FSA Store Inc. Mucus-DM Drug Facts
caring mill mucus dm by
Drug Labeling and Warnings
caring mill mucus dm by is a Otc medication manufactured, distributed, or labeled by FSA Store Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CARING MILL MUCUS DM- dextromethorphan hbr, guaifenesin tablet, extended release
FSA Store Inc.
----------
FSA Store Inc. Mucus-DM Drug Facts
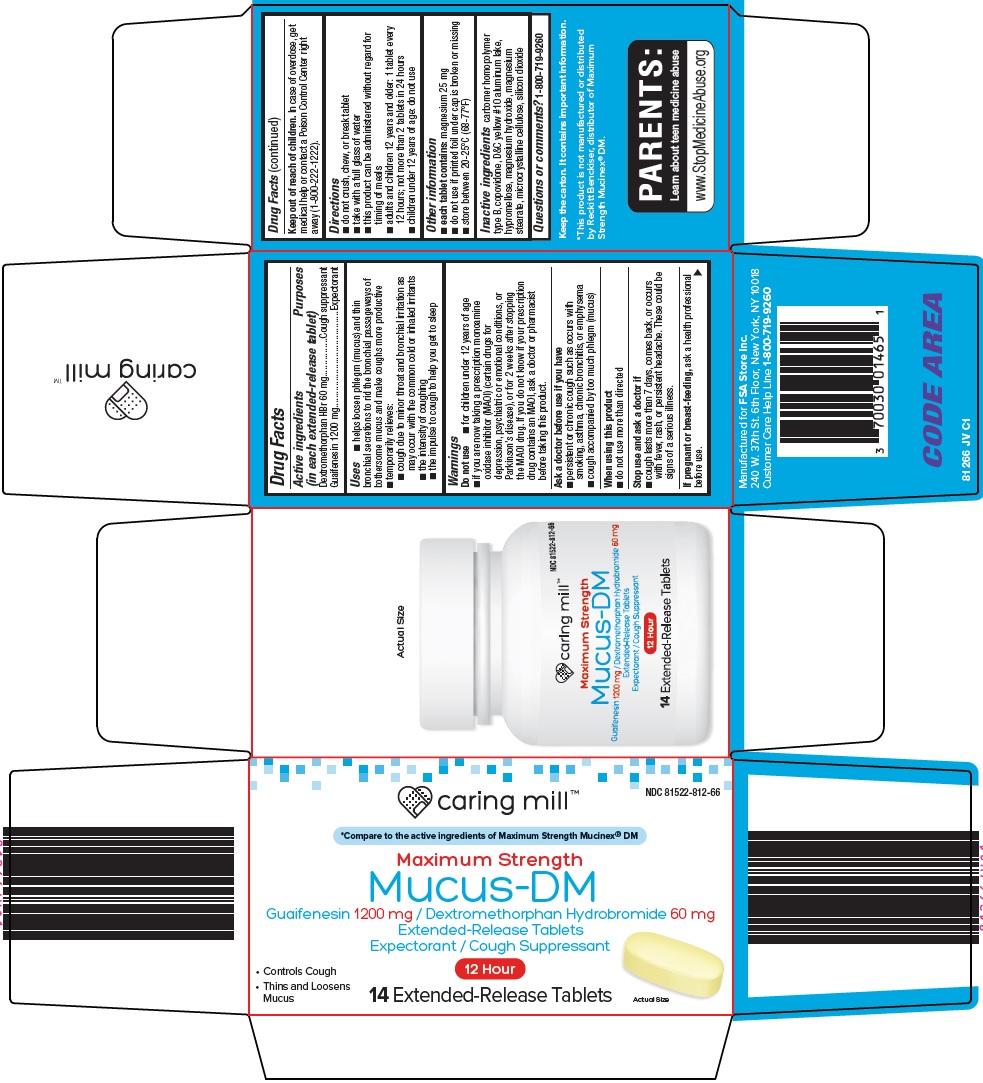
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- temporarily relieves:
- cough due to minor throat and bronchial irritation as may occur with the common cold or inhaled irritants
- the intensity of coughing
- the impulse to cough to help you get to sleep
Warnings
Do not use
- for children under 12 years of age
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- cough accompanied by too much phlegm (mucus)
Directions
- do not crush, chew, or break tablet
- take with a full glass of water
- this product can be administered without regard for timing of meals
- adults and children 12 years and older: 1 tablet every 12 hours; not more than 2 tablets in 24 hours
- children under 12 years of age: do not use
Other information
- each tablet contains: magnesium 25 mg
- do not use if printed foil under cap is broken or missing
- store between 20-25°C (68-77°F)
Inactive ingredients
carbomer homopolymer type B, copovidone, D&C yellow #10 aluminum lake, hypromellose, magnesium hydroxide, magnesium stearate, microcrystalline cellulose, silicon dioxide
Package/Label Principal Display Panel
Compare to the active ingredients of Maximum Strength Mucinex® DM
Maximum Strength
Mucus-DM
Guaifenesin 1200 mg / Dextromethorphan Hydrobromide 60 mg
Extended-Release Tablets
Expectorant / Cough Suppressant
Controls Cough
Thins and Loosens Mucus
12 Hour
14 Extended-Release Tablets
Actual Size
| CARING MILL MUCUS DM
dextromethorphan hbr, guaifenesin tablet, extended release |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - FSA Store Inc. (049283340) |
Revised: 1/2026
Document Id: 65af13b3-e623-4404-b710-13889cdd989a
Set id: 814bc1e4-85a0-4aa7-baa9-b9636fa38473
Version: 3
Effective Time: 20260120
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.