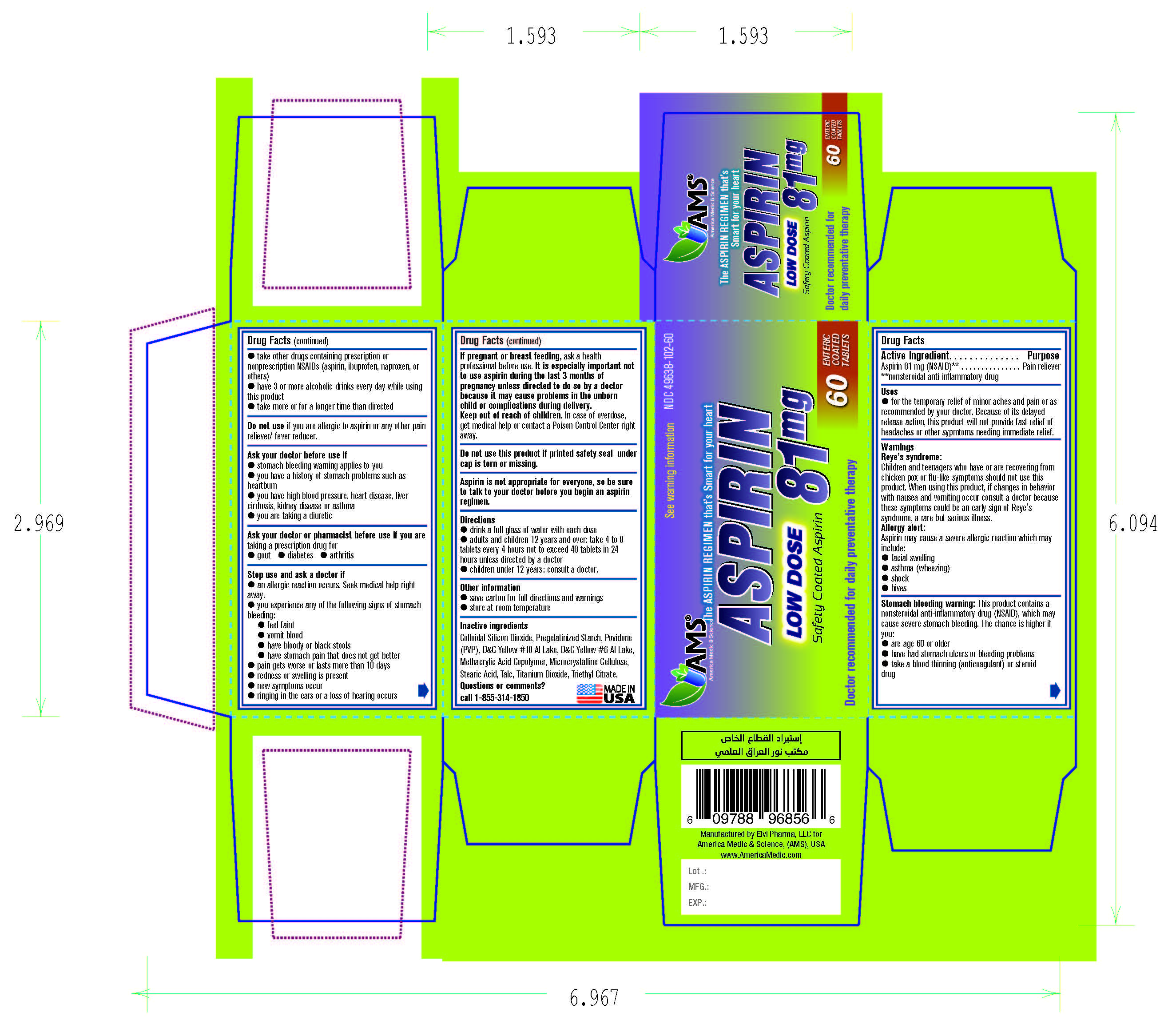
Aspirin Delayed Release Tablets USP, 81 MG Low Dose Safety Coated Aspirin
Aspirin by
Drug Labeling and Warnings
Aspirin by is a Otc medication manufactured, distributed, or labeled by America Medic & Science, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
ASPIRIN- aspirin tablet, delayed release
America Medic & Science, LLC
----------
Aspirin Delayed Release Tablets USP, 81 MG
Low Dose
Safety Coated Aspirin
Uses
- for the temporary relief of minor aches and pains or as recommended by your doctor. Because of its delayed release action, this product will not provide fast relief of headaches or other sypmtoms needing immediate relief.
Warnings
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include:
- facial swelling
- asthma (wheezing)
- shock
- hives
Stomach bleeding warning: This product contains a nonsteroidal anti-inflammatory drug (NSAID), which may cause severe stomach bleeding. The chance is higher if you:
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Ask your doctor before use
- stomach bleeding warning applies to you
- you have a history of stomach problems such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, kidney disease or asthma
- you are taking a diuretic
Ask your doctor or pharmacist before use if you are taking a prescription drug for
- gout
- diabetes
- arthritis
Stop use and ask doctor if
an allergic reaction occurs. Seek medical help right away.
you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- pain gets worse or lasts more than 10 days
- redness or swelling is present
- new symptoms occur
- ringing in the ears or a loss of hearing occurs
If pregnant or breast feeding, ask a health professional before use. It is especially important not to use aspirin during the last 3 months of pregnancy unless directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Aspirin is not appropriate for everyone, so be sure to talk to your doctor before you begin an aspirin regimen.
Directions
- drink a full glass of water with each dose
- adults and children 12 years and over: take 4 to 8 tablets every 4 hours not to exceed 48 tablets in 24 hours unless directed by a doctor
- children under 12 years: consult a doctor.
| ASPIRIN
aspirin tablet, delayed release |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - America Medic & Science, LLC (071065464) |
| Registrant - America Medic & Science, LLC (071065464) |
Trademark Results [Aspirin]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ASPIRIN 76481358 2788617 Dead/Cancelled |
Simon Carter Accessories, Ltd. 2003-01-10 |
 ASPIRIN 75209895 not registered Dead/Abandoned |
Bayer Aktiengesellschaft 1996-12-09 |
 ASPIRIN 73234351 1171777 Dead/Cancelled |
McIntyre; William A. 1979-10-09 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.