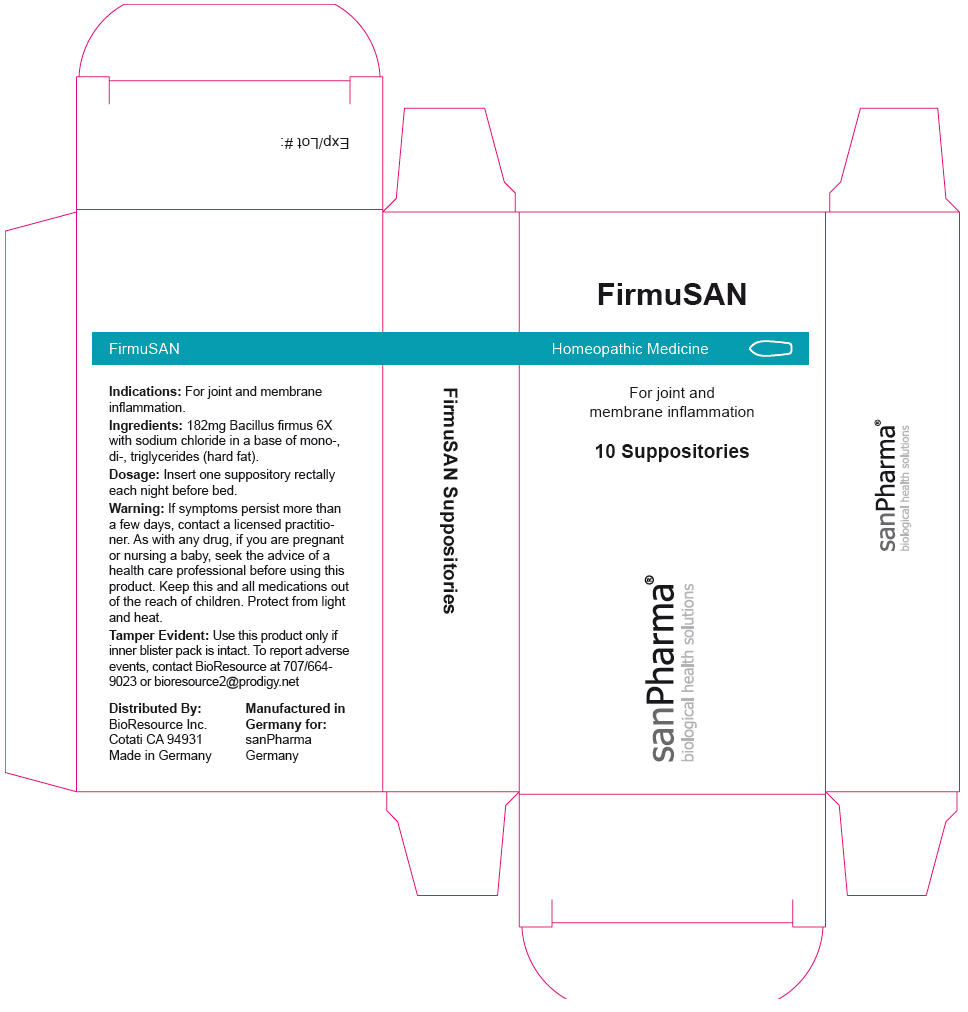
FirmuSAN by sanPharma GmbH FirmuSAN
FirmuSAN by
Drug Labeling and Warnings
FirmuSAN by is a Homeopathic medication manufactured, distributed, or labeled by sanPharma GmbH. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
FIRMUSAN- bacillus firmus suppository
sanPharma GmbH
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
FirmuSAN
Warning
If symptoms persist more than a few days, contact a licensed practitioner. As with any drug, if you are pregnant or nursing a baby, seek the advice of a health care professional before using this product.
| FIRMUSAN
bacillus firmus suppository |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - sanPharma GmbH (341409153) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| sanPharma GmbH | 341409153 | manufacture(64232-105) , label(64232-105) | |
Revised: 12/2019
Document Id: 99584552-2a26-4c4d-e053-2a95a90a288a
Set id: 854e4064-cbb2-49db-8f02-bd75cb94d9e6
Version: 4
Effective Time: 20191210
sanPharma GmbH