EXTRA STRENGTH STOPAIN COLD PAIN RELIEVING LIQUID ROLL ON
EXTRA STRENGTH STOPAIN COLD PAIN RELIEVING ROLL ON by
Drug Labeling and Warnings
EXTRA STRENGTH STOPAIN COLD PAIN RELIEVING ROLL ON by is a Otc medication manufactured, distributed, or labeled by TROY MANUFACTURING, INC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
EXTRA STRENGTH STOPAIN COLD PAIN RELIEVING ROLL ON- menthol liquid
TROY MANUFACTURING, INC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
EXTRA STRENGTH STOPAIN COLD PAIN RELIEVING LIQUID ROLL ON
Uses: For the temporary relief of minor aches and pains of muscle and joints associated with - simple backache -arthritis - strains - bruises -sprains
When using this product
use only as directed
do not bandage tightly or use a heating pad
do not apply to wounds or damaged skin.
Stop use and ask a doctor if
condition worsens or symptoms persist for more than 7 days or clear up and occur again within a few days.
redness is present
excessive irritation of the skin develops.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
Adults and children over 12 years: spray directly onto affected area without the need to rub, massage or bandage
repeat as necessary, but do not apply more than 4 times daily.
Children 12 years or younger: ask a doctor.
Inactive Ingredients
Boswellia serrata extract, carbomer, dimethylsulfone (MSM), eucalyptus oil, glucosamine sulfate, glycerin, peppermint oil, SD alcohol 39-C, triethanolamine, water
Manufactured for: Troy Healthcare, LLC 130 Lions Drive, Hazleton, PA 18202
800-201-PAIN (7246)
www.stopain.com
NOTHING LASTS LONGER
EXTRA STRENGTH
STOPAIN COLD
NO-DRIP, NO-STAIN
PAIN RELIVING LIQUID ROLL-ON
Immediate, Penetrating Pain Relief From
Arthritis
Muscle Aches
Back Pain
3fl oz (88 ml)
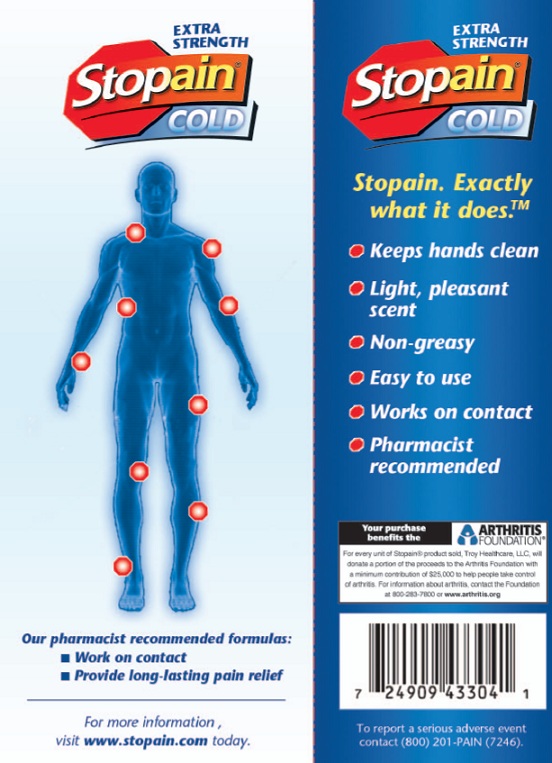
Stopain. Exactly what it does. tm
Keeps hands clean
Light, pleasant scent
Non Greasy
Easy to use
Works on contact
Pharmacist recommended
Your purchase benefits the Arthritis Foundation
For every unit of Stopain(r) product sold, Troy Healthcare, LLC will donate a portion of the proceeds to the ARthrits Foundation with a minimum contribution of $25,000 to help people take control of arthritis. For information about arthritis, contact the Foundation at 800-283-7800 or www.arthritis.org.
Our pharmacist recommended formulas:
Work on contact
Provide long-lasting pain relief
For more information, visit www.stopain.com today
made in USA
To report a serious adverse event, contact (800) 201 PAIN (7246)
| EXTRA STRENGTH STOPAIN COLD PAIN RELIEVING ROLL ON
menthol liquid |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - TROY MANUFACTURING, INC (160075248) |
| Registrant - TROY MANUFACTURING, INC (160075248) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| TROY MANUFACTURING, INC | 160075248 | manufacture(63936-8523) | |