Glycopyrrolate Injection, USP Rx onlyNOT FOR USE IN NEONATES CONTAINS BENZYL ALCOHOL
Glycopyrrolate by
Drug Labeling and Warnings
Glycopyrrolate by is a Prescription medication manufactured, distributed, or labeled by Zydus Lifesciences Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
GLYCOPYRROLATE- glycopyrrolate injection
Zydus Lifesciences Limited
----------
Glycopyrrolate Injection, USP
Rx only
NOT FOR USE IN NEONATES
CONTAINS BENZYL ALCOHOL
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
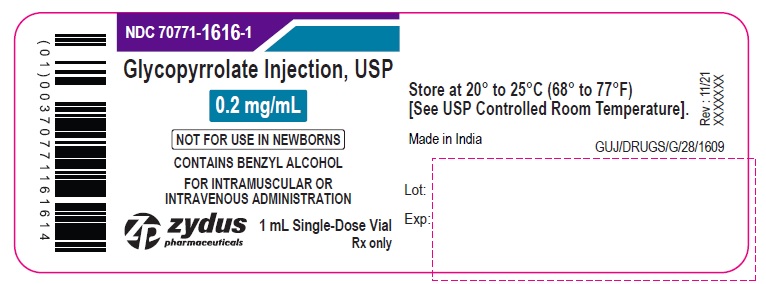
NDC: 70771-1616-1
Glycopyrrolate Injection, USP
0.2 mg/mL
NOT FOR USE IN NEWBORNS
CONTAINS BENZYL ALCOHOL
FOR INTRAMUSCULAR OR
INTRAVENOUS ADMINISTRATION
1 mL Single-Dose Vial
Rx only
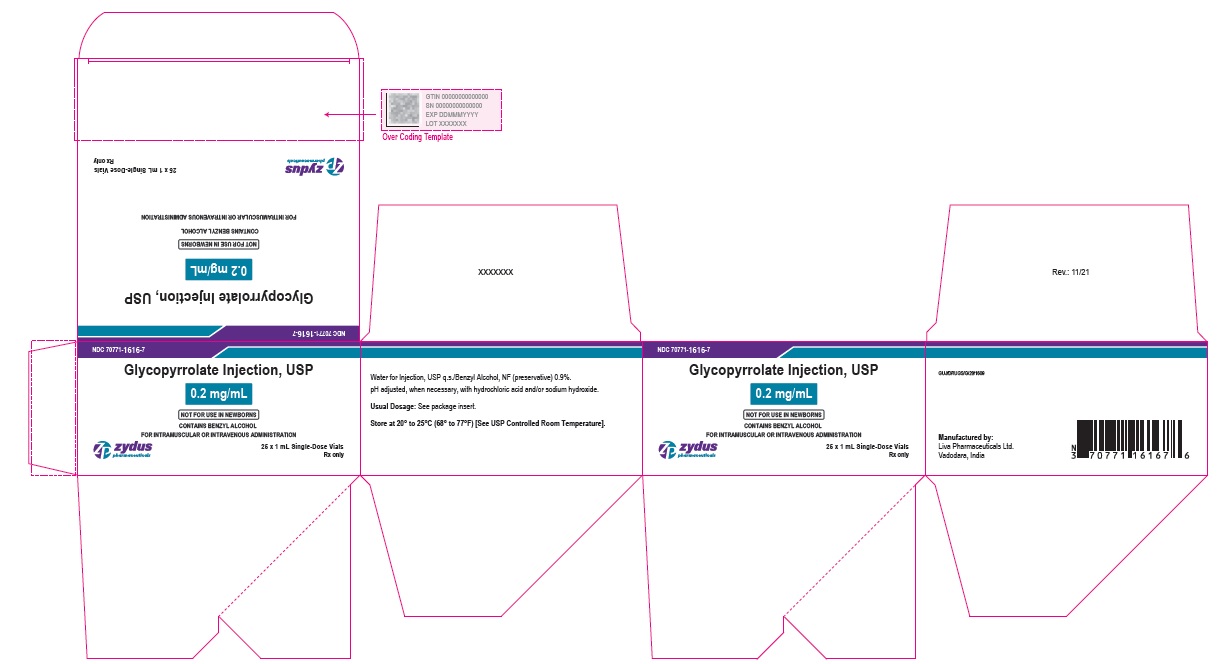
NDC: 70771-1616-7
Glycopyrrolate Injection, USP
0.2 mg/mL
NOT FOR USE IN NEWBORNS
CONTAINS BENZYL ALCOHOL
FOR INTRAMUSCULAR OR INTRAVENOUS ADMINISTRATION
25 x 1 mL Single-Dose Vials
Rx only
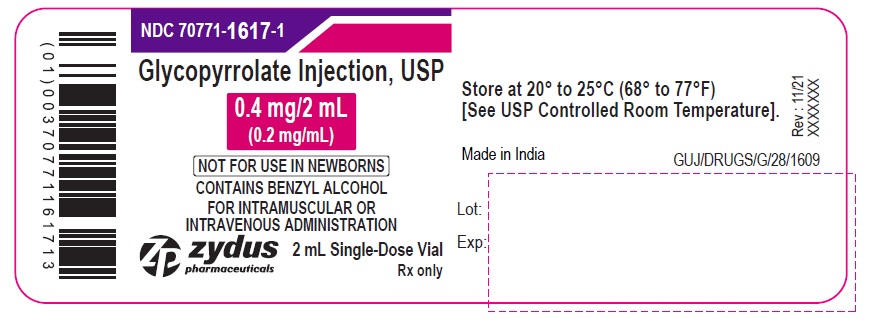
NDC: 70771-1617-1
Glycopyrrolate Injection, USP
0.4 mg/2 mL
(0.2 mg/mL)
NOT FOR USE IN NEWBORNS
CONTAINS BENZYL ALCOHOL
FOR INTRAMUSCULAR OR
INTRAVENOUS ADMINISTRATION
2 mL Single-Dose Vial
Rx only
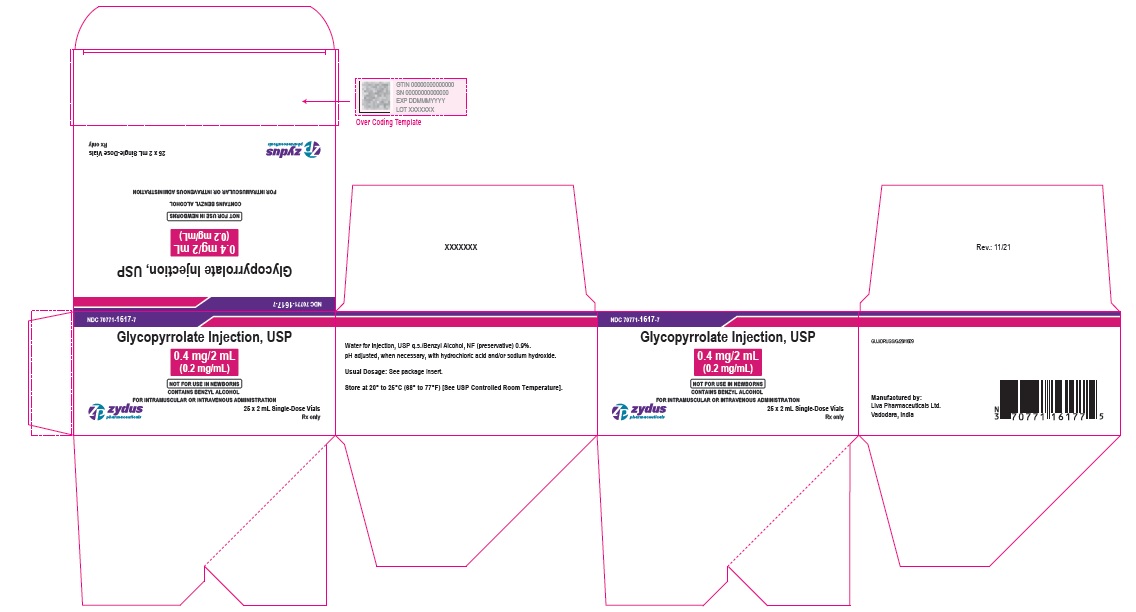
NDC: 70771-1617-7
Glycopyrrolate Injection, USP
0.4 mg/2 mL
(0.2 mg/mL)
NOT FOR USE IN NEWBORNS
CONTAINS BENZYL ALCOHOL
FOR INTRAMUSCULAR OR INTRAVENOUS ADMINISTRATION
25 x 2 mL Single-Dose Vials
Rx only
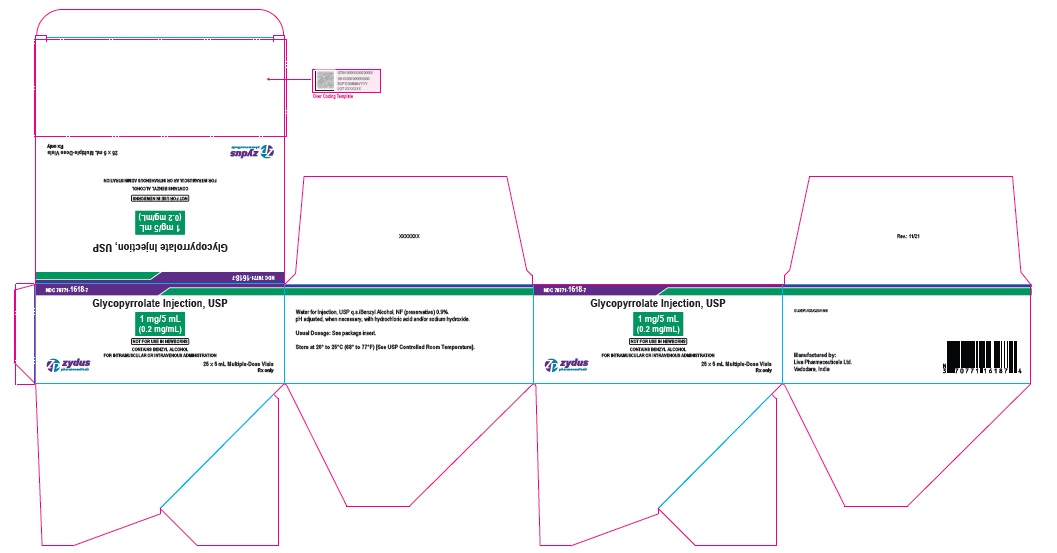
NDC: 70771-1618-1
Glycopyrrolate Injection, USP
1 mg/5 mL
(0.2 mg/mL)
NOT FOR USE IN NEWBORNS
CONTAINS BENZYL ALCOHOL
FOR INTRAMUSCULAR OR
INTRAVENOUS ADMINISTRATION
5 mL Multiple-Dose Vial
Rx only

NDC: 70771-1618-7
Glycopyrrolate Injection, USP
1 mg/5 mL
(0.2 mg/mL)
NOT FOR USE IN NEWBORNS
CONTAINS BENZYL ALCOHOL
FOR INTRAMUSCULAR OR INTRAVENOUS ADMINISTRATION
25 x 5 mL Multiple-Dose Vials
Rx only
NDC: 70771-1619-1
Glycopyrrolate Injection, USP
4 mg/20 mL
(0.2 mg/mL)
NOT FOR USE IN NEWBORNS
CONTAINS BENZYL ALCOHOL
FOR INTRAMUSCULAR OR
INTRAVENOUS ADMINISTRATION
20 mL Multiple-Dose Vial
Rx only

NDC: 70771-1619-6
Glycopyrrolate Injection, USP
4 mg/20 mL
(0.2 mg/mL)
NOT FOR USE IN NEWBORNS
CONTAINS BENZYL ALCOHOL
FOR INTRAMUSCULAR OR INTRAVENOUS ADMINISTRATION
10 x 20 mL Multiple-Dose Vials
Rx only

| GLYCOPYRROLATE
glycopyrrolate injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| GLYCOPYRROLATE
glycopyrrolate injection |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| GLYCOPYRROLATE
glycopyrrolate injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| GLYCOPYRROLATE
glycopyrrolate injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Zydus Lifesciences Limited (918596198) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Zydus Lifesciences Limited | 873671928 | MANUFACTURE(70771-1616, 70771-1617, 70771-1618, 70771-1619) , ANALYSIS(70771-1616, 70771-1617, 70771-1618, 70771-1619) | |