ELIZABETH HEALTH STYLE ICE COLD by AYUSHREE PHARMACEUTICALS PRIVATE LIMITED
ELIZABETH HEALTH STYLE ICE COLD by
Drug Labeling and Warnings
ELIZABETH HEALTH STYLE ICE COLD by is a Otc medication manufactured, distributed, or labeled by AYUSHREE PHARMACEUTICALS PRIVATE LIMITED. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
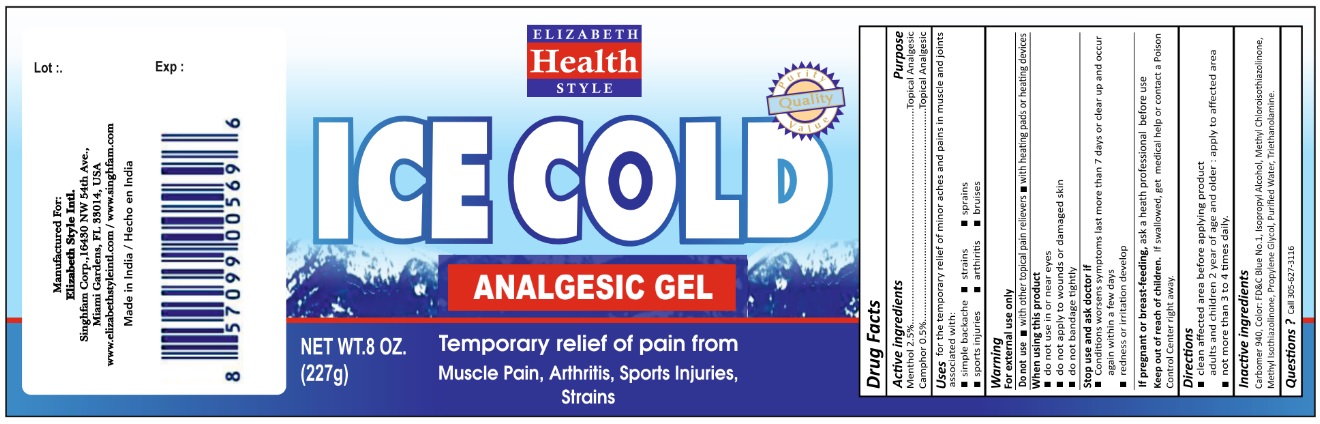
ELIZABETH HEALTH STYLE ICE COLD- camphor (synthetic), levomenthol gel
AYUSHREE PHARMACEUTICALS PRIVATE LIMITED
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Active Ingredient Purpose
Menthol 2.50% .......................... Topical analgesic
Camphor 0.5 % ......................... Topical analgesic
Use
for the temporary relief of minor aches and pains in muscle and joints associated with:
simple backache strains sprains
sports injuries arthritis bruises
Warnings
For external use only
Do not use with other topical pain relievers with heating pads or heating devices.
When using this product
do not use in or near eyes
do not apply to wounds or damaged skin
do not bandage tightly
When using this product
- do not use in or near eyes
- do not apply to wounds or damaged skin
- do not bandage tightly
Stop use and ask doctor if
- Conditions worsens symptoms last more than 7 days or clear up and occur again within a few days
- redness or irritation develop
If pregnant or breast-feeding, ask a heath professional before use
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- clean affected area before applying product
adults and children 2 year of age and older: apply to affected area
- not more than 3 to 4 times daily.
| ELIZABETH HEALTH STYLE ICE COLD
camphor (synthetic), levomenthol gel |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - AYUSHREE PHARMACEUTICALS PRIVATE LIMITED (861375952) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| AYUSHREE PHARMACEUTICALS PRIVATE LIMITED | 861375952 | manufacture(80591-002) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.