Benzocaine 20% by PAI Holdings, LLC dba PAI Pharma BENZOCAINE 20% solution
Benzocaine 20% by
Drug Labeling and Warnings
Benzocaine 20% by is a Otc medication manufactured, distributed, or labeled by PAI Holdings, LLC dba PAI Pharma. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- Active Ingredient
- Purpose
- Uses
-
Warnings
METHEMOGLOBINEMIA WARNING: Use of this product may cause methemoglobinemia, a rare but serious condition that must be treated promptly because it reduces the amount of oxygen carried in blood. This can occur even if you have used this product before. Stop use and seek immediate medical attention if you or a child in your care develops:
- pale, gray, or blue colored skin (cyanosis)
- headache
- rapid heart rate
- shortness of breath
- dizziness or lightheadedness
- fatigue or lack of energy
Allergy alert: Do not use if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine or other "caine" anesthetics.
Sore throat warning: if sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, swelling, nausea, or vomiting, consult a doctor promptly. If sore mouth symptoms do not improve in 7 days, or if irritation, pain, or redness persists or worsens, see your dentist or doctor promptly.
When using this product
- avoid contact with eyes
- do not exceed recommended dosage
- do not use for more than 7 days unless directed by a doctor or dentist
-
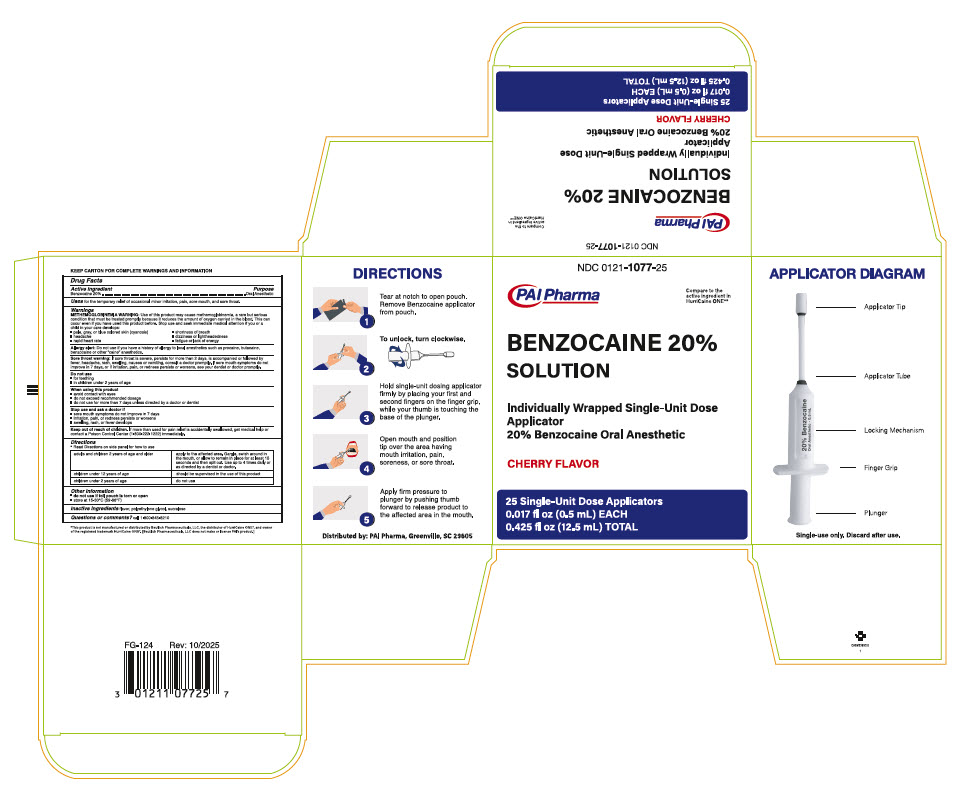
Directions
- Read Directions on side panel for how to use
adults and children 2 years of age and older apply to the affected area, Gargle, swish around in the mouth, or allow to remain in place for at least 15 seconds and then spit out. Use up to 4 times daily or as directed by a dentist or doctor. children under 12 years of age should be supervised in the use of the product children under 2 years of age do not use - Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel - pouch
NDC 0121-1077-01
BENZOCAINE 20% SOLUTION
Individually Wrapped Single-Unit Dose Applicator
20% Benzocaine Oral AnestheticCHERRY FLAVOR
Store at 15° - 30°C (59 - 86°F)
Warnings
METHEMOGLOBINEMIA WARNING: Use of this product may cause methemoglobinemia
a rare but serious condition that must be treated promptly because it reduces the amount of
oxygen carried in the blood. This can occur even if you have used this product before. stop use and
seek immediate medical attention if you or your child in your care develops:
■ pale, gray, or blue colored skin (cyanosis) ■ headache ■ rapid heart rate ■ shortness of breath
■ dizziness or lightheadedness ■ fatigue or lack of energy
Allergy alert: Do not use this product if you have a history of allergy to local anesthetics such as
procaine, butacaine, benzocaine, or other "caine" anesthetics.Please refer to Drug Facts for complete product information.
1 Single-Unit Dose Applicator 0.017 fl oz (0.5 mL)

- Principal Display Panel - carton
-
INGREDIENTS AND APPEARANCE
BENZOCAINE 20%
benzocaine 20% solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0121-1077 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 200 mg in 1 mL Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0121-1077-25 25 in 1 CARTON 03/20/2026 1 NDC: 0121-1077-01 1 in 1 POUCH 1 0.5 mL in 1 APPLICATOR; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M022 03/20/2026 Labeler - PAI Holdings, LLC dba PAI Pharma (044940096)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.