The mean AUC value for Temozolomide was 24.6 mcghr/mL and for MTIC was 891 nghr/mL.
Temozolomide exhibits linear kinetics over the therapeutic dosing range of 75 mg/m 2/day to 250 mg/m 2/day.
Absorption
The median T max is 1 hour.
Effect of Food
The mean C max and AUC decreased by 32% and 9%, respectively, and median T max increased by 2-fold (from 1-2.25 hours) when Temozolomide capsules were administered after a modified high-fat breakfast (587 calories of 1 fried egg, 2 strips of bacon, 2 slices of toast, 2 pats of butter, and 8 oz whole milk).
Distribution
Temozolomide has a mean apparent volume of distribution of 0.4 L/kg (%CV=13%). The mean percent bound of drug-related total radioactivity is 15%.
Elimination
Clearance of temozolomide is about 5.5 L/hr/m 2 and the mean elimination half-life is 1.8 hours.
Metabolism
Temozolomide is spontaneously hydrolyzed at physiologic pH to the active species, MTIC and to temozolomide acid metabolite. MTIC is further hydrolyzed to 5-amino-imidazole-4-carboxamide (AIC), which is known to be an intermediate in purine and nucleic acid biosynthesis, and to methylhydrazine, which is believed to be the active alkylating species. Cytochrome P450 enzymes play only a minor role in the metabolism of temozolomide and MTIC. Relative to the AUC of temozolomide, the exposure to MTIC and AIC is 2.4% and 23%, respectively.
Excretion
About 38% of the administered temozolomide total radioactive dose is recovered over 7 days: 38% in urine and 0.8% in feces. The majority of the recovery of radioactivity in urine is unchanged temozolomide (6%), AIC (12%), temozolomide acid metabolite (2.3%), and unidentified polar metabolite(s) (17%).
Specific Populations
No clinically meaningful differences in the pharmacokinetics of temozolomide were observed based on age (range: 19-78 years), gender, smoking status (smoker vs. non-smoker), creatinine clearance (CLcr) of 36 to 130 mL/min/m 2, or mild to moderate hepatic impairment (Child Pugh class A and B). The pharmacokinetics of temozolomide has not been studies in patients with CLcr < 36 mL/min/m 2, end-stage renal disease on dialysis, or severe hepatic impairment (Child Pugh class C).
Drug Interaction Studies
Effect of Other Drugs on Temozolomide Pharmacokinetics
In a multiple-dose study, administration of Temozolomide capsules with ranitidine did not change the C max or AUC values for temozolomide or MTIC.
A population analysis indicated that administration of valproic acid decreases the clearance of temozolomide by about 5%.
A population analysis did not demonstrate any influence of coadministered dexamethasone, prochlorperazine, phenytoin, carbamazepine, ondansetron, histamine-2-receptor antagonists, or phenobarbital on the clearance of orally administered temozolomide.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, mutagenesis, Impairment of Fertility
Temozolomide is carcinogenic in rats at doses less than the maximum recommended human dose. Temozolomide induced mammary carcinomas in both males and females at doses 0.13 to 0.63 times the maximum human dose (25-125 mg/m 2) when administered orally on 5 consecutive days every 28 days for 6 cycles. Temozolomide also induced fibrosarcomas of the heart, eye, seminal vesicles, salivary glands, abdominal cavity, uterus, and prostate, carcinomas of the seminal vesicles, schwannomas of the heart, optic nerve, and harderian gland, and adenomas of the skin, lung, pituitary, and thyroid at doses 0.5 times the maximum daily dose. Mammary tumors were also induced following 3 cycles of temozolomide at the maximum recommended daily dose.
Temozolomide is a mutagen and a clastogen. In a reverse bacterial mutagenesis assay (Ames assay), temozolomide increased revertant frequency in the absence and presence of metabolic activation. Temozolomide was clastogenic in human lymphocytes in the presence and absence of metabolic activation.
Temozolomide impairs male fertility. Temozolomide caused syncytial cells/immature sperm formation at doses of 50 and 125 mg/m 2 (0.25 and 0.63 times the human dose of 200 mg/m 2) in rats and dogs, respectively, and testicular atrophy in dogs at 125 mg/m 2.
13.2 Animal Toxicology and/or Pharmacology
Toxicology studies in rats and dogs identified a low incidence of hemorrhage, degeneration, and necrosis of the retina at temozolomide doses equal to or greater than 125 mg/m 2 (0.63 times the human dose of 200 mg/m 2) . These changes were most commonly seen at doses where mortality was observed.
-
14 CLINICAL STUDIES
14.1 Newly Diagnosed Glioblastoma
The efficacy of Temozolomide was evaluated in Study MK-7365-051, a randomized (1:1), multicenter, open-label trial. Eligible patients were required to have newly diagnosed glioblastoma. Patients were randomized to receive either radiation therapy alone or concomitant Temozolomide 75 mg/m 2 once daily starting the first day of radiation therapy and continuing the last day of radiation therapy for 42 days (with maximum of 49 days), followed by Temozolomide 150 mg/m 2 or 200 mg/m 2 once daily on Days 1 to 5 of each 28-day cycle, starting 4 weeks after the end of radiation therapy and continuing for 6 cycles. In both arms, focal radiation therapy was delivered as 60 Gy/30 fractions and included radiation to the tumor bed or resection site with a 2 to 3-cm margin. PCP prophylaxis was required during the concomitant phase, regardless of lymphocyte count and continued until recovery of lymphocyte count to grade 1 or less. The major efficacy outcome measure was overall survival.
A total of 573 patients were randomized, 287 to Temozolomide and radiation therapy and 286 to radiation therapy alone. At the time of disease progression, Temozolomide was administered as salvage therapy in 161 patients of the 282 (57%) in the radiation therapy alone arm, and 62 patients of the 277 (22%) in the Temozolomide and radiation therapy arm.
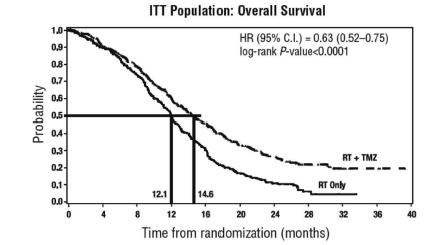
The addition of concomitant and maintenance Temozolomide to radiation therapy in the treatment of patients with newly diagnosed glioblastoma showed a statistically significant improvement in overall survival compared to radiotherapy alone ( Figure 1). The hazard ratio (HR) for overall survival was 0.63 (95% CI: 0.52, 0.75) with a log-rank P less than 0.0001 in favor of the Temozolomide arm. The median survival was increased by 2.5 months in the Temozolomide arm.
FIGURE 1: Kaplan-Meier Curves for Overall Survival (ITT Population) in Newly Diagnosed Glioblastoma Trial
14.2 Refractory Anaplastic Astrocytoma
The efficacy of Temozolomide was evaluated in Study MK-7365-006, a single-arm, multicenter trial. Eligible patients had anaplastic astrocytoma at first relapse and a baseline Karnofsky performance status (KPS) of 70 or greater. Patients had previously received radiation therapy and may also have previously received a nitrosourea with or without other chemotherapy. Fifty-four patients had disease progression on prior therapy with both a nitrosourea and procarbazine, and their malignancy was considered refractory to chemotherapy (refractory anaplastic astrocytoma population). Temozolomide capsules were given on Days 1 to 5 of each 28-day cycle at a starting dose of 150 mg/m 2/day. If ANC was greater than or equal to 1.5 x 10 9/L and platelet count was greater than or equal to 100 x 10 9/L at the nadir and on Day 1 of the next cycle, the Temozolomide dose was increased to 200 mg/m 2/day. The major efficacy outcome measure was progression-free survival at 6 months and the additional efficacy outcome measures were overall survival and overall response rate.
In the refractory anaplastic astrocytoma population (n=54), the median age was 42 years (range: 19 to 76); 65% were male; and 72% had a KPS of greater than 80. Sixty-three percent of patients had surgery other than a biopsy at the time of initial diagnosis. Of those patients undergoing resection, 73% underwent a subtotal resection and 27% underwent a gross total resection. Eighteen percent of patients had surgery at the time of first relapse. The median time from initial diagnosis to first relapse was 13.8 months (range: 4.2 months to 6.3 years).
In the refractory anaplastic astrocytoma population, the overall response rate (CR+PR) was 22% (12 of 54 patients) and the complete response rate was 9% (5 of 54 patients). The median duration of all responses was 50 weeks (range: 16 to 114 weeks) and the median duration of complete responses was 64 weeks (range: 52 to 114 weeks). In this population, progression-free survival at 6 months was 45% (95% CI: 31%, 58%) and progression-free survival at 12 months was 29% (95% CI: 16%, 42%). Median progression-free survival was 4.4 months. Overall survival at 6 months was 74% (95% CI: 62%, 86%) and 12-month overall survival was 65% (95% CI: 52%, 78%). Median overall survival was 15.9 months.
- 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Temozolomide is a cytotoxic drug. Follow applicable special handling and disposal procedures.
Temozolomide capsules
Temozolomide capsules are supplied in amber glass bottles with child-resistant caps containing the following capsule strengths:
Temozolomide capsules 5 mg: have opaque white bodies with green caps. The capsule body is imprinted with the dosage strength.
They are supplied as follows:
5-count – NDC: 59923-703-05
14-count – NDC: 59923-704-14
Temozolomide capsules 20 mg: have opaque white bodies with yellow caps. The capsule body is imprinted with the dosage strength.
They are supplied as follows:
5-count – NDC: 59923-705-05
14-count – NDC: 59923-706-14
Temozolomide capsules 100 mg: have opaque white bodies with pink caps. The capsule body is imprinted with the dosage strength.
They are supplied as follows:
5-count – NDC: 59923-707-05
14-count – NDC: 59923-708-14
Temozolomide capsules 140 mg: have opaque white bodies with blue caps. The capsule body is imprinted with the dosage strength.
They are supplied as follows:
5-count – NDC: 59923-709-05
14-count – NDC: 59923-710-14
Temozolomide capsules 180 mg: have opaque white bodies with orange caps. The capsule body is imprinted with the dosage strength.
They are supplied as follows:
5-count – NDC: 59923-711-05
14-count – NDC: 59923-712-14
Temozolomide capsules 250 mg: have opaque white bodies with white caps. The capsule body is imprinted with the dosage strength.
They are supplied as follows:
5-count – NDC: 59923-713-05
Store Temozolomide capsules at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-Approved Patient Labeling (Patient Information).
Myelosuppression
Inform patients that Temozolomide can cause low blood cell counts and the need for frequent monitoring of blood cell counts. Advise patients to contact their healthcare provider immediately for bleeding, fever, or other signs of infections [see Warnings and Precautions (5.1)].
Myelodysplastic Syndrome and Secondary Malignancies
Advise patients of the increased risk of myelodysplastic syndrome and secondary malignancies [see Warnings and Precautions (5.2)].
Pneumocystis Pneumonia
Advise patients of the increased risk of Pneumocystis pneumonia and to contact their healthcare provider immediately for new or worsening pulmonary symptoms. Inform patients that prophylaxis for Pneumocystis pneumonia may be needed [see Dosage and Administration (2.1), Warnings and Precautions (5.3)].
Hepatotoxicity
Advise patients of the increased risk of hepatotoxicity and to contact their healthcare provider immediately for signs or symptoms of hepatotoxicity [see Warnings and Precautions (5.4)].
Administration Instructions
Advise patients to not open capsules. If capsules are accidentally opened or damaged, advise patients to take rigorous precautions with capsule contents to avoid inhalation or contact with the skin or mucous membranes. In case of powder contact, the hands should be washed [see Dosage and Administration (2.3)].
Embryo-Fetal Toxicity
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females to inform their healthcare provider of a known or suspected pregnancy [see Warnings and Precautions (5.5), Use in Specific Population (8.1)].
Advise females of reproductive potential to use effective contraception during treatment with Temozolomide and for at least 6 months after the last dose [see Use in Specific Population (8.3)].
Advise male patients with pregnant partners or female partners of reproductive potential to use condoms during treatment with Temozolomide and for at least 3 months after the final dose [see Use in Specific Population (8.3), Nonclinical Toxicology (13.1)].
Advise male patients not to donate semen during treatment with Temozolomide and for at least 3 months after the final dose [see Use in Specific Population (8.3), Nonclinical Toxicology (13.1)].
Lactation
Advise women not to breastfeed during treatment with Temozolomide and for at least 1 week after the final dose [see Use in Specific Population (8.2)].
Infertility
Advise males of reproductive potential that Temozolomide may impair fertility [see Use in Specific Population (8.3), Nonclinical Toxicology (13.1)].
Distributed by:
Areva Pharmaceuticals Inc. Georgetown, IN 47122
Made in Italy
Revised: 02/2020
-
Patient Information
Patient Information
Temozolomide capsules (TEM-oh-ZOE-loe-mide)
What is the most important information I should know about Temozolomide capsules?
Temozolomide capsules may cause birth defects
Females and female partners of male patients who take Temozolomide capsules:
- Avoid becoming pregnant while taking Temozolomide capsules.
- Females who can become pregnant should use an effective form of birth control (contraception) during treatment and for at least 6 months after your last dose of Temozolomide capsules. Your doctor should to do a pregnancy test to make sure that you are not pregnant before you start taking Temozolomide capsules.
- Tell your doctor right away if you become pregnant or think you are pregnant during treatment with Temozolomide capsules.
Males taking Temozolomide capsules and have a female partner who is pregnant or who can become pregnant:
- Use a condom for birth control (contraception) during treatment and for at least 3 months after taking your final dose of Temozolomide capsules.
- Do not donate semen during treatment and for at least 3 months after your final dose of Temozolomide capsules.
See the section "What are the possible side effects of Temozolomide capsules?" for more information about side effects.
What are Temozolomide capsules?
Temozolomide capsules are a prescription medicine used to treat adults with certain brain cancer tumors.
It is not known if Temozolomide capsules are safe and effective in children.
Who should not take Temozolomide capsules?
Do not take Temozolomide capsules if you:
- have had an allergic reaction to temozolomide or any of the other ingredients in Temozolomide capsules. See the end of the leaflet for a list of ingredients in Temozolomide capsules. Symptoms of an allergic reaction with Temozolomide capsules may include: a red itchy rash, or a severe allergic reaction, such as trouble breathing, swelling of the face, throat, or tongue, or severe skin reaction. If you are not sure, ask your doctor.
- have had an allergic reaction to dacarbazine (DTIC), another cancer medicine.
What should I tell my doctor before taking Temozolomide capsules?
Tell your doctor about all your medical conditions, including if you:
- have kidney problems
- have liver problems
- are pregnant or plan to become pregnant. See “What is the most important information I should know about Temozolomide capsules?”
- are breast-feeding or plan to breastfeed. It is not known if Temozolomide capsules passes into your breast milk. Do not breastfeed during treatment and for at least 1 week after your last dose of Temozolomide capsules.
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Especially tell your doctor if you take a medicine that contains valproic acid.
Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist when you get a new medicine.
How should I take Temozolomide capsules?
Temozolomide capsules may be taken by mouth as a capsule.
Take Temozolomide capsules exactly as prescribed by your doctor.
There are 2 common dosing schedules for taking Temozolomide capsules depending on the type of brain cancer tumor that you have.
- People with certain brain cancer tumors take or receive Temozolomide capsules:
- 1 time each day for 42 days in a row (possibly 49 days depending on side effects) along with receiving radiation treatment. This is one cycle of treatment.
- After this, your doctor may prescribe 6 more cycles of Temozolomide capsules as “maintenance” treatment. For each of these cycles, you take or receive Temozolomide capsules 1 time each day for 5 days in a row and then you stop taking it for the next 23 days. This is a 28-day maintenance treatment cycle.
- People with certain other brain cancer tumors take or receive Temozolomide capsules:
- 1 time each day for 5 days in a row only, and then you stop taking it for the next 23 days. This is one cycle of treatment (28 days).
- Your doctor will watch your progress on Temozolomide and decide how long you should take it. You might take Temozolomide capsules until your tumor gets worse or for possibly up to 2 years.
- If your doctor prescribes a treatment regimen that is different from the information in this leaflet, make sure you follow the instructions given to you by your doctor.
- Your doctor may change your dose of Temozolomide capsules, or tell you to stop Temozolomide capsules for a short period of time or permanently if you have certain side effects.
- Your doctor will decide how many treatment cycles of Temozolomide capsules that you will receive, depending on how you respond to and tolerate this treatment.
Temozolomide capsules:
- Take Temozolomide capsules exactly as your doctor tells you to.
- Temozolomide capsules contain a white capsule body with a color cap and the colors vary based on the dosage strength. Your doctor may prescribe more than 1 strength of Temozolomide capsules for you, so it is important that you understand how to take your medicine the right way. Be sure that you understand exactly how many capsules you need to take on each day of your treatment, and what strengths to take. This maybe different whenever you start a new cycle.
- Do not take more Temozolomide capsules than prescribed.
- Talk to your doctor or pharmacist before taking your dose if you are not sure how much Temozolomide capsules to take. This will help to prevent taking too much Temozolomide capsules and decrease your chances of getting serious side effects.
- Take each day's dose of Temozolomide capsules at one time, with a full glass of water.
- Swallow Temozolomide capsules whole. Do not chew, open, or split the capsules.
- Take Temozolomide capsules at the same time each day.
- Take Temozolomide capsules the same way each time, either with food or without food.
- If Temozolomide capsules are accidentally opened or damaged, be careful not to breathe in (inhale) the powder from the capsules or get the powder on your skin or mucous membranes (for example, in your nose or mouth). If contact with any of these areas happens, flush the area with water.
- To help reduce nausea and vomiting, try to take Temozolomide capsules on an empty stomach or bedtime. Your doctor may prescribe medicine to prevent or treat nausea, or other medicines to lessen side effects with Temozolomide capsules.
- See your doctor regularly to check your progress. Your doctor will check you for side effects.
- If you take more Temozolomide capsules than prescribed, call your doctor or get emergency medical help right away.
What are the possible side effects of Temozolomide capsules?
Temozolomide capsules can cause serious side effects, including:
- See “What is the most important information I should know about Temozolomide capsules?”
- Decreased blood cell counts. Temozolomide capsules can affect your bone marrow and cause you to have decreased blood cell counts. Decreased white blood cell count, red blood cell count and platelet count are common with Temozolomide capsules but it can also be severe and lead to death.
- Your doctor will do blood tests regularly to check your blood cell counts before you start and during treatment with Temozolomide capsules.
- Your doctor may need to change the dose of Temozolomide capsules, or when you get it depending on your blood cell counts.
- People who are age 70 or older and women have a higher risk for developing decreased blood cell counts during treatment with Temozolomide capsules.
- Secondary cancers. Blood problems such as myelodysplastic syndrome (MDS) and new cancers (secondary cancers), including a certain kind of leukemia, can happen in people who take Temozolomide capsules. Your doctor will monitor you for this.
- Pneumocystis pneumonia (PCP). PCP is an infection that people can get when their immune system is weak. Temozolomide capsules decreases white blood cells, which makes your immune system weaker and can increase your risk of getting PCP.
- People who are taking steroid medicines or who stay on Temozolomide capsules for a longer period of time may have an increased risk of getting PCP infection.
- Anyone who takes Temozolomide capsules will be watched carefully by their doctor for low blood cell counts and this infection.
- Tell your doctor if you have any of the following signs and symptoms of PCP infection: shortness of breath, or fever, chills, dry cough.
- Liver problems. Liver problems can happen with Temozolomide capsules and can sometimes be severe and lead to death. Your doctor will do blood tests to check your liver function before you start taking Temozolomide capsules, during treatment, and about 2 to 4 weeks after your last dose of Temozolomide capsules.
Common side effects with Temozolomide capsules include:
- hair loss
- unable to move (paralysis) on one
- feeling tired
side of the body
- nausea and vomiting
- weakness
- headache
- fever
- constipation
- dizziness
- loss of appetite
- coordination problems
- convulsions
- viral infection
- rash
- memory loss
- diarrhea
- sleep problems
Temozolomide capsules can affect fertility in males and may affect your ability to father a child. Talk with your doctor if fertility is a concern for you.
Tell your doctor about any side effect that bothers you or that does not go away.
These are not all the possible side effects with Temozolomide capsules. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Temozolomide capsules?
- Store Temozolomide capsules at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep Temozolomide capsules out of the reach of children.
General information about the safe and effective use of Temozolomide capsules.
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information leaflet. Do not use Temozolomide capsules for a condition for which it was not prescribed. Do not give Temozolomide capsules to other people, even if they have the same symptoms that you have. It may harm them.
This leaflet summarizes the most important information about Temozolomide capsules. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about Temozolomide capsules that is written for health professionals.
For more information, contact Areva at 1-855-853-4760.
What are the ingredients in Temozolomide capsules?
Temozolomide capsules:
Active ingredient: temozolomide.
Inactive ingredients: lactose anhydrous, colloidal silicon dioxide, sodium starch glycolate, tartaric acid, stearic acid.
The body of the capsules is made of gelatin, and is opaque white. The cap is also made of gelatin, and the colors vary based on the dosage strength. The capsule body is imprinted with pharmaceutical branding ink, which contains shellac, propylene glycol, strong ammonia solution, potassium hydroxide, and black iron oxide.
Temozolomide capsules 5 mg: The green cap contains gelatin, titanium dioxide, iron oxide yellow and FD&C Blue #2.
Temozolomide capsules 20 mg: The yellow cap contains gelatin, titanium dioxide, and iron oxide yellow.
Temozolomide capsules 100 mg: The pink cap contains gelatin, titanium dioxide and iron oxide red.
Temozolomide capsules 140 mg: The blue cap contains gelatin, titanium dioxide, and FD&C Blue #2.
Temozolomide capsules 180 mg: The orange cap contains gelatin, iron oxide red and titanium dioxide.
Temozolomide capsules 250 mg: The white cap contains gelatin and titanium dioxide.
Trademarks are the property of their respective owners.
Distributed by:
Areva Pharmaceuticals Inc. Georgetown, IN 47122
Made in Italy
Revised: 02/2020
-
PRINCIPAL DISPLAY PANEL
Principal Display Panel - 5 mg per capsule, 5 capsules
NDC: 59923-703-05
Temozolomide Capsules
5 mg per capsule
For Oral Administration
Rx Only 5 Capsules
Principal Display Panel - 5 mg per capsule, 14 capsules
NDC: 59923-704-14
Temozolomide Capsules
5 mg per capsule
For Oral Administration
Rx Only 14 Capsules
Principal Display Panel - 20 mg per capsule, 5 capsules
NDC: 59923-705-05
Temozolomide Capsules
20 mg per capsule
For Oral Administration
Rx Only 5 Capsules
Principal Display Panel - 20 mg per capsule, 14 capsules
NDC: 59923-706-14
Temozolomide Capsules
20 mg per capsule
For Oral Administration
Rx Only 14 Capsules
Principal Display Panel - 100 mg per capsule, 5 capsules
NDC: 59923-707-05
Temozolomide Capsules
100 mg per capsule
For Oral Administration
Rx Only 5 Capsules
Principal Display Panel - 100 mg per capsule, 14 capsules
NDC: 59923-708-14
Temozolomide Capsules
100 mg per capsule
For Oral Administration
Rx Only 14 Capsules
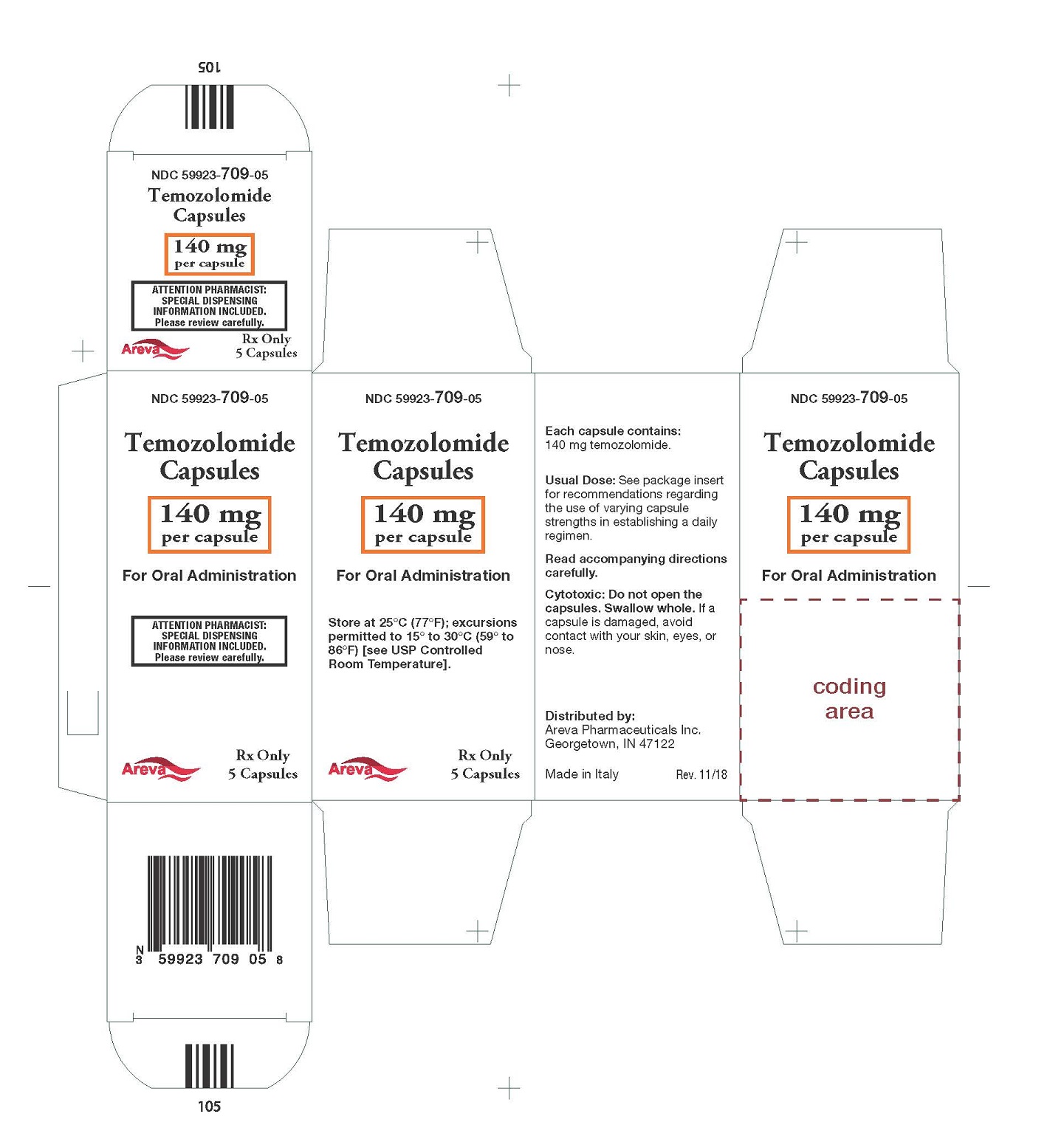
Principal Display Panel - 140 mg per capsule, 5 capsules
NDC: 59923-709-05
Temozolomide Capsules
140 mg per capsule
For Oral Administration
Rx Only 5 Capsules
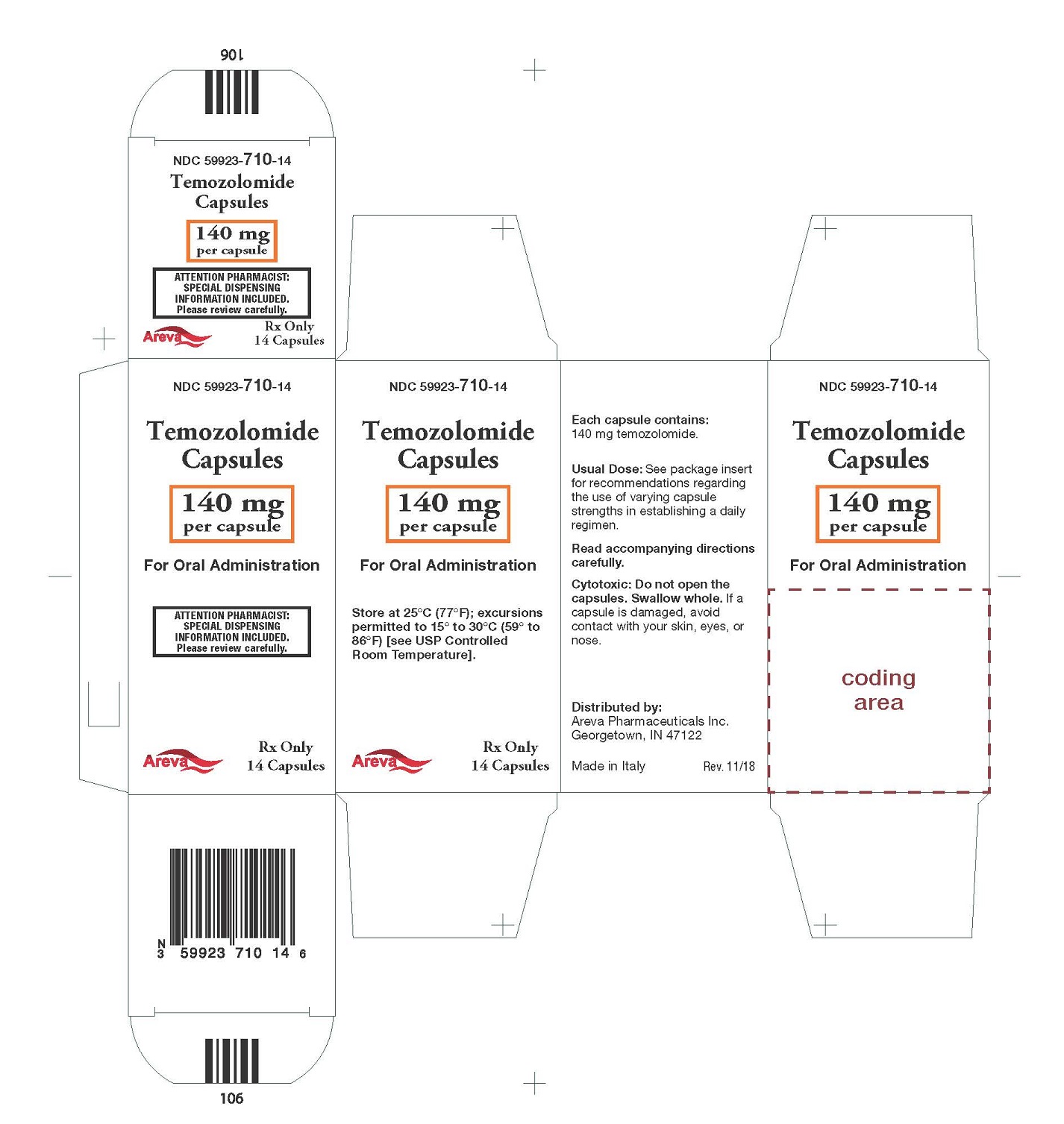
Principal Display Panel - 140 mg per capsule, 14 capsules
NDC: 59923-710-14
Temozolomide Capsules
140 mg per capsule
For Oral Administration
Rx Only 14 Capsules
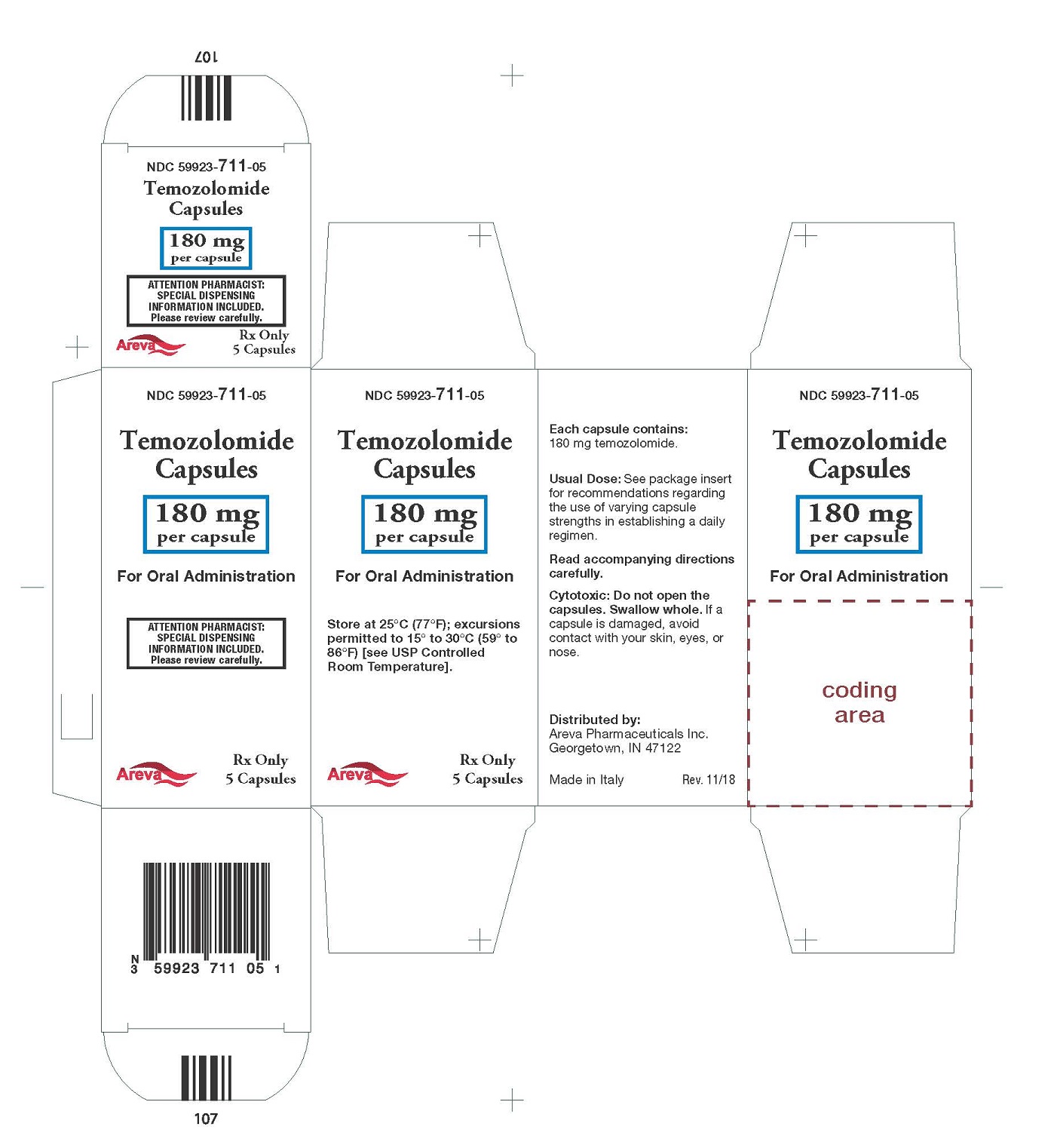
Principal Display Panel - 180 mg per capsule, 5 capsules
NDC: 59923-711-05
Temozolomide Capsules
180 mg per capsule
For Oral Administration
Rx Only 5 Capsules
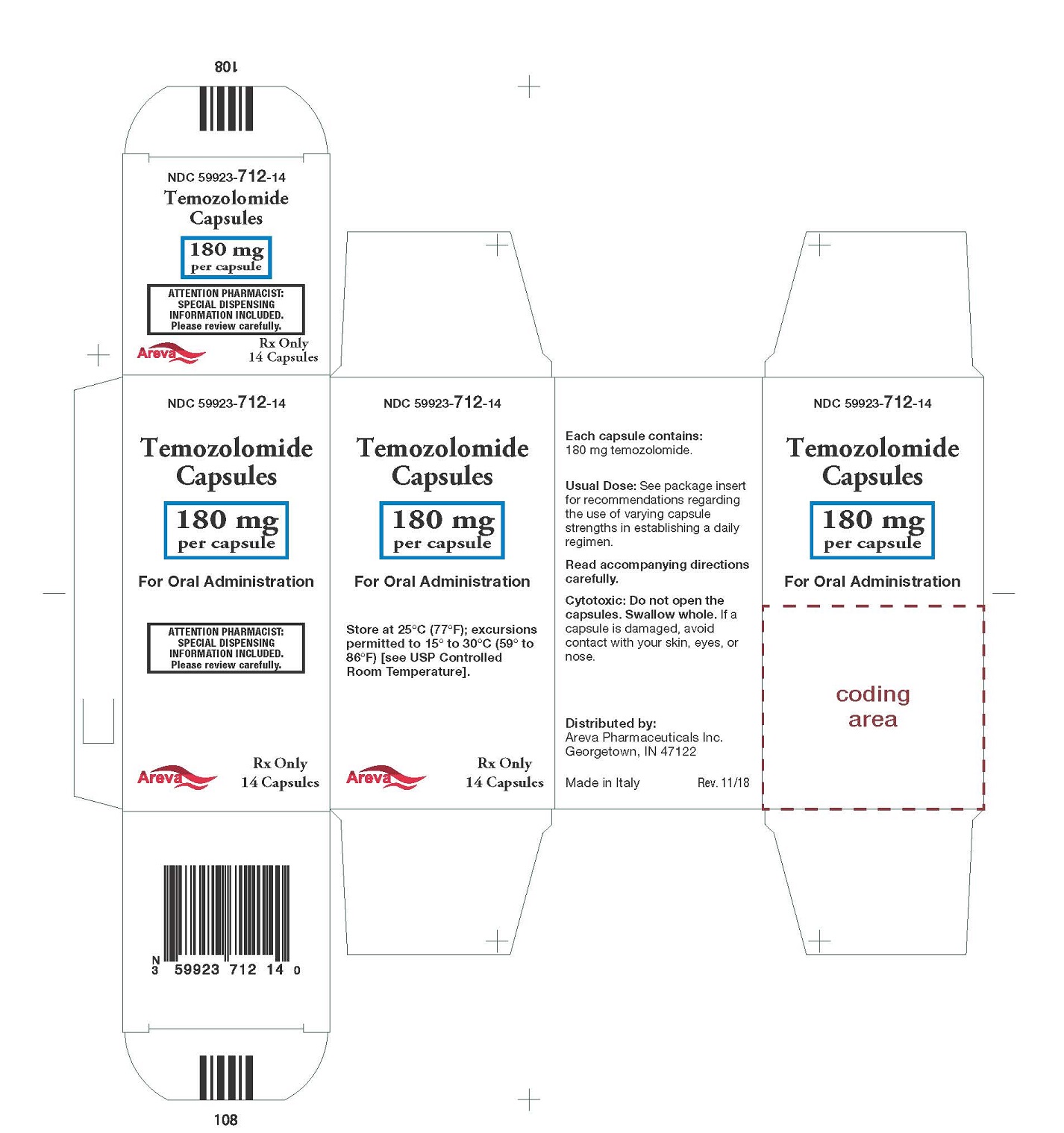
Principal Display Panel - 180 mg per capsule, 14 capsules
NDC: 59923-712-14
Temozolomide Capsules
180 mg per capsule
For Oral Administration
Rx Only 14 Capsules
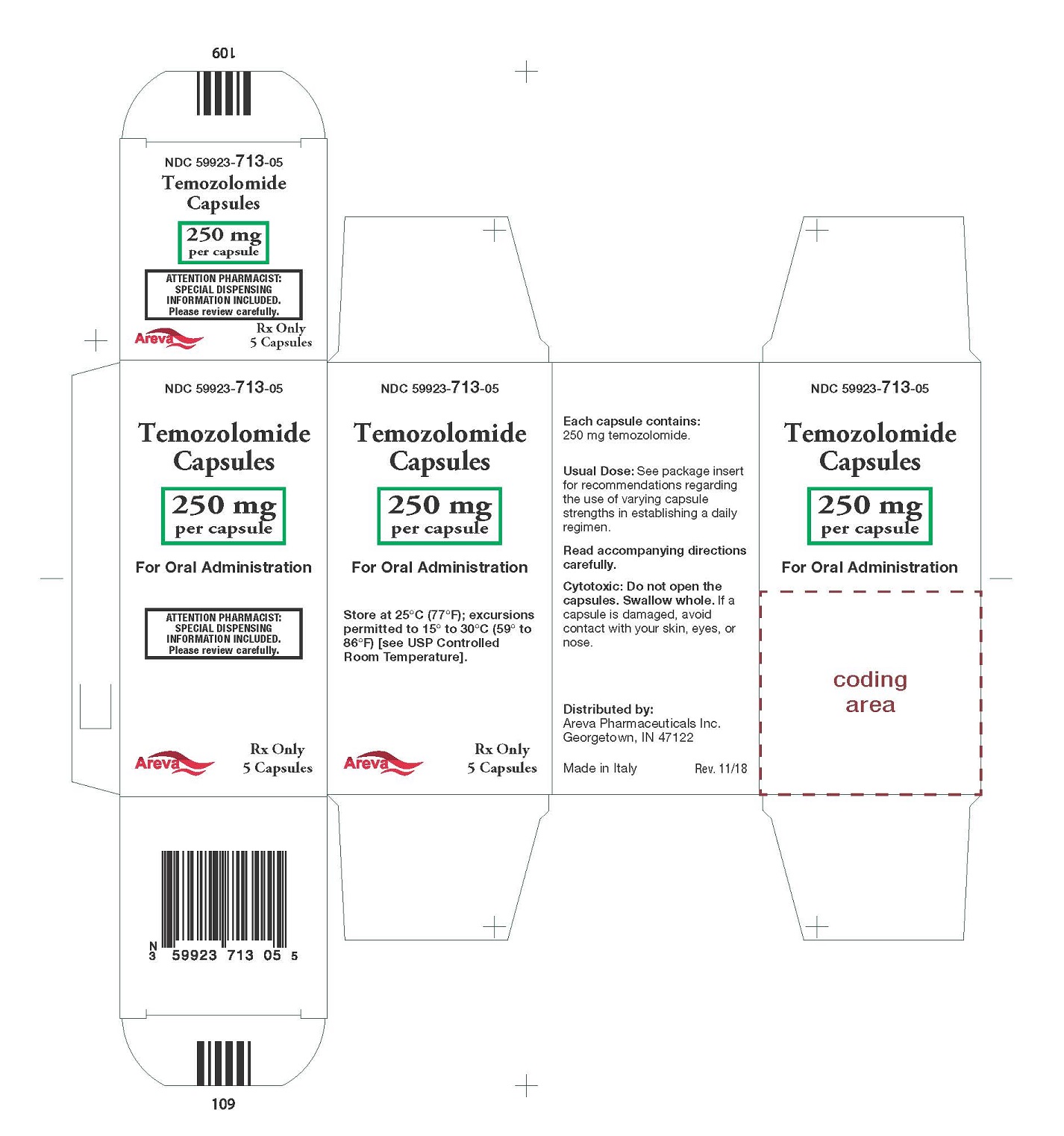
Principal Display Panel - 250 mg per capsule, 5 capsules
NDC: 59923-713-05
Temozolomide Capsules
250 mg per capsule
For Oral Administration
Rx Only 5 Capsules
-
INGREDIENTS AND APPEARANCE
TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-712 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 180 mg Inactive Ingredients Ingredient Name Strength STEARIC ACID (UNII: 4ELV7Z65AP) GELATIN (UNII: 2G86QN327L) SHELLAC (UNII: 46N107B71O) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) AMMONIA (UNII: 5138Q19F1X) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) TARTARIC ACID (UNII: W4888I119H) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color white, orange Score no score Shape CAPSULE Size 22mm Flavor Imprint Code 180 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-712-14 1 in 1 CARTON 01/25/2019 1 14 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-713 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 250 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) TARTARIC ACID (UNII: W4888I119H) STEARIC ACID (UNII: 4ELV7Z65AP) WATER (UNII: 059QF0KO0R) AMMONIA (UNII: 5138Q19F1X) GELATIN (UNII: 2G86QN327L) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) SHELLAC (UNII: 46N107B71O) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Product Characteristics Color white Score no score Shape CAPSULE Size 22mm Flavor Imprint Code 250 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-713-05 1 in 1 CARTON 01/25/2019 1 5 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-706 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 20 mg Inactive Ingredients Ingredient Name Strength TARTARIC ACID (UNII: W4888I119H) SHELLAC (UNII: 46N107B71O) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) AMMONIA (UNII: 5138Q19F1X) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) STEARIC ACID (UNII: 4ELV7Z65AP) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) GELATIN (UNII: 2G86QN327L) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Product Characteristics Color white, yellow Score no score Shape CAPSULE Size 18mm Flavor Imprint Code 20 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-706-14 1 in 1 CARTON 01/25/2019 1 14 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-704 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 5 mg Inactive Ingredients Ingredient Name Strength STEARIC ACID (UNII: 4ELV7Z65AP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FERROSOFERRIC OXIDE (UNII: XM0M87F357) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TARTARIC ACID (UNII: W4888I119H) SHELLAC (UNII: 46N107B71O) WATER (UNII: 059QF0KO0R) AMMONIA (UNII: 5138Q19F1X) GELATIN (UNII: 2G86QN327L) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color green, white Score no score Shape CAPSULE Size 16mm Flavor Imprint Code 5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-704-14 1 in 1 CARTON 01/25/2019 1 14 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-703 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 5 mg Inactive Ingredients Ingredient Name Strength FERROSOFERRIC OXIDE (UNII: XM0M87F357) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) AMMONIA (UNII: 5138Q19F1X) STEARIC ACID (UNII: 4ELV7Z65AP) SHELLAC (UNII: 46N107B71O) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) TARTARIC ACID (UNII: W4888I119H) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) GELATIN (UNII: 2G86QN327L) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color green, white Score no score Shape CAPSULE Size 16mm Flavor Imprint Code 5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-703-05 1 in 1 CARTON 01/25/2019 1 5 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-707 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 100 mg Inactive Ingredients Ingredient Name Strength SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) SHELLAC (UNII: 46N107B71O) WATER (UNII: 059QF0KO0R) AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) TARTARIC ACID (UNII: W4888I119H) STEARIC ACID (UNII: 4ELV7Z65AP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) FERRIC OXIDE RED (UNII: 1K09F3G675) GELATIN (UNII: 2G86QN327L) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color pink, white Score no score Shape CAPSULE Size 19mm Flavor Imprint Code 100 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-707-05 1 in 1 CARTON 01/25/2019 1 5 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-711 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 180 mg Inactive Ingredients Ingredient Name Strength SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) TARTARIC ACID (UNII: W4888I119H) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) GELATIN (UNII: 2G86QN327L) SHELLAC (UNII: 46N107B71O) WATER (UNII: 059QF0KO0R) AMMONIA (UNII: 5138Q19F1X) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) Product Characteristics Color white, orange Score no score Shape CAPSULE Size 22mm Flavor Imprint Code 180 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-711-05 1 in 1 CARTON 01/25/2019 1 5 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-705 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 20 mg Inactive Ingredients Ingredient Name Strength SHELLAC (UNII: 46N107B71O) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TARTARIC ACID (UNII: W4888I119H) AMMONIA (UNII: 5138Q19F1X) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) WATER (UNII: 059QF0KO0R) GELATIN (UNII: 2G86QN327L) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color yellow, white Score no score Shape CAPSULE Size 18mm Flavor Imprint Code 20 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-705-05 1 in 1 CARTON 01/25/2019 1 5 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-708 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 100 mg Inactive Ingredients Ingredient Name Strength POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) FERRIC OXIDE RED (UNII: 1K09F3G675) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) TARTARIC ACID (UNII: W4888I119H) SHELLAC (UNII: 46N107B71O) GELATIN (UNII: 2G86QN327L) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) AMMONIA (UNII: 5138Q19F1X) Product Characteristics Color pink, white Score no score Shape CAPSULE Size 19mm Flavor Imprint Code 100 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-708-14 1 in 1 CARTON 01/25/2019 1 14 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-710 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 140 mg Inactive Ingredients Ingredient Name Strength STEARIC ACID (UNII: 4ELV7Z65AP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) GELATIN (UNII: 2G86QN327L) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) TARTARIC ACID (UNII: W4888I119H) SHELLAC (UNII: 46N107B71O) AMMONIA (UNII: 5138Q19F1X) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) WATER (UNII: 059QF0KO0R) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) FERROSOFERRIC OXIDE (UNII: XM0M87F357) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color blue, white Score no score Shape CAPSULE Size 22mm Flavor Imprint Code 140 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-710-14 1 in 1 CARTON 01/25/2019 1 14 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 TEMOZOLOMIDE
temozolomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59923-709 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TEMOZOLOMIDE (UNII: YF1K15M17Y) (TEMOZOLOMIDE - UNII:YF1K15M17Y) TEMOZOLOMIDE 140 mg Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) AMMONIA (UNII: 5138Q19F1X) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) GELATIN (UNII: 2G86QN327L) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FERROSOFERRIC OXIDE (UNII: XM0M87F357) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TARTARIC ACID (UNII: W4888I119H) STEARIC ACID (UNII: 4ELV7Z65AP) SHELLAC (UNII: 46N107B71O) Product Characteristics Color white, blue Score no score Shape CAPSULE Size 22mm Flavor Imprint Code 140 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59923-709-05 1 in 1 CARTON 01/25/2019 1 5 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204639 01/25/2019 Labeler - Areva Pharmaceuticals (833189835) Establishment Name Address ID/FEI Business Operations NerPharMa SRL 338839192 manufacture(59923-703, 59923-704, 59923-705, 59923-706, 59923-707, 59923-708, 59923-709, 59923-710, 59923-711, 59923-712, 59923-713)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.