ETODOLAC capsule ETODOLAC tablet
ETODOLAC by
Drug Labeling and Warnings
ETODOLAC by is a Prescription medication manufactured, distributed, or labeled by STAT Rx USA LLC, PSS World Medical Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
Cardiovascular Risk
- NSAIDs may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction (MI), and stroke, which can be fatal. This risk may increase with duration of use. Patients with cardiovascular disease or risk factors for cardiovascular disease may be at greater risk. (See WARNINGS).
- Etodolac capsules USP, 200 mg and 300 mg, and Etodolac tablets USP, 400 mg and 500 mg are contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery (See WARNINGS).
Gastrointestinal Risk
- NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal (GI) events. (See WARNINGS).
-
DESCRIPTION
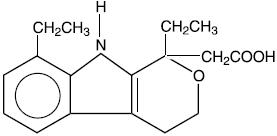
Etodolac capsules and tablets, USP are members of the pyranocarboxylic acid group of nonsteroidal anti-inflammatory drugs (NSAIDs). Each tablet and capsule contains etodolac for oral administration. Etodolac is a racemic mixture of [+]S and [-]R-enantiomers. Etodolac is a white crystalline compound, insoluble in water but soluble in alcohols, chloroform, dimethyl sulfoxide, and aqueous polyethylene glycol.
The chemical name is (±) 1,8-diethyl-1,3,4,9-tetrahydropyrano-[3,4-b]indole-1-acetic acid. The molecular weight of the base is 287.37. It has a pKa of 4.65 and an n-octanol: water partition coefficient of 11.4 at pH 7.4. The molecular formula for etodolac is C17H21NO3, and it has the following structural formula:
Each Capsule, for oral administration, contains 200 or 300 mg of Etodolac. In addition, each capsule contains the following inactive ingredients: Ammonium Hydroxide USP, Black Iron Oxide USP, Colloidal Silicone Dioxide NF, Erythrosine (200 mg only), Ethyl Alcohol USP, Gelatin, Isopropyl Alcohol USP, Lactose Monohydrate NF, Magnesium Stearate NF, Microcrystalline Cellulose NF, N-Butyl Alcohol USP, Povidone USP, Propylene Glycol USP, Purified Water USP, Shellac, Titanium Dioxide.
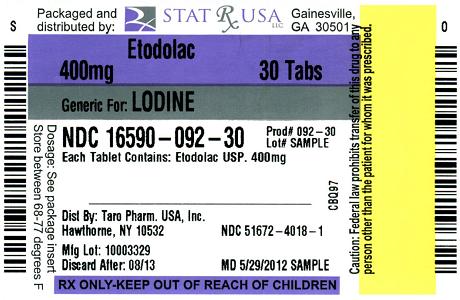
Each Tablet, for oral administration, contains 400 mg or 500 mg of Etodolac. In addition, each tablet contains the following inactive ingredients: Hydroxypropyl Methylcellulose USP, Lactose Monohydrate NF, Magnesium Stearate, Microcrystalline Cellulose NF, Polyethylene Glycol, Povidone USP, Sodium Starch Glycolate NF and Titanium Dioxide. Also, each 400 mg tablet contains Iron Oxide Red and Iron Oxide Yellow. Each 500 mg tablet contains D&C Yellow #10 Aluminum Lake, FD&C Blue #1 Aluminum Lake, and FD&C Red #40 Aluminum Lake.
-
CLINICAL PHARMACOLOGY
Pharmacodynamics
Etodolac is a nonsteroidal anti-inflammatory drug (NSAID) that exhibits anti-inflammatory, analgesic, and antipyretic activities in animal models. The mechanism of action of etodolac, like that of other NSAIDs, is not completely understood, but may be related to the prostaglandin synthetase inhibition.
Etodolac is a racemic mixture of [-]R- and [+]S-etodolac. As with other NSAIDs, it has been demonstrated in animals that the [+]S-form is biologically active. Both enantiomers are stable and there is no [-]R to [+] S conversion in vivo.
Pharmacokinetics
Absorption
The systemic bioavailability of etodolac from etodolac capsules and tablets, USP is 100% as compared to solution and at least 80% as determined from mass balance studies. Etodolac is well absorbed and had a relative bioavailability of 100% when 200 mg capsules were compared with a solution of etodolac. Based on mass balance studies, the systemic availability of etodolac from either the tablet or capsule formulation is at least 80%. Etodolac does not undergo significant first-pass metabolism following oral administration. Mean (± 1 SD) peak plasma concentrations (Cmax) range from approximately 14 ± 4 to 37 ± 9 µg/mL after 200 to 600 mg single doses and are reached in 80 ± 30 minutes (see Table 1 for summary of pharmacokinetic parameters). The dose-proportionality based on the area under the plasma concentration-time curve (AUC) is linear following doses up to 600 mg every 12 hours. Peak concentrations are dose proportional for both total and free etodolac following doses up to 400 mg every 12 hours, but following a 600 mg dose, the peak is about 20% higher than predicted on the basis of lower doses. The extent of absorption of etodolac is not affected when etodolac tablets or capsules are administered after a meal. Food intake, however, reduces the peak concentration reached by approximately one-half and increases the time to peak concentration by 1.4 to 3.8 hours.
Table 1. Mean (CV%)* Pharmacokinetic Parameters of etodolac in Normal Healthy Adults and Various Special Populations PK
ParametersNormal Healthy Adults
(18-65)†Healthy Males
(18-65)Healthy Females
(27-65)Elderly
(>65)
(70-84)Hemodialysis
(24-65)
(n=9)Renal Impairment
(46-73)Hepatic Impairment
(34-60)(n=179) (n=176) (n=3) Dialysis On Dialysis Off (n=10) (n=9) NA = not available - * % Coefficient of variation
- † Age Range (years)
Tmax,h 1.4
(61%)*1.4
(60%)1.7
(60%)1.2
(43%)1.7
(88%)0.9
(67%)2.1
(46%)1.1
(15%)Oral Clearance, mL/h/kg (CL/F) 49.1
(33%)49.4
(33%)35.7
(28%)45.7
(27%)NA NA 58.3
(19%)42.0
(43%)Apparent Volume of Distribution, mL/kg (Vd/F) 393
(29%)394
(29%)300
(8%)414
(38%)NA NA NA NA Terminal Half-Life,h 6.4
(22%)6.4
(22%)7.9
(35%)6.5
(24%)5.1
(22%)7.5
(34%)NA 5.7
(24%)Distribution
The mean apparent volume of distribution (Vd/F) of etodolac is approximately 390 mL/kg. Etodolac is more than 99% bound to plasma proteins, primarily to albumin. The free fraction is less than 1% and is independent of etodolac total concentration over the dose range studied. It is not known whether etodolac is excreted in human milk; however, based on its physical-chemical properties, excretion into breast milk is expected. Data from in vitro studies, using peak serum concentrations at reported therapeutic doses in humans, show that the etodolac free fraction is not significantly altered by acetaminophen, ibuprofen, indomethacin, naproxen, piroxicam, chlorpropamide, glipizide, glyburide, phenytoin, and probenecid.
Metabolism
Etodolac is extensively metabolized in the liver. The role, if any, of a specific cytochrome P450 system in the metabolism of etodolac is unknown. Several etodolac metabolites have been identified in human plasma and urine. Other metabolites remain to be identified. The metabolites include 6-, 7-, and 8- hydroxylated-etodolac and etodolac glucuronide. After a single dose of 14C-etodolac, hydroxylated metabolites accounted for less than 10% of total drug in serum. On chronic dosing, hydroxylated-etodolac metabolite does not accumulate in the plasma of patients with normal renal function. The extent of accumulation of hydroxylated-etodolac metabolites in patients with renal dysfunction has not been studied. The hydroxylated-etodolac metabolites undergo further glucuronidation followed by renal excretion and partial elimination in the feces.
Excretion
The mean oral clearance of etodolac following oral dosing is 49 (± 16) mL/h/kg. Approximately 1% of an etodolac dose is excreted unchanged in the urine with 72% of the dose excreted into urine as parent drug plus metabolite:
-etodolac, unchanged 1% -etodolac glucuronide 13% -hydroxylated metabolites (6-, 7-, and 8-OH) 5% -hydroxylated metabolite glucuronides 20% -unidentified metabolites 33% Although renal elimination is a significant pathway of excretion for etodolac metabolites, no dosing adjustment in patients with mild to moderate renal dysfunction is generally necessary. The terminal half-life (t1/2) of etodolac is 6.4 hours (22% CV). In patients with severe renal dysfunction or undergoing hemodialysis, dosing adjustment is not generally necessary.
Fecal excretion accounted for 16% of the dose.
Special Populations
Geriatric
In etodolac clinical studies, no overall differences in safety or effectiveness were observed between these patients and younger patients. In pharmacokinetic studies, age was shown not to have any effect on etodolac half-life or protein binding, and there was no change in expected drug accumulation. Therefore no dosage adjustment is generally necessary in the elderly on the basis of pharmacokinetics (see PRECAUTIONS, Geriatric Use).
Etodolac is eliminated primarily by the kidney. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see WARNINGS, Renal Effects).
Pediatric
Safety and effectiveness in pediatric patients below the age of 18 years have not been established.
Race
Pharmacokinetic differences due to race have not been identified. Clinical studies included patients of many races, all of whom responded in a similar fashion.
Hepatic Insufficiency
Etodolac is predominantly metabolized by the liver. In patients with compensated hepatic cirrhosis, the disposition of total and free etodolac is not altered. Patients with acute and chronic hepatic diseases do not generally require reduced doses of etodolac compared to patients with normal hepatic function. However, etodolac clearance is dependent on liver function and could be reduced in patients with severe hepatic failure. Etodolac plasma protein binding did not change in patients with compensated hepatic cirrhosis given etodolac.
Renal Insufficiency
Etodolac pharmacokinetics have been investigated in subjects with renal insufficiency. Etodolac renal clearance was unchanged in the presence of mild-to-moderate renal failure (creatinine clearance 37 to 88 mL/min). Furthermore, there were no significant differences in the disposition of total and free etodolac in these patients. However, etodolac should be used with caution in such patients because, as with other NSAIDs, it may further decrease renal function in some patients. In patients undergoing hemodialysis, there was a 50% greater apparent clearance of total etodolac, due to a 50% greater unbound fraction. Free etodolac clearance was not altered, indicating the importance of protein binding in etodolac's disposition. Etodolac is not significantly removed from the blood in patients undergoing hemodialysis.
-
CLINICAL TRIALS
Analgesia
Controlled clinical trials in analgesia were single-dose, randomized, double-blind, parallel studies in three pain models, including dental extractions. The analgesic effective dose for etodolac established in these acute pain models was 200 to 400 mg. The onset of analgesia occurred approximately 30 minutes after oral administration. Etodolac 200 mg provided efficacy comparable to that obtained with aspirin (650 mg). Etodolac 400 mg provided efficacy comparable to that obtained with acetaminophen with codeine (600 mg + 60 mg). The peak analgesic effect was between 1 to 2 hours. Duration of relief averaged 4 to 5 hours for 200 mg of etodolac and 5 to 6 hours for 400 mg of etodolac as measured by when approximately half of the patients required remedication.
Osteoarthritis
The use of etodolac in managing the signs and symptoms of osteoarthritis of the hip or knee was assessed in double-blind, randomized, controlled clinical trials in 341 patients. In patients with osteoarthritis of the knee, etodolac, in doses of 600 to 1000 mg/day, was better than placebo in two studies. The clinical trials in osteoarthritis used b.i.d. dosage regimens.
Rheumatoid Arthritis
In a 3-month study with 426 patients, etodolac 300 mg b.i.d. was effective in management of rheumatoid arthritis and comparable in efficacy to piroxicam 20 mg/day. In a long-term study with 1,446 patients in which 60% of patients completed 6 months of therapy and 20% completed 3 years of therapy, etodolac in a dose of 500 mg b.i.d. provided efficacy comparable to that obtained with ibuprofen 600 mg q.i.d. In clinical trials of rheumatoid arthritis patients, etodolac has been used in combination with gold, d-penicillamine, chloroquine, corticosteroids, and methotrexate.
-
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of etodolac capsules and tablets, USP, and other treatment options before deciding to use etodolac capsules and tablets, USP. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
Etodolac Capsules and Tablets, USP are indicated:
- For acute and long-term use in the management of signs and symptoms of the following:
- Osteoarthritis
- Rheumatoid arthritis
- For the management of acute pain
- For acute and long-term use in the management of signs and symptoms of the following:
-
CONTRAINDICATIONS
Etodolac Capsules and Tablets, USP are contraindicated in patients with known hypersensitivity to etodolac or other ingredients in etodolac.
Etodolac Capsules and Tablets, USP should not be given to patients who have experienced asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see WARNINGS, Anaphylactoid Reactions and PRECAUTIONS, Pre-existing Asthma).
Etodolac capsules and tablets, USP are contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS).
-
WARNINGS
CARDIOVASCULAR EFFECTS
Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, myocardial infarction, and stroke, which can be fatal. All NSAIDs, both COX-2 selective and nonselective, may have a similar risk. Patients with known CV disease or risk factors for CV disease may be at greater risk. To minimize the potential risk for an adverse CV event in patients treated with an NSAID, the lowest effective dose should be used for the shortest duration possible. Physicians and patients should remain alert for the development of such events, even in the absence of previous CV symptoms. Patients should be informed about the signs and/or symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID does increase the risk of serious GI events (see WARNINGS, Gastrointestinal Effects - Risk of Ulceration, Bleeding, and Perforation).
Two large, controlled, clinical trials of a COX-2 selective NSAID for the treatment of pain in the first 10-14 days following CABG surgery found an increased incidence of myocardial infarction and stroke (see CONTRAINDICATIONS).
Hypertension
NSAIDs, including etodolac capsules and tablets, USP, can lead to onset of new hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events. Patients taking thiazides or loop diuretics may have impaired response to these therapies when taking NSAIDs. NSAIDs, including etodolac capsules and tablets,USP, should be used with caution in patients with hypertension. Blood pressure (BP) should be monitored closely during the initiation of NSAID treatment and throughout the course of therapy.
Congestive Heart Failure and Edema
Fluid retention and edema have been observed in some patients taking NSAIDs. Etodolac capsules and tablets, USP should be used with caution in patients with fluid retention or heart failure.
Gastrointestinal Effects - Risk of Ulceration, Bleeding, and Perforation
NSAIDs, including etodolac capsules and tablets, USP, can cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the stomach, small intestine or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients, who develop a serious upper GI adverse event on NSAID therapy, is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occur in approximately 1% of patients treated for 3-6 months, and in about 2-4% of patients treated for one year. These trends continue with longer duration of use, increasing the likelihood of developing a serious GI event at some time during the course of therapy. However, even short-term therapy is not without risk. Physicians should inform patients about the signs and/or symptoms of serious GI toxicity and what steps to take if they occur.
NSAIDs should be prescribed with extreme caution in those with a prior history of ulcer disease or gastrointestinal bleeding. Patients with a prior history of peptic ulcer disease, and/or gastrointestinal bleeding, and who use NSAIDs have a greater than 10-fold increased risk for developing a GI bleed compared to patients with neither of these risk factors. Other factors that increase the risk for GI bleeding in patients treated with NSAIDs include concomitant use of oral corticosteroids or anticoagulants, longer duration of NSAID therapy, smoking, use of alcohol, older age, and poor general health status. Most spontaneous reports of fatal GI events are in elderly or debilitated patients, and therefore, special care should be taken in treating this population.
To minimize the potential risk for an adverse GI event in patients treated with an NSAID, the lowest effective dose should be used for the shortest possible duration. Patients and physicians should remain alert for signs and symptoms of GI ulceration and bleeding during NSAID therapy and promptly initiate additional evaluation and treatment if a serious GI adverse event is suspected. This should include discontinuation of the NSAID until a serious GI adverse event is ruled out. For high risk patients, alternate therapies that do not involve NSAIDs should be considered.
Renal Effects
Long-term administration of NSAIDs has resulted in renal papillary necrosis and other renal injury. Renal toxicity has also been seen in patients in whom renal prostaglandins have a compensatory role in the maintenance of renal perfusion. In these patients, administration of a nonsteroidal antiinflammatory drug may cause a dose-dependent reduction in prostaglandin formation and, secondarily, in renal blood flow, which may precipitate overt renal decompensation. Patients at greater risk of this reaction are those with impaired renal function, heart failure, liver dysfunction, those taking diuretics and ACE inhibitors, and the elderly. Discontinuation of NSAID therapy is usually followed by recovery to the pretreatment state.
Renal pelvic transitional epithelial hyperplasia, a spontaneous change occurring with variable frequency, was observed with increased frequency in treated male rats in a 2-year chronic study.
Caution is recommended in patients with pre-existing kidney disease.
Advanced Renal Disease
No information is available from controlled clinical studies regarding the use of etodolac capsules and tablets, USP, in patients with advanced renal disease. Therefore, treatment with etodolac capsules and tablets, USP is not recommended in these patients with advanced renal disease. If etodolac capsules and tablets, USP therapy must be initiated, close monitoring of the patient's renal function is advisable.
Anaphylactoid Reactions
As with other NSAIDs, anaphylactoid reactions may occur in patients without prior exposure to etodolac capsules and tablets, USP. Etodolac capsules and tablets, USP should not be given to patients with the aspirin triad. This symptom complex typically occurs in asthmatic patients who experience rhinitis with or without nasal polyps, or who exhibit severe, potentially fatal bronchospasm after taking aspirin or other NSAIDs. Fatal reactions have been reported in such patients (see CONTRAINDICATIONS and PRECAUTIONS, General, Pre-existing Asthma). Emergency help should be sought in cases where an anaphylactoid reaction occurs.
Skin Reactions
NSAIDs, including etodolac capsules and tablets, USP, can cause serious skin adverse events such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Patients should be informed about the signs and symptoms of serious skin manifestations and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity.
Pregnancy
In late pregnancy, the third trimester, as with other NSAIDs, etodolac capsules and tablets, USP should be avoided because it may cause premature closure of the ductus arteriosus (see PRECAUTIONS, Pregnancy, Nonteratogenic Effects).
-
PRECAUTIONS
General
Etodolac capsules and tablets, USP cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Patients on prolonged corticosteroid therapy should have their therapy tapered solely if a decision is made to discontinue corticosteroids. The pharmacological activity of etodolac capsules and tablets, USP in reducing fever and inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
Hepatic Effects
Borderline elevations of one or more liver tests may occur in up to 15% of patients taking NSAIDs including etodolac capsules and tablets, USP. These laboratory abnormalities may progress, may remain unchanged, or may be transient with continuing therapy. Notable elevations of ALT or AST (approximately three or more times the upper limit of normal) have been reported in approximately 1% of patients in clinical trials with NSAIDs. In addition, rare cases of severe hepatic reactions, including jaundice and fatal fulminant hepatitis, liver necrosis and hepatic failure, some of them with fatal outcomes, have been reported.
A patient with symptoms and/or signs suggesting liver dysfunction, or in whom an abnormal liver test has occurred, should be evaluated for evidence of the development of a more severe hepatic reaction while on therapy with etodolac. If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), etodolac capsules and tablets, USP should be discontinued.
Hematological Effects
Anemia is sometimes seen in patients receiving NSAIDs including etodolac capsules and tablets, USP. This may be due to fluid retention, occult or gross GI blood loss, or an incompletely described effect upon erythropoiesis. Patients on long-term treatment with NSAIDs, including etodolac capsules and tablets, USP, should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia.
NSAIDs inhibit platelet aggregation and have been shown to prolong bleeding time in some patients. Unlike aspirin, their effect on platelet function is quantitatively less, of shorter duration, and reversible. Patients receiving etodolac capsules and tablets, USP, who may be adversely affected by alterations in platelet function, such as those with coagulation disorders or patients receiving anticoagulants, should be carefully monitored.
Pre-existing Asthma
Patients with asthma may have aspirin-sensitive asthma. The use of aspirin in patients with aspirin-sensitive asthmas has been associated with severe bronchospasm which can be fatal. Since cross reactivity, including bronchospasm, between aspirin and other nonsteroidal anti-inflammatory drugs has been reported in such aspirin-sensitive patients, etodolac capsules and tablets, USP should not be administered to patients with this form of aspirin sensitivity and should be used with caution in all patients with pre-existing asthma.
Information for Patients
Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
- Etodolac capsules and tablets, USP, like other NSAIDs, may cause serious CV side effects, such as MI or stroke, which may result in hospitalization and even death. Although serious CV events can occur without warning symptoms, patients should be alert for the signs and symptoms of chest pain, shortness of breath, weakness, slurring of speech, and should ask for medical advice when observing any indicative sign or symptoms. Patients should be apprised of the importance of this follow-up (see WARNINGS, CARDIOVASCULAR EFFECTS).
- Etodolac capsules and tablets, USP, like other NSAIDs, can cause GI discomfort and, rarely, serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (see WARNINGS, Gastrointestinal Effects - Risk of Ulceration, Bleeding, and Perforation).
- Etodolac capsules and tablets, USP, like other NSAIDs, can cause serious skin side effects such as exfoliative dermatitis, SJS, and TEN, which may result in hospitalizations and even death. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and blisters, fever, or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Patients should be advised to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible.
- Patients should promptly report signs or symptoms of unexplained weight gain or edema to their physicians.
- Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy.
- Patients should be informed of the signs of an anaphylactoid reaction (e.g. difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help (see WARNINGS).
- In late pregnancy, the third trimester, as with other NSAIDs, etodolac capsules and tablets, USP should be avoided because they may cause premature closure of the ductus arteriosus.
Laboratory Tests
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. Patients on long-term treatment with NSAIDs should have their CBC and a chemistry profile checked periodically for signs or symptoms of anemia. Appropriate measures should be taken in case such signs of anemia occur. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, etodolac capsules and tablets, USP should be discontinued.
Drug Interactions
ACE-inhibitors
Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE-inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE-inhibitors (see WARNINGS).
Antacids
The concomitant administration of antacids has no apparent effect on the extent of absorption of etodolac capsules and tablets, USP. However, antacids can decrease the peak concentration reached by 15% to 20% but have no detectable effect on the time-to-peak.
Aspirin
When etodolac capsules and tablets, USP is administered with aspirin, its protein binding is reduced, although the clearance of free etodolac is not altered. The clinical significance of this interaction is not known; however, as with other NSAIDs, concomitant administration of etodolac and aspirin is not generally recommended because of the potential of increased adverse effects.
Cyclosporine, Digoxin, Methotrexate
Etodolac, like other NSAIDs, through effects on renal prostaglandins, may cause changes in the elimination of these drugs leading to elevated serum levels of cyclosporine, digoxin, methotrexate, and increased toxicity. Nephrotoxicity associated with cyclosporine may also be enhanced. Patients receiving these drugs who are given etodolac, or any other NSAID, and particularly those patients with altered renal function, should be observed for the development of the specific toxicities of these drugs. NSAIDs, such as etodolac, should not be administered prior to or concomitantly with high doses of methotrexate. NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. In general, caution should be used when NSAIDs are administered concomitantly with methotrexate.
Diuretics
Etodolac has no apparent pharmacokinetic interaction when administered with furosemide or hydrochlorothiazide. Nevertheless, clinical studies, as well as postmarketing observations have shown that etodolac can reduce the natriuretic effect of furosemide and thiazides in some patients with possible loss of blood pressure control. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal insufficiency or failure (see WARNINGS, Renal Effects), as well as to assure diuretic efficacy.
Lithium
NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity. Careful monitoring of lithium levels is advised in the event NSAID dosage adjustments are required.
Phenylbutazone
Phenylbutazone causes increase (by about 80%) in the free fraction of etodolac. Although in vivo studies have not been done to see if etodolac clearance is changed by coadministration of phenylbutazone, it is not recommended that they be coadministered.
Warfarin
The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than that of users of either drug alone. Short-term pharmacokinetic studies have demonstrated that concomitant administration of warfarin and etodolac capsules and tablets, USP results in reduced protein binding of warfarin, but there was no change in the clearance of free warfarin. There was no significant difference in the pharmacodynamic effect of warfarin administered alone and warfarin administered with etodolac capsules and tablets, USP as measured by prothrombin time. Thus, concomitant therapy with warfarin and etodolac should not require dosage adjustment of either drug. However, caution should be exercised because there have been a few spontaneous reports of prolonged prothrombin times, with or without bleeding, in etodolac-treated patients receiving concomitant warfarin therapy. Close monitoring of such patients is therefore recommended.
Drug/Laboratory Test Interactions
The urine of patients who take etodolac can give a false-positive reaction for urinary bilirubin (urobilin) due to the presence of phenolic metabolites of etodolac. Diagnostic dip-stick methodology, used to detect ketone bodies in urine, has resulted in false-positive findings in some patients treated with etodolac. Generally, this phenomenon has not been associated with other clinically significant events. No dose relationship has been observed. Etodolac treatment is associated with a small decrease in serum uric acid levels. In clinical trials, mean decreases of 1 to 2 mg/dL were observed in arthritic patients receiving etodolac (600 to 1000 mg/day) after 4 weeks of therapy. These levels then remained stable for up to 1 year of therapy.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
No carcinogenic effect of etodolac was observed in mice or rats receiving oral doses of 15 mg/kg/day (45 to 89 mg/m2, respectively) or less for periods of 2 years or 18 months, respectively. Etodolac was not mutagenic in in vitro tests performed with S. typhimurium and mouse lymphoma cells as well as in an in vivo mouse micronucleus test. However, data from the in vitro human peripheral lymphocyte test showed an increase in the number of gaps (3.0 to 5.3% unstained regions in the chromatid without dislocation) among the etodolac-treated cultures (50 to 200 µg/mL) compared to negative controls (2.0%); no other difference was noted between the controls and drug-treated groups. Etodolac showed no impairment of fertility in male and female rats up to oral doses of 16 mg/kg (94 mg/m2). However, reduced implantation of fertilized eggs occurred in the 8 mg/kg group.
Pregnancy
Teratogenic Effects
Pregnancy Category C
In teratology studies, isolated occurrences of alterations in limb development were found and included polydactyly, oligodactyly, syndactyly, and unossified phalanges in rats and oligodactyly and synostosis of metatarsals in rabbits. These were observed at dose levels (2 to 14 mg/kg/day) close to human clinical doses. However, the frequency and the dosage group distribution of these findings in initial or repeated studies did not establish a clear drug or dose-response relationship. Animal reproduction studies are not always predictive of human response. There are no adequate and well-controlled studies in pregnant women. Etodolac capsules and tablets, USP should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects
Etodolac capsules and tablets, USP should be used during pregnancy only if the potential benefits justify the potential risk to the fetus. Because of the known effects of nonsteroidal anti-inflammatory drugs on the fetal cardiovascular system (closure of ductus arteriosus), use during pregnancy (particularly during the third trimester) should be avoided.
Labor and Delivery
In rat studies with NSAIDs, as with other drugs known to inhibit prostaglandin synthesis, an increased incidence of dystocia, delayed parturition, and decreased pup survival occurred. The effects of etodolac capsules and tablets, USP on labor and delivery in pregnant women are unknown.
Nursing Mothers
Trace amounts of some NSAIDs have been reported in human milk. It is not known whether etodalac is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from etodolac capsules and tablets, USP, a decision should be made whether to discontinue nursing or to discontinue the drug taking into account the importance of the drug to the mother.
Pediatric Use
Safety and effectiveness in pediatric patients below the age of 18 have not been established.
Geriatric Use
As with any NSAID, caution should be exercised in treating the elderly (65 years and older) and when increasing the dose (see WARNINGS).
In etodolac capsules and tablets, USP clinical studies, no overall differences in safety or effectiveness were observed between these patients and younger patients. In pharmacokinetic studies, age was shown not to have any effect on etodolac half-life or protein binding, and there was no change in expected drug accumulation. Therefore, no dosage adjustment is generally necessary in the elderly on the basis of pharmacokinetics (see CLINICAL PHARMACOLOGY, Special Populations).
Elderly patients may be more sensitive to the antiprostaglandin effects of NSAIDs (on the gastrointestinal tract and kidneys) than younger patients (see WARNINGS). In particular, elderly or debilitated patients who receive NSAID therapy seem to tolerate gastrointestinal ulceration or bleeding less well than other individuals, and most spontaneous reports of fatal GI events are in this population.
Etodolac is eliminated primarily by the kidney. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see WARNINGS, Renal Effects).
-
ADVERSE REACTIONS
In patients taking etodolac capsules and tablets, USP or other NSAIDs, the most frequently reported adverse experiences occurring in approximately 1-10% of patients are:
Gastrointestinal experiences including: abdominal pain, constipation, diarrhea, dyspepsia, flatulence, gross bleeding/perforation, heartburn, nausea, GI ulcers (gastric/duodenal), vomiting.
Other events including: abnormal renal function, anemia, dizziness, edema, elevated liver enzymes, headaches, increased bleeding time, pruritis, rashes, tinnitus.
Adverse-reaction information for etodolac was derived from 2,629 arthritic patients treated with etodolac capsules and tablets in double-blind and open-label clinical trials of 4 to 320 weeks in duration and worldwide postmarketing surveillance studies. In clinical trials, most adverse reactions were mild and transient. The discontinuation rate in controlled clinical trials, because of adverse events, was up to 10% for patients treated with etodolac.
New patient complaints (with an incidence greater than or equal to 1%) are listed below by body system. The incidences were determined from clinical trials involving 465 patients with osteoarthritis treated with 300 to 500 mg of etodolac b.i.d. (i.e., 600 to 1000 mg/day).
Incidence Greater Than Or Equal To 1% - Probably Causally Related
Body as a whole - Chills and fever.
Digestive system - Dyspepsia (10%), abdominal pain1, diarrhea1, flatulence1, nausea1, abdominal distension, epigastric pain, abnormal stools, constipation, gastritis, melena, vomiting.
Nervous system - Asthenia/malaise1, dizziness1, depression, nervousness, fatigue.
Skin and appendages - Pruritus, rash.
Special senses - Blurred vision, tinnitus.
Urogenital system - Dysuria, urinary frequency.
Musculoskeletal—Arthralgia.
Drug-related patient complaints occurring in fewer than 3%, but more than 1%, are unmarked.
- 1 Drug-related patient complaints occurring in 3 to 9% of patients treated with etodolac.
Incidence Less Than 1% - Probably Causally Related
(Adverse reactions reported only in worldwide postmarketing experience, not seen in clinical trials, are considered rarer and are italicized.)
Body as a whole - Allergic reaction, anaphylactic/anaphylactoid reactions (including shock).
Cardiovascular system - Hypertension, congestive heart failure, flushing, palpitations, syncope, vasculitis (including necrotizing and allergic).
Digestive system - Thirst, dry mouth, ulcerative stomatitis, anorexia, eructation, elevated liver enzymes, cholestatic hepatitis, hepatitis, cholestatic jaundice, duodenitis, jaundice, hepatic failure, liver necrosis, peptic ulcer with or without bleeding and/or perforation, intestinal ulceration, pancreatitis.
Hemic and lymphatic system - Ecchymosis, anemia, thrombocytopenia, bleeding time increased, agranulocytosis, hemolytic anemia, aplastic anemia, leukopenia, neutropenia, pancytopenia.
Metabolic and nutritional - Edema, serum creatinine increase, hyperglycemia in previously controlled diabetic patients.
Nervous system - Insomnia, somnolence.
Respiratory system - Asthma, pulmonary infiltration with eosinophilia.
Skin and appendages - Angioedema, sweating, urticaria, exfoliative dermatitis, vesiculobullous rash, cutaneous vasculitis with purpura, Stevens-Johnson Syndrome, toxic epidermal necrolysis, leukocytoclastic vasculitis, hyperpigmentation, erythema multiforme.
Special senses - Photophobia, transient visual disturbances.
Urogenital system - Elevated BUN, renal failure, renal insufficiency, renal papillary necrosis.
Incidence Less Than 1% - Causal Relationship Unknown
(Medical events occurring under circumstances where causal relationship to etodolac is uncertain. These reactions are listed as alerting information for physicians.)
Body as a whole - Infection, headache.
Cardiovascular system - Arrhythmias, myocardial infarction, cerebrovascular accident.
Digestive system - Esophagitis with or without stricture or cardiospasm, colitis, GI discomfort, burning sensation, blood in stools, gastralgia, upper abdominal discomfort.
Metabolic and nutritional - Change in weight.
Nervous system - Paresthesia, confusion, irritability.
Respiratory system - Bronchitis, bronchospasm, dyspnea, pharyngitis, rhinitis, sinusitis.
Skin and appendages - Alopecia, maculopapular rash, photosensitivity, skin peeling.
Special senses - Conjunctivitis, deafness, taste perversion, loss of taste.
Urogenital system - Cystitis, hematuria, leukorrhea, renal calculus, interstitial nephritis, uterine bleeding irregularities, renal impairment.
Musculoskeletal—Muscle pain.
Additional Adverse Reactions Reported with NSAIDs
Body as a whole - Sepsis, death
Cardiovascular system - Tachycardia
Digestive system - Gastric ulcers, gastritis, gastrointestinal bleeding, glossitis, hematemesis
Hemic and lymphatic system - Lymphadenopathy
Nervous system - Anxiety, dream abnormalities, convulsions, coma, hallucinations, meningitis, tremors, vertigo
Respiratory system - Respiratory depression, pneumonia
Urogenital system - Oliguria/polyuria, proteinuria
-
OVERDOSAGE
Symptoms following acute NSAID overdose are usually limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which are generally reversible with supportive care. Gastrointestinal bleeding can occur and coma has occurred following massive ibuprofen or mefenamic-acid overdose. Hypertension, acute renal failure, and respiratory depression may occur but are rare. Anaphylactoid reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following overdose.
Patients should be managed by symptomatic and supportive care following an NSAID overdose. There are no specific antidotes. Emesis and/or activated charcoal (60 to 100 g in adults, 1 to 2 g/kg in children) and/or osmotic cathartic may be indicated in patients seen within 4 hours of ingestion with symptoms or following a large overdose (5 to 10 times the usual dose). Forced diuresis, alkalinization of the urine, hemodialysis, or hemoperfusion would probably not be useful due to etodolac's high protein binding.
-
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of etodolac capsules and tablets, USP and other treatment options before deciding to use etodolac capsules and tablets, USP. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
After observing the response to initial therapy with etodolac capsules and tablets, USP, the dose and frequency should be adjusted to suit an individual patient's needs.
Dosage adjustment of etodolac capsules and tablets, USP is generally not required in patients with mild to moderate renal impairment. Etodolac should be used with caution in such patients, because, as with other NSAIDs, it may further decrease renal function in some patients with impaired renal function (see WARNINGS, Renal Effects).
Analgesia
The recommended total daily dose of etodolac for acute pain is up to 1000 mg, given as 200-400 mg every 6 to 8 hours. Doses of etodolac greater than 1000 mg/day have not been adequately evaluated in wellcontrolled trials.
Osteoarthritis and Rheumatoid Arthritis
The recommended starting dose of etodolac for the management of the signs and symptoms of osteoarthritis or rheumatoid arthritis is: 300 mg b.i.d., t.i.d., or 400 mg b.i.d., or 500 mg b.i.d. A lower dose of 600 mg/day may suffice for long-term administration. Physicians should be aware that doses above 1000 mg/day have not been adequately evaluated in well-controlled clinical trials.
In chronic conditions, a therapeutic response to therapy with etodolac is sometimes seen within one week of therapy, but most often is observed by two weeks. After a satisfactory response has been achieved, the patient's dose should be reviewed and adjusted as required.
-
HOW SUPPLIED
Etodolac Capsules and Tablets, USP are available as:
Etodolac Capsules, USP
300 mg capsules (light pink gelatin capsule, with black imprint "ETO 300 MG" on cap and body, white to off-white powder filling)
- in bottles of 21 NDC: 16590-488-21
- in bottles of 30 NDC: 16590-488-30
- in bottles of 60 NDC: 16590-488-60
- in bottles of 90 NDC: 16590-488-90
- in bottles of 180 NDC: 16590-488-82
Etodolac Tablets, USP
400 mg tablets (peach, oval shaped, film coated tablet engraved with "T88" on one side and other side plain)
- in bottles of 20 NDC: 16590-092-20
- in bottles of 28 NDC: 16590-092-28
- in bottles of 30 NDC: 16590-092-30
- in bottles of 56 NDC: 16590-092-56
- in bottles of 60 NDC: 16590-092-60
- in bottles of 90 NDC: 16590-092-90
- in bottles of 120 NDC: 16590-092-72
- in bottles of 180 NDC: 16590-092-82
Relabeling and Repackaging by:
STAT Rx USA LLC
Gainesville, GA 30501
-
Medication GuideforNon-Steroidal Anti-Inflammatory Drugs (NSAIDs)
(See the end of this Medication Guide for a list of prescription NSAID medicines.)
What is the most important information I should know about medicines called Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
NSAID medicines may increase the chance of a heart attack or stroke that can lead to death. This chance increases:
- with longer use of NSAID medicines
- in people who have heart disease
NSAID medicines should never be used right before or after a heart surgery called a "coronary artery bypass graft (CABG)."
NSAID medicines can cause ulcers and bleeding in the stomach and intestines at any time during treatment. Ulcers and bleeding:
- can happen without warning symptoms
- may cause death
The chance of a person getting an ulcer or bleeding increases with:
- taking medicines called "corticosteroids" and "anticoagulants"
- longer use
- smoking
- drinking alcohol
- older age
- having poor health
NSAID medicines should only be used:
- exactly as prescribed
- at the lowest dose possible for your treatment
- for the shortest time needed
What are Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
NSAID medicines are used to treat pain and redness, swelling, and heat (inflammation) from medical conditions such as:
- different types of arthritis
- menstrual cramps and other types of short-term pain
Who should not take a Non-Steroidal Anti-Inflammatory Drug (NSAID)?
Do not take an NSAID medicine:
- if you had an asthma attack, hives, or other allergic reaction with aspirin or any other NSAID medicine
- for pain right before or after heart bypass surgery
Tell your healthcare provider:
- about all of your medical conditions.
- about all of the medicines you take. NSAIDs and some other medicines can interact with each other and cause serious side effects. Keep a list of your medicines to show to your healthcare provider and pharmacist.
- if you are pregnant. NSAID medicines should not be used by pregnant women late in their pregnancy.
- if you are breastfeeding. Talk to your doctor.
What are the possible side effects of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
Serious side effects include: Other side effects include: - heart attack
- stroke
- high blood pressure
- heart failure from body swelling (fluid retention)
- kidney problems including kidney failure
- bleeding and ulcers in the stomach and intestine
- low red blood cells (anemia)
- life-threatening skin reactions
- life-threatening allergic reactions
- liver problems including liver failure
- asthma attacks in people who have asthma
- stomach pain
- constipation
- diarrhea
- gas
- heartburn
- nausea
- vomiting
- dizziness
Get emergency help right away if you have any of the following symptoms:
- shortness of breath or trouble breathing
- chest pain
- weakness in one part or side of your body
- slurred speech
- swelling of the face or throat
Stop your NSAID medicine and call your healthcare provider right away if you have any of the following symptoms:
- nausea
- more tired or weaker than usual
- itching
- your skin or eyes look yellow
- stomach pain
- flu-like symptoms
- vomit blood
- there is blood in your bowel movement or it is black and sticky like tar
- unusual weight gain
- skin rash or blisters with fever
- swelling of the arms and legs, hands and feet
These are not all the side effects with NSAID medicines. Talk to your healthcare provider or pharmacist for more information about NSAID medicines.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Other information about Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
- Aspirin is an NSAID medicine but it does not increase the chance of a heart attack. Aspirin can cause bleeding in the brain, stomach, and intestines. Aspirin can also cause ulcers in the stomach and intestines.
- Some of these NSAID medicines are sold in lower doses without a prescription (over–the–counter). Talk to your healthcare provider before using over–the–counter NSAIDs for more than 10 days.
NSAID medicines that need a prescription
Generic Name Tradename - * Vicoprofen contains the same dose of ibuprofen as over-the-counter (OTC) NSAIDs, and is usually used for less than 10 days to treat pain. The OTC NSAID label warns that long term continuous use may increase the risk of heart attack or stroke.
Celecoxib Celebrex Diclofenac Cataflam, Voltaren, Arthrotec (combined with misoprostol) Diflunisal Dolobid Etodolac Lodine, Lodine XL Fenoprofen Nalfon, Nalfon 200 Flurbiprofen Ansaid Ibuprofen Motrin, Tab-Profen, Vicoprofen* (combined with hydrocodone), Combunox (combined with oxycodone) Indomethacin Indocin, Indocin SR, Indo-Lemmon, Indomethagan Ketoprofen Oruvail Ketorolac Toradol Mefenamic Acid Ponstel Meloxicam Mobic Nabumetone Relafen Naproxen Naprosyn, Anaprox, Anaprox DS, EC-Naprosyn, Naprelan, Naprapac (copackaged with lansoprazole) Oxaprozin Daypro Piroxicam Feldene Sulindac Clinoril Tolmetin Tolectin, Tolectin DS, Tolectin 600 This Medication Guide has been approved by the U.S. Food and Drug Administration.
Manufactured by: Taro Pharmaceutical Industries Ltd., Haifa Bay, Israel 26110
Distributed by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532Revised: February, 2010
89927-0210-4
- PACKAGE LABEL - ETODOLAC - 300 MG CAPSULES
- PACKAGE LABEL - ETODOLAC - 400 MG TABLETS
-
INGREDIENTS AND APPEARANCE
ETODOLAC
etodolac capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 16590-488(NDC:51672-4017) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ETODOLAC (UNII: 2M36281008) (ETODOLAC - UNII:2M36281008) ETODOLAC 300 mg Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) FERROSOFERRIC OXIDE (UNII: XM0M87F357) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ALCOHOL (UNII: 3K9958V90M) GELATIN (UNII: 2G86QN327L) ISOPROPYL ALCOHOL (UNII: ND2M416302) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color purple (LIGHT PINK) Score no score Shape CAPSULE Size 22mm Flavor Imprint Code ETO;300;MG Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 16590-488-21 21 in 1 BOTTLE 2 NDC: 16590-488-60 60 in 1 BOTTLE 3 NDC: 16590-488-82 180 in 1 BOTTLE 4 NDC: 16590-488-90 90 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075078 04/30/1998 ETODOLAC
etodolac tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 16590-092(NDC:51672-4018) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ETODOLAC (UNII: 2M36281008) (ETODOLAC - UNII:2M36281008) ETODOLAC 400 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSES (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color pink (Peach) Score no score Shape OVAL Size 19mm Flavor Imprint Code T88 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 16590-092-20 20 in 1 BOTTLE 2 NDC: 16590-092-28 28 in 1 BOTTLE 3 NDC: 16590-092-30 30 in 1 BOTTLE 4 NDC: 16590-092-56 56 in 1 BOTTLE 5 NDC: 16590-092-60 60 in 1 BOTTLE 6 NDC: 16590-092-72 120 in 1 BOTTLE 7 NDC: 16590-092-82 180 in 1 BOTTLE 8 NDC: 16590-092-90 90 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075074 03/11/1998 Labeler - STAT Rx USA LLC (786036330) Registrant - PSS World Medical Inc. (101822682) Establishment Name Address ID/FEI Business Operations STAT Rx USA LLC 786036330 relabel, repack
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.