GOCOVRI- amantadine capsule, coated pellets
GOCOVRI by
Drug Labeling and Warnings
GOCOVRI by is a Prescription medication manufactured, distributed, or labeled by Adamas Pharma, LLC, Catalent Pharma Solutions (Somerset, NJ), Catalent Pharma Solutions (Morrisville, NC), Sharp Corporation, Allentown, PA, Sharp Corporation, Conshohocken, PA, Moehs Catalana SL. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use GOCOVRI® safely and effectively. See full prescribing information for GOCOVRI.
GOCOVRI® (amantadine) extended release capsules, for oral use
Initial U.S. Approval: 1968RECENT MAJOR CHANGES
Dosage and Administration, Dosing Information (2.1) 03/2020
INDICATIONS AND USAGE
GOCOVRI® is indicated for the treatment of dyskinesia in patients with Parkinson’s disease receiving levodopa-based therapy, with or without concomitant dopaminergic medications (1)
DOSAGE AND ADMINISTRATION
- Administer orally once daily at bedtime (2.1, 2.3)
- The initial daily dosage is 137 mg; after 1 week, increase to the recommended daily dosage of 274 mg (2.1)
- Swallow whole; may sprinkle contents on soft food (2.2)
- May be taken with or without food; avoid use with alcohol (2.2)
- A lower dosage is recommended for patients with moderate or severe renal impairment (2.3)
DOSAGE FORMS AND STRENGTHS
Extended release capsules: 68.5 mg and 137 mg (3)
CONTRAINDICATIONS
GOCOVRI is contraindicated in patients with end stage renal disease (4)
WARNINGS AND PRECAUTIONS
- Falling Asleep During Activities of Daily Living: Advise patients prior to treatment; ordinarily discontinue if occurs (5.1)
- Suicidality and Depression: Monitor patients for depressed mood, depression, or suicidal ideation or behavior (5.2)
- Hallucinations/Psychotic Behavior: Patients with major psychotic disorder should ordinarily not be treated with GOCOVRI; observe patients for the occurrence of hallucinations throughout treatment, especially at initiation and after dose increases (5.3)
- Dizziness and Orthostatic Hypotension: Monitor patients for dizziness and orthostatic hypotension, especially after starting GOCOVRI or increasing the dose (5.4)
- Withdrawal-Emergent Hyperpyrexia and Confusion: Avoid sudden discontinuation (5.5)
- Impulse Control/Compulsive Behaviors: Ask patients about increased gambling urges, sexual urges, uncontrolled spending or other urges; consider dose reduction or discontinuation if occurs (5.6)
ADVERSE REACTIONS
The most commonly observed adverse reactions occurring at a frequency of >10% and greater than placebo were hallucination, dizziness, dry mouth, peripheral edema, constipation, fall, and orthostatic hypotension (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Adamas Pharma, LLC at 1-833-223-2627 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch.
DRUG INTERACTIONS
- Other Anticholinergic Drugs: Doses should be reduced if atropine-like effects occur (7.1)
- Drugs Affecting Urinary pH: Excretion increases with acidic urine; possible accumulation with urine change towards alkaline (7.2)
- Live Attenuated Influenza Vaccines: Not recommended during use (7.3)
- Alcohol: Concomitant use not recommended (7.4)
USE IN SPECIFIC POPULATIONS
- Pregnancy: Based on animal data, may cause fetal harm (8.1)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Information
2.2 Administration Information
2.3 Dosing in Patients with Renal Impairment
2.4 Discontinuation and Missed Dose
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Falling Asleep During Activities of Daily Living and Somnolence
5.2 Suicidality and Depression
5.3 Hallucinations/Psychotic Behavior
5.4 Dizziness and Orthostatic Hypotension
5.5 Withdrawal-Emergent Hyperpyrexia and Confusion
5.6 Impulse Control/Compulsive Behaviors
6 ADVERSE REACTIONS
6.1. Clinical Trial Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Other Anticholinergic Drugs
7.2 Drugs Affecting Urinary pH
7.3 Live Attenuated Influenza Vaccines
7.4 Alcohol
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6. Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Information
The initial daily dosage of GOCOVRI is 137 mg, administered orally once daily at bedtime. After one week, increase to the recommended dosage of 274 mg (two 137 mg capsules) once daily at bedtime.
GOCOVRI is not interchangeable with other amantadine immediate- or extended-release products.
2.2 Administration Information
GOCOVRI should be swallowed whole. Do not crush, chew or divide capsules. If needed, GOCOVRI may be administered by carefully opening and sprinkling the entire contents on a small amount (teaspoonful) of soft food, such as applesauce. The drug/food mixture should be swallowed immediately without chewing. Do not store mixture for future use.
GOCOVRI can be taken with or without food [see Clinical Pharmacology (12.3)].
Concomitant use of GOCOVRI with alcohol is not recommended [see Drug Interactions (7.4)].
It is recommended to avoid sudden discontinuation of GOCOVRI [see Dosage and Administration (2.4)].
2.3 Dosing in Patients with Renal Impairment
The initial and maximum recommended daily dosage of GOCOVRI for patients with renal impairment (creatinine clearance estimated by Modification of Diet in Renal Disease (MDRD) method) is provided below.
1 Increase, if needed, after one week of the initial dosage Creatinine Clearance Dosage Mild renal impairment
(60 to 89 mL/min/1.73 m2)Initial dosage: 137 mg once daily at bedtime. After one week, increase to recommended dosage of 274 mg once daily at bedtime. Moderate renal impairment
(30 to 59 mL/min/1.73 m2)Initial dosage: 68.5 mg once daily at bedtime
Maximum recommended dosage: 137 mg once daily at bedtime1Severe renal impairment
(15 to 29 mL/min/1.73 m2)68.5 mg once daily at bedtime End Stage Renal Disease
(below 15 mL/min/1.73 m2)Contraindicated 2.4 Discontinuation and Missed Dose
Rapid dose reduction or withdrawal of GOCOVRI may cause adverse reactions [see Warnings and Precautions (5.5)]. Therefore, to stop GOCOVRI in patients who have been on the drug for more than 4 weeks, their dose should typically, if possible, be reduced by half for their final week of dosing.
If a dose of GOCOVRI is missed, the next dose should be taken as scheduled.
-
3 DOSAGE FORMS AND STRENGTHS
GOCOVRI is available as extended release capsules for oral administration. Each capsule contains 68.5 mg or 137 mg of amantadine.
The 68.5 mg capsule is a white opaque size #2 capsule, with black printing of ‘ADAMAS’ on front and ‘85’ on back of the cap and three black bands printed on body of capsule.
The 137 mg capsule is a light blue opaque size #0 capsule, with black printing of ‘ADAMAS’ on front and ‘170’ on back of the cap and three black bands printed on body of capsule.
-
4 CONTRAINDICATIONS
GOCOVRI is contraindicated in patients with end stage renal disease (ie., creatinine clearance below 15 mL/min/1.73 m2) [see Clinical Pharmacology (12.3)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Falling Asleep During Activities of Daily Living and Somnolence
Patients treated for Parkinson’s disease have reported falling asleep while engaged in activities of daily living, including the operation of motor vehicles, which sometimes has resulted in accidents. Patients may not perceive warning signs, such as excessive drowsiness, or they may report feeling alert immediately prior to the event. In controlled clinical trials, somnolence and fatigue were reported as adverse reactions in 4% of patients treated with GOCOVRI 274 mg and 1% for placebo.
Before initiating treatment with GOCOVRI, advise patients of the potential to develop drowsiness and specifically ask about factors that may increase the risk for somnolence with GOCOVRI, such as concomitant sedating medications or the presence of a sleep disorder. If a patient develops daytime sleepiness or episodes of falling asleep during activities that require full attention (e.g., driving a motor vehicle, conversations, eating), GOCOVRI should ordinarily be discontinued.
If a decision is made to continue GOCOVRI, patients should be advised not to drive and to avoid other potentially dangerous activities. There is insufficient information to establish that dose reduction will eliminate episodes of falling asleep while engaged in activities of daily living or daytime somnolence.
5.2 Suicidality and Depression
In controlled clinical trials, suicidal ideation or suicide attempt was reported in 2% of GOCOVRI-treated patients and 0% of placebo-treated patients. Depression or depressed mood was reported in 6% of GOCOVRI-treated patients and 1% of placebo-treated patients. Confusional state was reported in 3% of GOCOVRI-treated patients and 2% of placebo-treated patients. Apathy was reported in 2% of GOCOVRI-treated patients and 0% of placebo-treated patients.
Monitor patients for depression, including suicidal ideation or behavior. Prescribers should consider whether the benefits outweigh the risks of treatment with GOCOVRI in patients with a history of suicidality or depression.
5.3 Hallucinations/Psychotic Behavior
Patients with a major psychotic disorder should ordinarily not be treated with GOCOVRI because of the risk of exacerbating psychosis. In controlled trials, the incidence of patients who experienced visual hallucination, auditory hallucination, delusions, illusions, or paranoia was 25% in patients treated with GOCOVRI 274 mg and 3% in placebo-treated patients. Hallucinations caused discontinuation of treatment in 8% of GOCOVRI-treated patients and 0% of placebo-treated patients.
Observe patients for the occurrence of hallucinations throughout treatment, especially at initiation and after dose increases.
5.4 Dizziness and Orthostatic Hypotension
In controlled clinical trials, 29% of GOCOVRI-treated patients and 2% of placebo-treated patients experienced dizziness, syncope, orthostatic hypotension, presyncope, postural dizziness or hypotension. In GOCOVRI-treated patients, 3% discontinued study treatment because of dizziness, postural dizziness, or syncope, compared to 0% of placebo-treated patients.
Monitor patients for dizziness and orthostatic hypotension, especially after starting GOCOVRI or increasing the dose. Concomitant use of alcohol when using GOCOVRI is not recommended [see Drug Interactions (7.4)].
5.5 Withdrawal-Emergent Hyperpyrexia and Confusion
A symptom complex resembling neuroleptic malignant syndrome (characterized by elevated temperature, muscular rigidity, altered consciousness, and autonomic instability), with no other obvious etiology, has been reported in association with rapid dose reduction, withdrawal of, or changes in drugs that increase central dopaminergic tone.
Abrupt discontinuation of GOCOVRI may cause an increase in the symptoms of Parkinson’s disease or cause delirium, agitation, delusions, hallucinations, paranoid reaction, stupor, anxiety, depression, or slurred speech. It is recommended to avoid sudden discontinuation of GOCOVRI [see Dosing Information (2.4)].
5.6 Impulse Control/Compulsive Behaviors
Patients can experience intense urges to gamble, increased sexual urges, intense urges to spend money, binge eating, and/or other intense urges, and the inability to control these urges while taking one or more of the medications, including GOCOVRI, that increase central dopaminergic tone. In some cases, these urges were reported to have stopped when the dose was reduced or the medication was discontinued. Because patients may not recognize these behaviors as abnormal, it is important for prescribers to specifically ask patients or their caregivers about the development of new or increased gambling urges, sexual urges, uncontrolled spending, or other urges while being treated with GOCOVRI. Consider dose reduction or stopping the medication if a patient develops such urges while taking GOCOVRI.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are described in more detail elsewhere in the labeling:
- Falling Asleep During Activities of Daily Living and Somnolence [see Warnings and Precautions (5.1)]
- Suicidality and Depression [see Warnings and Precautions (5.2)]
- Hallucinations/Psychotic Behavior [see Warnings and Precautions (5.3)]
- Dizziness and Orthostatic Hypotension [see Warnings and Precautions (5.4)]
- Withdrawal-Emergent Hyperpyrexia and Confusion [see Warnings and Precautions (5.5)]
- Impulse Control/Compulsive Behaviors [see Warnings and Precautions (5.6)]
6.1. Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Placebo-Controlled Trials
GOCOVRI was evaluated in two double-blind, placebo-controlled efficacy trials of similar design and population: Study 1 (123 patients) and Study 2 (75 patients). The study population was approximately 56% male and 94% white, with a mean age of 65 years (age range from 34 years to 82 years). The mean duration of levodopa-induced dyskinesia was 4 years (range 0.1 to 14 years). Active treatment started at 137 mg once daily for one week, followed by a dose increase to 274 mg once daily. The treatment duration was 25 weeks for Study 1 and 13 weeks for Study 2. Of the 100 patients in the safety population described below, 39 patients were treated with GOCOVRI for 24 weeks. The safety data for these trials were pooled.
The most common adverse reactions reported in >10% of GOCOVRI-treated patients and more frequently than on placebo were: hallucination, dizziness, dry mouth, peripheral edema, constipation, falls, and orthostatic hypotension.
The overall rate of discontinuation because of adverse reactions for GOCOVRI-treated patients was 20%, compared to 8% for placebo-treated patients. Adverse reactions that led to treatment discontinuation in at least 2% of patients were hallucination (8% GOCOVRI vs. 0% placebo), dry mouth (3% GOCOVRI vs. 0% placebo), peripheral edema (3% GOCOVRI vs. 0% placebo), blurred vision (GOCOVRI 3% vs. 0% placebo), postural dizziness and syncope (GOCOVRI 2% vs. 0% placebo), abnormal dreams (GOCOVRI 2% vs. 1% placebo), dysphagia (GOCOVRI 2% vs. 0% placebo), and gait disturbance (GOCOVRI 2% vs. 0% placebo).
Table 1: Adverse Reactions Reported for ≥ 3% of Patients Treated with 274 mg GOCOVRI in Study 1 and Study 2 (Pooled Analysis) a=Includes visual hallucinations and auditory hallucinations
b=Includes anxiety and generalized anxiety
c=Includes orthostatic hypotension, postural dizziness, syncope, presyncope, and hypotension
d=The denominator is all male patients in the safety population randomized to GOCOVRI (n=54) or placebo (n=57)Adverse Reactions GOCOVRI 274 mg
N=100
%Placebo
N=98
%Psychiatric disorders Hallucinationa 21 3 Anxietyb 7 3 Insomnia 7 2 Depression/Depressed mood 6 1 Abnormal dreams 4 2 Confusional state 3 2 Nervous system disorders Dizziness 16 1 Headache 6 4 Dystonia 3 1 Gastrointestinal disorders Dry mouth 16 1 Constipation 13 3 Nausea 8 3 Vomiting 3 0 General disorders and administration site conditions Peripheral edema 16 1 Gait disturbance 3 0 Injury, poisoning and procedural complications Fall 13 7 Contusion 6 1 Infections and infestations Urinary tract infection 10 5 Skin and subcutaneous tissue disorders Livedo reticularis 6 0 Pigmentation disorder 3 0 Metabolism and nutrition disorders Decreased appetite 6 1 Vascular disorders Orthostatic hypotensionc 13 1 Eye disorders Blurred vision 4 1 Cataract 3 1 Dry eye 3 0 Musculoskeletal and connective tissue disorders Joint swelling 3 0 Muscle spasms 3 0 Reproductive system and breast disorders Benign prostatic hyperplasiad 6 2 Respiratory, thoracic and mediastinal disorders Cough 3 0 Other clinically relevant adverse reactions observed at <3% included somnolence, fatigue, suicide ideation or attempt, apathy, delusions, illusions, and paranoia [see Warnings and Precautions (5.1, 5.2, 5.3)].
Difference in the Frequency of Adverse Reactions by Gender
Adverse reactions reported more frequently in women treated with 274 mg of GOCOVRI (n=46), compared to men (n=54), were: dry mouth (22% women, 11% men), nausea (13% women, 4% men), livedo reticularis (13% women, 0% men), abnormal dreams (9% women, 0% men) and cataracts (7% women, 0% men).
Men treated with 274 mg of GOCOVRI reported the following adverse reactions more frequently than women: dizziness (20% men, 11% women), peripheral edema (19% men, 11% women), anxiety (11% men, 2% women), orthostatic hypotension (7% men, 2% women) and gait disturbance (6% men, 0% women).
Difference in the Frequency of Adverse Reactions by Age
Hallucinations (visual or auditory) were reported in 31% of GOCOVRI-treated patients age 65 years and over (n=52), compared to 10% in patients below the age of 65 years (n=48). Falls were reported in 17% of GOCOVRI-treated patients age 65 and over, compared to 8% of patients below age 65. Orthostatic hypotension was reported in 8% of patients age 65 and over, compared to 2% of patients below age 65.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of GOCOVRI. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Nervous System Disorders: Seizure.
-
7 DRUG INTERACTIONS
7.1 Other Anticholinergic Drugs
Products with anticholinergic properties may potentiate the anticholinergic-like side effects of amantadine. The dose of anticholinergic drugs or of GOCOVRI should be reduced if atropine-like effects appear when these drugs are used concurrently.
7.2 Drugs Affecting Urinary pH
The pH of the urine has been reported to influence the excretion rate of amantadine. Urine pH is altered by diet, drugs (e.g., carbonic anhydrase inhibitors, sodium bicarbonate), and clinical state of the patient (e.g., renal tubular acidosis or severe infections of the urinary tract).
Since the excretion rate of amantadine increases rapidly when the urine is acidic, the administration of urine acidifying drugs may increase the elimination of the drug from the body. Alterations of urine pH towards the alkaline condition may lead to an accumulation of the drug with a possible increase in adverse reactions. Monitor for efficacy or adverse reactions under conditions that alter the urine pH to more acidic or alkaline, respectively.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate data on the developmental risk associated with use of amantadine in pregnant women. Animal studies suggest a potential risk for fetal harm with amantadine. In mice and rats, adverse developmental effects (embryolethality, increased incidence of malformations, and reduced fetal body weight) were observed when amantadine was administered to pregnant animals at clinically relevant doses [see Data].
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The background risk for major birth defects and miscarriage in patients with Parkinson’s disease is unknown.
Data
Animal Data
The effects of amantadine on development have not been tested in studies conducted in animals using currently recommended methodology; however, developmental toxicity studies of amantadine have been reported in the published literature.
In mice, oral administration of amantadine (0, 10, or 40 mg/kg/day) to pregnant animals during organogenesis (gestation days 7-12) resulted in embryolethality and reduced fetal body weight at the highest dose tested, which was associated with maternal toxicity. The no-effect dose for developmental toxicity in mice (10 mg/kg/day) is less than the recommended human dose (RHD) of 274 mg/day, based on body surface area (mg/m2).
In rats, oral administration of amantadine (0, 40 or 120 mg/kg/day) to pregnant animals during organogenesis (gestation days 7-12) resulted in embryolethality and reduced fetal body weight at the highest dose. The no-effect dose for developmental toxicity in this study (40 mg/kg/day) is approximately equal to the RHD on a mg/m2 basis.
In another study in pregnant rats, oral administration of amantadine during organogenesis (gestation days 7-14) resulted in an increase in visceral and skeletal malformations at oral doses of 50 and 100 mg/kg/day. The no-effect dose for teratogenicity in this study (37 mg/kg/day) is approximately equal to the RHD on a mg/m2 basis.
Evaluation of parturition, lactation, and post-natal development in a limited number of litters from the mouse and rat studies described above revealed reductions in live litter size and pup weights at birth at 40 mg/kg/day in mice and 120 mg/kg/day in rats.
8.2 Lactation
Risk Summary
Amantadine is excreted into human milk, but amounts have not been quantified. There is no information on the risk to a breastfed infant.
Amantadine may alter breast milk production or excretion [see Data].
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for GOCOVRI and any potential adverse effects on the breastfed infant from GOCOVRI or from the underlying maternal condition.
Data
In published studies, amantadine reduced serum prolactin levels and the symptoms of galactorrhea in patients taking neuroleptic drugs. The effect of amantadine on milk supply has not been evaluated in nursing mothers.
8.4 Pediatric Use
The safety and effectiveness of GOCOVRI in pediatric patients have not been established.
8.5 Geriatric Use
The majority of people with Parkinson’s disease are 65 years and older. In Phase 3 clinical trials, the mean age of patients at study entry was 65 years. Of the total number of patients in clinical studies of GOCOVRI, 46% were less than 65 years of age, 39% were 65-74 years of age, and 15% were 75 years of age or older.
Hallucinations and falls occurred more frequently in patients 65 years of age or older, compared to those less than 65 years of age [see Adverse Reactions (6.1)].
No dose adjustment is recommended on the basis of age. GOCOVRI is known to be substantially excreted by the kidney, and the risk of adverse reactions may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.3)].
8.6. Renal Impairment
GOCOVRI is contraindicated for use in patients with end-stage renal disease (creatinine clearance lower than 15 mL/min/1.73 m2).
For patients with moderate renal impairment (creatinine clearance between 30 and 59 mL/min/1.73 m2), a 50% dose reduction of GOCOVRI dosage to a starting daily dose of 68.5 mg daily at bedtime for a week, to a maximum dosage of 137 mg daily at bedtime is recommended.
For patients with severe renal impairment (creatinine clearance between 15 and 29 mL/min/1.73 m2), a daily dose of 68.5 mg at bedtime is the recommended initial and maximum dosage [see Dosage and Administration (2.3)].
Creatinine clearance values are estimated by the Modification of Diet in Renal Disease (MDRD) method.
-
10 OVERDOSAGE
Deaths have been reported from overdose with amantadine. The lowest reported acute lethal dose was 1 gram of amantadine hydrochloride (equivalent to 0.8 g amantadine). Acute toxicity may be attributable to the anticholinergic effects of amantadine. Drug overdose has resulted in cardiac, respiratory, renal, or central nervous system toxicity. Pulmonary edema and respiratory distress (including adult respiratory distress syndrome, ARDS) have been reported with amantadine; renal dysfunction, including increased BUN and decreased creatinine clearance, can occur.
Central nervous system effects that have been reported with overdose include agitation, aggressive behavior, hypertonia, hyperkinesia, ataxia, tremor, disorientation, depersonalization, fear, delirium, psychotic reactions, lethargy, and coma. Seizures may be exacerbated in patients with prior history of seizure disorders. Hyperthermia has occurred with amantadine overdose.
For acute overdosing, general supportive measures should be employed along with immediate gastric decontamination if appropriate. Give intravenous fluids if necessary. The excretion rate of amantadine increases with acidification of urine, which may increase the elimination of the drug. Monitor patients for arrhythmias and hypotension. Electrocardiographic monitoring may be needed after ingestion because arrhythmias have been reported after overdose, including arrhythmias with fatal outcomes. Adrenergic agents, such as isoproterenol, in patients with an amantadine overdose has been reported to induce arrhythmias.
Monitor blood electrolytes, urine pH, and urinary output. Although amantadine is not efficiently removed by hemodialysis, this procedure may be useful in the treatment of amantadine toxicity in patients with renal failure.
-
11 DESCRIPTION
GOCOVRI contains amantadine in an extended release formulation. The active ingredient in GOCOVRI is amantadine hydrochloride.
The chemical name for amantadine hydrochloride is tricyclo [3.3.1.1 3,7] decan-1-amine, hydrochloride or 1-adamantanamine hydrochloride with the following structural formula:
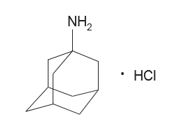
The molecular formula is C10H17NHCl and the molecular weight is 187.71 (g/mol). Amantadine hydrochloride is a white crystalline powder and is non-hygroscopic, practically insoluble in ether, sparingly soluble in methylene chloride, soluble in chloroform, and freely soluble in water, ethanol, and methanol.
GOCOVRI capsules are for oral use. Each capsule contains 68.5 mg or 137 mg amantadine (as 85 mg or 170 mg amantadine hydrochloride, respectively). Capsules also contain the following inactive ingredients: copovidone, ethylcellulose, hypromellose, magnesium stearate, medium-chain triglycerides, microcrystalline cellulose, povidone, and talc in a hard gelatin capsule.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism by which amantadine exerts efficacy in the treatment of dyskinesia in patients with Parkinson’s disease is unknown. Amantadine is a weak uncompetitive antagonist of the NMDA receptor. Amantadine has not been shown to possess direct anticholinergic activity in animal studies; however, it exhibits anticholinergic-like side effects such as dry mouth, urinary retention, and constipation in humans. Amantadine may have direct and indirect effects on dopamine neurons; it exerts dopaminergic-like side effects such as hallucinations and dizziness in humans.
12.2 Pharmacodynamics
The effect of amantadine on QT prolongation was not studied in a dedicated thorough QT study.
Alcohol consumption may increase the potential for CNS effects such as dizziness, confusion, lightheadedness, and orthostatic hypotension [see Drug Interactions (7.4)].
12.3 Pharmacokinetics
GOCOVRI is an extended release formulation. The pharmacokinetics of amantadine from 68.5 mg, 137 mg, and 274 mg of GOCOVRI are dose proportional in healthy subjects.
Absorption
After a single dose bedtime administration of GOCOVRI, the median Tmax for plasma amantadine was around 12 hours (range 6-20 hours). The steady-state concentrations of GOCOVRI were achieved 4 days after the dose initiation. The steady-state total exposures (AUC0-tau) were 20-30% higher than after single dose, suggesting an accumulation ratio of 1.2-1.3.
Effect of Food
A single dose crossover study of GOCOVRI established the lack of effect of high-fat, high-calorie meal on plasma amantadine pharmacokinetics; additionally, administration of entire capsule contents sprinkled on applesauce also did not affect plasma amantadine pharmacokinetics.
Distribution
The volume of distribution determined after the intravenous administration of amantadine to 15 healthy subjects was 3 to 8 L/kg, suggesting potential extravascular distribution. Amantadine is approximately 67% bound to plasma proteins over a concentration range of 0.1 to 2.0 µg/mL.
Elimination
In a study with healthy volunteers after oral administration, the apparent plasma clearance of amantadine was estimated to be 0.27 ± 0.094 L/hr/kg (range 0.13 to 0.57 L/hr/kg). Amantadine is primarily excreted unchanged in the urine, and in a study of six healthy volunteers, the ratio of amantadine renal clearance to apparent plasma clearance was 0.79 ± 0.17 (mean ± SD). The mean plasma amantadine half-life at steady-state was approximately 16 hours.
Metabolism
Eight metabolites of amantadine have been identified in human urine. One metabolite, an N-acetylated compound, was quantified in human urine and accounted for 0-15% of the administered dose in multiple studies. The contribution of this metabolite to efficacy or toxicity is not known.
Excretion
Amantadine is primarily excreted unchanged in the urine by both glomerular filtration and tubular secretion.
Specific Population
Male/Female Patients
In an integrated analysis of five studies in healthy volunteers (n=147), the mean total amantadine clearance following administration of GOCOVRI, adjusted for body weight in kilograms, was 1.2 fold higher in males compared to females (95% CI: 1.1, 1.3, P=0.007). No dose adjustment by gender is warranted.
Renal Impairment
The renal clearance of amantadine is significantly lower in adult patients with moderate or severe renal impairment, compared to healthy adults. Since the renal pathway is a major elimination pathway, impairment in renal function can result in significant accumulation in the plasma, warranting dose adjustment. The impact of renal impairment on dose adjustment was not investigated in a dedicated study.
Based on PK simulations, the range of the total exposures (AUC0-tau) in subjects with normal renal function (creatinine clearance >90 mL/min/1.73 m2) or mild renal impairment (creatinine clearance between 60 and 89 mL/min/1.73 m2) were comparable for the same dosing regimen. However, patients with moderate renal impairment (creatinine clearance between 30 and 59 mL/min/1.73 m2) had higher exposures relative to patients with normal renal function or mild renal impairment. Severe renal impairment (creatinine clearance between 15 and 29 mL/min/1.73 m2) resulted in even higher total exposures. Dosage adjustment is recommended in patients with moderate and severe renal impairment [see Use in Special Populations (8.6) and Dosage and Administration (2.3)]. GOCOVRI is contraindicated in patients with end stage renal disease (creatinine clearance less than 15 mL/min/1.73 m2).
Amantadine is inefficiently removed by hemodialysis.
Drug Interaction Studies
The in-vitro dissolution-release profiles showed 52% drug release after 45 minutes, and up to 95% after 2 hours, at concentrations of 40% alcohol/0.1N HCl.
In vitro studies indicate that amantadine has negligible or no inhibitory activity against cellular transporters (P-gp, BCRP, MATE2-K, OAT1, OAT3, OATP1B1, and OATP1B3) at plasma concentrations observed in patients with Parkinson’s disease on a GOCOVRI 274 mg dose.
In vitro studies in MDCK-II cells demonstrated that amantadine is not a substrate of the anionic transporters OAT1 or OAT3, or the cationic transporter MATE2-K. Amantadine was a poor substrate of the cationic transporters OCT2 and MATE1. Renal elimination of amantadine may be mediated in part by one or more organic cation transporters independent of OCT2. An in vivo study demonstrated that quinidine, a known organic cation transporter inhibitor, reduced amantadine clearance by approximately 33% in humans. The clinical significance is unknown. In vitro studies show that amantadine does not significantly inhibit the enzyme activity of drug metabolizing cytochrome P450 isoforms (CYP1A2, 2B6, 2C19, 2C8, 2C9, 2D6, 2E1, 3A4, and 3A5).
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Animal studies designed to evaluate the carcinogenic potential of amantadine have not been conducted.
Mutagenesis
Amantadine was negative for genotoxicity in in vitro (Ames and mammalian cell [Chinese Hamster ovary and human peripheral blood lymphocytes]) assays in the presence or absence of metabolic activation and in an in vivo mouse bone marrow micronucleus assay.
Impairment of Fertility
The effects of amantadine on fertility have not been adequately tested in a study in animals conducted according to current standards. In a reproduction study reported in the literature, oral administration of amantadine to male and female rats at a dose of 32 mg/kg/day resulted in impaired fertility. The no-effect dose for adverse effects on fertility (10 mg/kg/day) is less than the recommended human dose of 274 mg/day on a mg/m2 basis.
-
14 CLINICAL STUDIES
Overview of Studies
GOCOVRI for the treatment of dyskinesia in patients with Parkinson’s disease was assessed in two randomized, double-blind, placebo-controlled efficacy trials: Study 1 and Study 2. Key inclusion criteria in both studies included at least 1 hour of troublesome dyskinesia time during the day and at least mild functional impact because of dyskinesia.
Study 1 was conducted in 121 (modified Intention to Treat (mITT) population) Parkinson’s disease patients with dyskinesia in the United States (US) and Canada. The duration of treatment in Study 1 was up to 25 weeks. Study 2 was conducted in 75 (mITT population) patients with dyskinesia in the US, Germany, France, Spain, and Austria. The duration of treatment was 13 weeks.
In both studies, the primary efficacy endpoint was the change in total score of the Unified Dyskinesia Rating Scale (UDysRS) between baseline and Week 12. Key secondary endpoints derived from a Parkinson’s disease home diary included changes from baseline to Week 12 in ON time without troublesome dyskinesia and OFF time.
Study Population
In Study 1 and Study 2, the mean age of patients at the time of Parkinson’s disease diagnosis was 55 years (range: 29-75 years).
At baseline, patients had a mean UDysRS total score of 40.1 (range: 8-76), a mean duration of ON time without troublesome dyskinesia (Parkinson’s disease home diary) of 8.4 hours (range: 0-15.3), and a mean duration of OFF time of 2.8 hours (range: 0-9.5).
Patients in Study 1 and Study 2 were treated with a stable dose of levodopa, with 32% of patients on levodopa monotherapy. Patients were also treated with concomitant dopamine agonists (54%) and/or MAO-B inhibitors (44%).
Study 1
In Study 1, a significant decrease in mean UDysRS total score (reduction in dyskinesia) was observed at Week 12 in patients treated with GOCOVRI, compared with placebo (Table 2).
Study 2
In Study 2, a significant decrease in mean UDysRS total score (reduction in dyskinesia) was observed at Week 12 in patients treated with GOCOVRI, compared with placebo (Table 2).
Table 2: UDysRS Total Score at Week 12 for Study 1 and Study 2 mITT Population Study 1 Study 2 Placebo
(N=58)GOCOVRI
274 mg
(N=63)Placebo
(N=38)GOCOVRI
274 mg
(N=37)UDysRS total score,
LS Mean (SE)Change from Baseline -8.0 (1.64) -15.9 (1.62) -6.3 (2.08) -20.7 (2.20) Treatment Difference -7.9 (2.30) -14.4 (3.03) P-value 0.0009 <0.0001 In Study 1 and Study 2, there was a significant increase in ON time without troublesome dyskinesia, and a significant decrease in OFF time between baseline and Week 12 in patients treated with GOCOVRI, compared with placebo (Table 3).
Table 3: PD Home Diary Results at Week 12 for Study 1 and Study 2 mITT Population Study 1 Study 2 Placebo
(N=58)GOCOVRI
274 mg
(N=63)Placebo
(N=38)GOCOVRI
274 mg
(N=37)ON Time w/o
Troublesome
Dyskinesia, LS
Mean (SE)Change from Baseline 0.8 (0.43) 3.6 (0.43) 2.1 (0.53) 4.0 (0.56) Treatment Difference 2.7 (0.61) 1.9 (0.78) P-value <0.0001 0.0168 OFF Time, LS
Mean (SE)Change from Baseline 0.3 (0.26) -0.6 (0.27) 0.6 (0.31) -0.5 (0.34) Treatment Difference -0.9 (0.37) -1.1 (0.46) P-value 0.0171 0.0199 -
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
GOCOVRI is supplied as extended release capsules in the following configurations:
The 68.5 mg capsule is a white opaque size #2 capsule, with black printing of ‘ADAMAS’ on front and ‘85’ on back of the cap and three black bands printed on body of capsule.
- 60 count bottles NDC# 70482-085-60
The 137 mg capsule is a light blue opaque size #0 capsule, with black printing of ‘ADAMAS’ on front and ‘170’ on back of the cap and three black bands printed on body of capsule.
- 60 count bottles NDC# 70482-170-60
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Administration
Instruct patients and caregivers that GOCOVRI capsules should be swallowed whole and can be administered with or without food. Alternatively, GOCOVRI capsules may be opened and the contents sprinkled on applesauce; the entire contents should be consumed immediately without chewing [see Dosage and Administration (2.2)]. Advise patients to speak to their healthcare provider before discontinuation of GOCOVRI.
Falling Asleep During Activities of Daily Living
Advise patients that sleepiness and fatigue that have been reported with GOCOVRI and patients treated with Parkinson’s disease medications have reported falling asleep while engaged in activities of daily living. These adverse reactions may affect some patients’ ability to drive and operate machinery safely [see Warnings and Precautions (5.1)].
Suicidality and Depression
Instruct patients, family members, and caregivers to notify their healthcare provider if depressed mood, depression, changes in behavior or thinking, and suicidal ideation or behavior develop during treatment [see Warnings and Precautions (5.2)].
Hallucinations/Psychotic Behavior
Inform patients and caregivers that hallucinations and paranoia can occur while taking GOCOVRI. Tell patients to report unreal visions, sounds, or sensations or other psychotic behavior to their healthcare provider promptly should they develop [see Warnings and Precautions (5.3)].
Dizziness and Orthostatic Hypotension
An increased incidence of dizziness, orthostatic hypotension, and syncope was observed with administration of GOCOVRI. Caution patients against standing rapidly after sitting or lying down, especially if they have been doing so for prolonged periods and especially at the initiation of treatment with GOCOVRI [see Warnings and Precautions (5.4)].
Withdrawal-Emergent Hyperpyrexia and Confusion
Advise patients to contact their healthcare provider before stopping GOCOVRI. Tell patients to inform their healthcare provider if they develop withdrawal symptoms such as fever, confusion, or severe muscle stiffness [see Warnings and Precautions (5.5)].
Impulse Control/Compulsive Disorders
Inform patients of the potential for experiencing intense urges to gamble, increased sexual urges, intense urges to spend money, binge eating, and other intense urges and the inability to control these urges while taking one or more of the medications that increase central dopaminergic tone, that are generally used for the treatment of Parkinson’s disease [see Warnings and Precautions (5.6)].
Drug Interactions
Certain medications can cause an interaction with GOCOVRI. Advise patients and/or caregivers to inform their healthcare provider of all the medicines the patient is taking, including over-the-counter medicines, dietary supplements, and herbal products. Inform patients that live influenza vaccines and consumption of alcohol are not recommended during treatment with GOCOVRI [see Drug Interactions (7.1, 7.2, 7.3, 7.4)].
Manufactured for:
Adamas Pharma, LLC
Emeryville, CA 94608 -
PATIENT PACKAGE INSERT
PATIENT INFORMATION
GOCOVRI (goh-KUV-ree)
(amantadine) extended release capsules, for oral useWhat is GOCOVRI?
GOCOVRI is a prescription medicine used for the treatment of:
- dyskinesia (sudden uncontrolled movements) in people with Parkinson’s disease who are treated with levodopa therapy or levodopa therapy with other medicines that increase the effects of dopamine in the brain.
It is not known if GOCOVRI is safe and effective in children.
Do not take GOCOVRI if you have severe kidney problems
Before you take GOCOVRI, tell your doctor about all of your medical conditions, including if you:
- have kidney problems.
- have daytime sleepiness from a sleep disorder, have unexpected or unpredictable sleepiness or periods of sleep, take a medicine to help you sleep, or take any medicine that makes you drowsy.
- have mental problems, such as suicidal thoughts, depression, or hallucinations.
- have unusual urges including gambling, increased sex drive, compulsive eating, or compulsive shopping.
- drink alcoholic beverages. This may increase your chances of becoming drowsy or sleepy while taking GOCOVRI.
- are pregnant or plan to become pregnant. GOCOVRI may harm your unborn baby.
- are breastfeeding or plan to breastfeed. GOCOVRI can pass into your breastmilk. Talk to your doctor about the best way to feed your baby if you take GOCOVRI.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Especially tell your doctor if you:
- take medicines like sodium bicarbonate.
- have had or are planning to have a live flu (influenza) vaccination (nasal spray). You can receive the influenza vaccination shot but should not get a live influenza vaccine while taking GOCOVRI.
Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine.
How should I take GOCOVRI?
- Take GOCOVRI as exactly as your doctor tells you to.
- Start GOCOVRI with 1 capsule at bedtime. Your doctor may change your dose if needed. If your doctor tells you to take 2 capsules, take them together at bedtime.
- Do not stop or change GOCOVRI before talking with your doctor. Call your doctor if you have symptoms of withdrawal such as fever, confusion, or severe muscle stiffness.
- GOCOVRI may be taken with food or without food.
- Swallow GOCOVRI capsules whole. Do not crush, chew, or divide.
- GOCOVRI capsules may be opened and sprinkled on applesauce before swallowing. Sprinkle all of the medicine in the capsule on the applesauce. Take all of the medicine right away without chewing.
- If you miss a dose of GOCOVRI, do not take an extra dose. Take your usual dose of GOCOVRI on the next day at bedtime.
- If you have forgotten to take GOCOVRI for several days, contact your doctor.
- Do not drink alcohol with your dose of GOCOVRI.
- Do not use GOCOVRI capsules that are damaged or show signs of tampering.
- If you take too much GOCOVRI, call your doctor or go to the nearest hospital emergency room right away.
What should I avoid while taking GOCOVRI?
- Do not drive, operate machinery, or do other dangerous activities until you know how GOCOVRI affects you.
- Do not drink alcohol while taking GOCOVRI. It can increase your chances of getting serious side effects.
What are the possible side effects of GOCOVRI?
GOCOVRI may cause serious side effects, including:
- falling asleep during normal activities. You may fall asleep while doing normal activities such as driving a car, talking, or eating while taking GOCOVRI or other medicines that treat Parkinson’s disease. You may fall asleep without being drowsy or without warning. This may result in having accidents. Your chances of falling asleep while doing normal activities while taking GOCOVRI are greater if you take other medicines that cause drowsiness. Tell your doctor right away if this happens.
- suicidal thoughts or actions and depression. Some people taking GOCOVRI have had suicidal thoughts, attempted suicide, or depression. Tell your doctor if you have new or sudden changes in mood, behaviors, thoughts, or feelings, including thoughts about hurting yourself or ending your life.
- hallucinations. GOCOVRI can cause or worsen hallucinations (seeing or hearing things that are not real) or psychotic behavior. Contact your doctor if you have hallucinations.
- feeling dizzy, faint or light headed, especially when you stand up (orthostatic hypotension). Light headedness or fainting may happen when getting up too quickly from a sitting or lying position especially after long periods of time, when first starting GOCOVRI, or if your dose has been increased. Contact your doctor if you become light headed or faint when standing up.
- unusual urges. Some people taking GOCOVRI get urges to behave in a way unusual for them. Examples of this are an unusual urge to gamble, increased sexual urges, strong urges to spend money, binge eating and the inability to control these urges. If you notice or your family notices that you are developing any unusual behaviors, talk to your doctor.
The most common side effects of GOCOVRI include dry mouth, swelling of legs and feet, constipation, and falls.
These are not all the possible side effects of GOCOVRI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store GOCOVRI?
- Store GOCOVRI at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep GOCOVRI out of the reach of children.
General information about the safe and effective use of GOCOVRI.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use GOCOVRI for a condition for which it was not prescribed. Do not give GOCOVRI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or doctor for information about GOCOVRI that is written for health professionals.
What are the ingredients in GOCOVRI?
Active ingredient: amantadine hydrochloride
Inactive ingredients: copovidone, ethylcellulose, hypromellose, magnesium stearate, medium chain triglycerides, microcrystalline cellulose, povidone, and talc
Manufactured for: Adamas Pharma LLC. Emeryville, CA 94608
For more information, go to www.GOCOVRI.com or call 1-833-223-2627.
This Patient Information has been approved by the U.S. Food and Drug Administration Issued:3/2020
- PRINCIPAL DISPLAY PANEL - NDC: 70482-085-60 - 68.5mg 60-count Bottle
- PRINCIPAL DISPLAY PANEL - NDC: 70482-170-60 - 135mg 60-count Bottle
-
INGREDIENTS AND APPEARANCE
GOCOVRI
amantadine capsule, coated pelletsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 70482-085 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AMANTADINE (UNII: BF4C9Z1J53) (AMANTADINE - UNII:BF4C9Z1J53) AMANTADINE 68.5 mg Inactive Ingredients Ingredient Name Strength COPOVIDONE K25-31 (UNII: D9C330MD8B) 5.66 mg ETHYLCELLULOSE (7 MPA.S) (UNII: H3UP11403C) 31.06 mg HYPROMELLOSE 2910 (3 MPA.S) (UNII: 0VUT3PMY82) 12.89 mg MAGNESIUM STEARATE (UNII: 70097M6I30) 0.20 mg MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) 3.75 mg MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) 24.29 mg POVIDONE K90 (UNII: RDH86HJV5Z) 4.20 mg TALC (UNII: 7SEV7J4R1U) 5.54 mg Product Characteristics Color WHITE (Cap and Body) , BLACK (Imprint) Score no score Shape CAPSULE Size 18mm Flavor Imprint Code ADAMAS;85;Three;black;bands Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 70482-085-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 08/24/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208944 08/24/2017 GOCOVRI
amantadine capsule, coated pelletsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 70482-170 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AMANTADINE (UNII: BF4C9Z1J53) (AMANTADINE - UNII:BF4C9Z1J53) AMANTADINE 137 mg Inactive Ingredients Ingredient Name Strength COPOVIDONE K25-31 (UNII: D9C330MD8B) 11.32 mg ETHYLCELLULOSE (7 MPA.S) (UNII: H3UP11403C) 62.13 mg HYPROMELLOSE 2910 (3 MPA.S) (UNII: 0VUT3PMY82) 31.06 mg MAGNESIUM STEARATE (UNII: 70097M6I30) 0.39 mg MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) 7.50 mg MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) 48.57 mg POVIDONE K90 (UNII: RDH86HJV5Z) 8.40 mg TALC (UNII: 7SEV7J4R1U) 11.07 mg Product Characteristics Color BLUE (Cap and Body) , BLACK (Imprint) Score no score Shape CAPSULE Size 22mm Flavor Imprint Code ADAMAS;170;Three;black;bands Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 70482-170-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 08/24/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208944 08/24/2017 Labeler - Adamas Pharma, LLC (192255110) Establishment Name Address ID/FEI Business Operations Catalent Pharma Solutions (Winchester, KY) 829672745 MANUFACTURE(70482-085, 70482-170)
Trademark Results [GOCOVRI]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 GOCOVRI 86698587 5649541 Live/Registered |
Adamas Pharmaceuticals, Inc. 2015-07-20 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

