STRIBILD- elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate tablet, film coated
Stribild by
Drug Labeling and Warnings
Stribild by is a Prescription medication manufactured, distributed, or labeled by Avera McKennan Hospital. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use STRIBILD safely and effectively. See full prescribing information for STRIBILD.
STRIBILD® (elvitegravir, cobicistat, emtricitabine, tenofovir disoproxil fumarate) tablets, for oral use
Initial U.S. Approval: 2012WARNING: LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS and POSTTREATMENT ACUTE EXACERBATION OF HEPATITIS B
See full prescribing information for complete boxed warning.
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs, including tenofovir disoproxil fumarate (DF), a component of STRIBILD. (5.1)
STRIBILD is not approved for the treatment of chronic hepatitis B virus (HBV) infection. Severe acute exacerbations of hepatitis B have been reported in patients coinfected with HIV-1 and HBV who have discontinued EMTRIVA or VIREAD, two of the components of STRIBILD. Hepatic function should be monitored closely in these patients. If appropriate, initiation of anti-hepatitis B therapy may be warranted. (5.2)
RECENT MAJOR CHANGES
Indications and Usage (1) 01/2017 Dosage and Administration, Testing Prior to Initiation and During Treatment with STRIBILD (2.1) 01/2017 Dosage and Administration, Recommended Dosage (2.2) 01/2017 Dosage and Administration, Dosage Adjustment in Patients with Renal Impairment (2.3) 01/2017 Contraindications (4) 09/2016 Warnings and Precautions, New Onset or Worsening Renal Impairment (5.3) 01/2017 Warnings and Precautions, Avoid Use with Other Antiretroviral Products (5.4) Removed 01/2017 Warnings and Precautions, Bone Loss and Mineralization Defects (5.5) 01/2017 INDICATIONS AND USAGE
STRIBILD is a four-drug combination of elvitegravir, an HIV integrase strand transfer inhibitor (HIV-1 INSTI), cobicistat, a CYP3A inhibitor, and emtricitabine and tenofovir DF, both HIV nucleoside analog reverse transcriptase inhibitors (HIV NRTI) and is indicated as a complete regimen for the treatment of HIV-1 infection in adults and pediatric patients 12 years of age and older weighing at least 35 kg who have no antiretroviral treatment history or to replace the current antiretroviral regimen in those who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable antiretroviral regimen for at least 6 months with no history of treatment failure and no known substitutions associated with resistance to the individual components of STRIBILD. (1, 14)
DOSAGE AND ADMINISTRATION
- Testing: Prior to initiation of STRIBILD, patients should be tested for hepatitis B virus infection. Assess serum creatinine, serum phosphorous, estimated serum creatinine clearance, urine glucose, and urine protein before initiating STRIBILD and during therapy in all patients as clinically appropriate. (2.1)
- Recommended dosage: One tablet taken once daily with food. (2.2)
- Dosage in renal impairment: Initiation of STRIBILD in patients with estimated creatinine clearance below 70 mL per minute is not recommended. Discontinue in patients with estimated creatinine clearance below 50 mL per minute. (2.3)
DOSAGE FORMS AND STRENGTHS
Tablets: 150 mg of elvitegravir, 150 mg of cobicistat, 200 mg of emtricitabine, and 300 mg of tenofovir disoproxil fumarate. (3)
CONTRAINDICATIONS
Coadministration of STRIBILD is contraindicated with drugs that:
WARNINGS AND PRECAUTIONS
- New onset or worsening renal impairment: Can include acute renal failure and Fanconi syndrome. Assessment of serum creatinine, serum phosphorous, estimated creatinine clearance, urine glucose, and urine protein is recommended before initiating STRIBILD therapy and during therapy as clinically appropriate. Avoid administering STRIBILD with concurrent or recent use of nephrotoxic drugs. (5.3)
- Risk of adverse reactions or loss of virologic response due to drug interactions: The concomitant use of STRIBILD and other drugs may result in known or potentially significant drug interactions, some of which may lead to loss of therapeutic effect of STRIBILD and possible development of resistance; and possible clinically significant adverse reactions from greater exposures of concomitant drugs. (5.4)
- Decreases in bone mineral density (BMD): Consider monitoring BMD in patients with a history of pathologic fracture or other risk factors of osteoporosis or bone loss. (5.5)
- Redistribution/accumulation of body fat: Observed in patients receiving antiretroviral therapy. (5.6)
- Immune reconstitution syndrome: May necessitate further evaluation and treatment. (5.7)
ADVERSE REACTIONS
Most common adverse drug reactions to STRIBILD (incidence greater than or equal to 10%, all grades) are nausea and diarrhea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Gilead Sciences, Inc. at 1-800-GILEAD-5 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- STRIBILD is a complete regimen for the treatment of HIV-1 infection; therefore, STRIBILD should not be administered with other antiretroviral medications for treatment of HIV-1 infection. (7.1)
- STRIBILD can alter the concentration of drugs metabolized by CYP3A or CYP2D6. Drugs that induce CYP3A can alter the concentrations of one or more components of STRIBILD. Consult the full prescribing information prior to and during treatment for potential drug-drug interactions. (4, 7.2, 7.3, 12.3)
USE IN SPECIFIC POPULATIONS
- Pregnancy: Use during pregnancy only if the potential benefit justifies the potential risk. (8.1)
- Lactation: Women infected with HIV should be instructed not to breastfeed due to the potential for HIV transmission. (8.2)
- Pediatrics: Not recommended for patients less than 12 years of age or weighing less than 35 kg. (8.4)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2017
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS and POSTTREATMENT ACUTE EXACERBATION OF HEPATITIS B
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Testing Prior to Initiation and During Treatment with STRIBILD
2.2 Recommended Dosage
2.3 Dosage Adjustment in Patients with Renal Impairment
2.4 Dosage in Patients with Hepatic Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Lactic Acidosis/Severe Hepatomegaly with Steatosis
5.2 Severe Acute Exacerbation of Hepatitis B in Patients Coinfected with HIV-1 and HBV
5.3 New Onset or Worsening Renal Impairment
5.4 Risk of Adverse Reactions or Loss of Virologic Response Due to Drug Interactions
5.5 Bone Loss and Mineralization Defects
5.6 Fat Redistribution
5.7 Immune Reconstitution Syndrome
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Not Recommended with Other Antiretroviral Medications
7.2 Potential for STRIBILD to Affect Other Drugs
7.3 Potential for Other Drugs to Affect One or More Components of STRIBILD
7.4 Drugs Affecting Renal Function
7.5 Established and Other Potentially Significant Interactions
7.6 Drugs without Clinically Significant Interactions with STRIBILD
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Description of Clinical Trials
14.2 Clinical Trial Results in HIV-1-Infected Adult Subjects with No Antiretroviral Treatment History
14.3 Clinical Trial Results in Virologically Suppressed HIV-1-Infected Adult Subjects with No History of Virologic Failure
14.4 Clinical Trial Results in HIV-1Treatment-Naïve Adolescent Subjects Aged 12 to Less than 18 Years
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS and POSTTREATMENT ACUTE EXACERBATION OF HEPATITIS B
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs, including tenofovir disoproxil fumarate, a component of STRIBILD, in combination with other antiretrovirals [see Warnings and Precautions (5.1)].
STRIBILD is not approved for the treatment of chronic hepatitis B virus (HBV) infection, and the safety and efficacy of STRIBILD have not been established in patients coinfected with HBV and human immunodeficiency virus-1 (HIV-1). Severe acute exacerbations of hepatitis B have been reported in patients who are coinfected with HBV and HIV-1 and have discontinued EMTRIVA or VIREAD, which are components of STRIBILD. Hepatic function should be monitored closely, with both clinical and laboratory follow-up for at least several months in patients who are coinfected with HIV-1 and HBV and discontinue STRIBILD. If appropriate, initiation of anti-hepatitis B therapy may be warranted [see Warnings and Precautions (5.2)].
-
1 INDICATIONS AND USAGE
STRIBILD® is indicated as a complete regimen for the treatment of HIV-1 infection in adults and pediatric patients 12 years of age and older weighing at least 35 kg who have no antiretroviral treatment history or to replace the current antiretroviral regimen in those who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable antiretroviral regimen for at least 6 months with no history of treatment failure and no known substitutions associated with resistance to the individual components of STRIBILD [see Clinical Studies (14)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Testing Prior to Initiation and During Treatment with STRIBILD
Prior to initiation of STRIBILD, patients should be tested for hepatitis B virus infection [see Warnings and Precautions (5.2)].
It is recommended that serum creatinine, serum phosphorous, estimated creatinine clearance, urine glucose, and urine protein should be assessed before initiating STRIBILD and during therapy in all patients as clinically appropriate [see Warnings and Precautions (5.3)].
2.2 Recommended Dosage
STRIBILD is a four-drug fixed dose combination product containing 150 mg of elvitegravir, 150 of cobicistat, 200 mg of emtricitabine, and 300 mg of tenofovir DF. The recommended dosage of STRIBILD is one tablet taken orally once daily with food in adults and pediatric patients 12 years of age and older with a body weight at least 35 kg (at least 77 lbs) and creatinine clearance greater than or equal to 70 mL per minute [see Clinical Pharmacology (12.3)].
2.3 Dosage Adjustment in Patients with Renal Impairment
Initiation of STRIBILD in patients with estimated creatinine clearance below 70 mL per minute is not recommended. Because STRIBILD is a fixed-dose combination tablet, STRIBILD should be discontinued if estimated creatinine clearance declines below 50 mL per minute during treatment with STRIBILD, as the dose interval adjustment required for emtricitabine and tenofovir disoproxil fumarate (DF) cannot be achieved [see Warnings and Precautions (5.3), Adverse Reactions (6.1), Use in Specific Populations (8.6), Clinical Pharmacology (12.3), and Clinical Studies (14)].
No data are available to make dose recommendations for pediatric patients with renal impairment.
2.4 Dosage in Patients with Hepatic Impairment
STRIBILD is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
-
3 DOSAGE FORMS AND STRENGTHS
Each STRIBILD tablet contains 150 mg of elvitegravir, 150 mg of cobicistat, 200 mg of emtricitabine, and 300 mg of tenofovir DF (equivalent to 245 mg of tenofovir disoproxil).
The tablets are green, capsule shaped, film coated, and debossed with "GSI" on one side and the number "1" surrounded by a square box (
 ) on the other side.
) on the other side. -
4 CONTRAINDICATIONS
Coadministration of STRIBILD is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events. These drugs and other contraindicated drugs (which may lead to reduced efficacy of STRIBILD and possible resistance) are listed in Table 1 [see Drug Interactions (7.5) and Clinical Pharmacology (12.3)].
Table 1 Drugs that are Contraindicated with STRIBILD Drug Class Drugs within Class that are Contraindicated with STRIBILD Clinical Comment - * See Drug Interactions (7), Table 6, for sildenafil when dosed as Viagra for erectile dysfunction.
- † See Drug Interactions (7), Table 6, for parenterally administered midazolam.
Alpha 1-Adrenoreceptor Antagonist Alfuzosin Potential for increased alfuzosin concentrations, which can result in hypotension. Anticonvulsants Carbamazepine
Phenobarbital
PhenytoinPotential for decreased cobicistat and elvitegravir plasma concentrations, which may result in loss of therapeutic effect and development of resistance. Antimycobacterial Rifampin Rifampin is a potent inducer of CYP450 metabolism and may cause significant decrease in the plasma concentration of elvitegravir and cobicistat. This may result in loss of therapeutic effect to STRIBILD. Antipsychotic Lurasidone Potential for serious and/or life-threatening reactions. Pimozide Potential for serious and/or life-threatening reactions such as cardiac arrhythmias. Ergot Derivatives Dihydroergotamine
Ergotamine
MethylergonovinePotential for serious and/or life-threatening events such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. GI Motility Agent Cisapride Potential for serious and/or life-threatening events such as cardiac arrhythmias. Herbal Products St. John's wort (Hypericum perforatum) Coadministration of products containing St. John's wort and STRIBILD may result in reduced plasma concentrations of elvitegravir and cobicistat. This may result in loss of therapeutic effect and development of resistance. HMG-CoA Reductase Inhibitors Lovastatin
SimvastatinPotential for serious reactions such as myopathy, including rhabdomyolysis. Phosphodiesterase-5 (PDE-5) Inhibitor Sildenafil* when dosed as Revatio for the treatment of pulmonary arterial hypertension There is increased potential for sildenafil-associated adverse events (which include visual disturbances, hypotension, priapism, and syncope). Sedatives/Hypnotics Triazolam
Orally administered midazolam†Triazolam and orally administered midazolam are extensively metabolized by CYP3A4. Coadministration of triazolam or orally administered midazolam with STRIBILD may cause large increases in the concentration of these benzodiazepines. The potential exists for serious and/or life threatening events such as prolonged or increased sedation or respiratory depression. -
5 WARNINGS AND PRECAUTIONS
5.1 Lactic Acidosis/Severe Hepatomegaly with Steatosis
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs, including tenofovir DF, a component of STRIBILD, in combination with other antiretrovirals. A majority of these cases have been in women. Obesity and prolonged nucleoside exposure may be risk factors. Particular caution should be exercised when administering nucleoside analogs to any patient with known risk factors for liver disease; however, cases have also been reported in patients with no known risk factors. Treatment with STRIBILD should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
5.2 Severe Acute Exacerbation of Hepatitis B in Patients Coinfected with HIV-1 and HBV
All patients with HIV-1 should be tested for the presence of hepatitis B virus (HBV) before initiating antiretroviral therapy [see Dosage and Administration (2.1)]. STRIBILD is not approved for the treatment of chronic HBV infection, and the safety and efficacy of STRIBILD have not been established in patients coinfected with HIV-1 and HBV.
Severe acute exacerbations of hepatitis B (e.g., liver decompensated and liver failure) have been reported in patients who are coinfected with HBV and HIV-1 and have discontinued emtricitabine or tenofovir DF, two of the components of STRIBILD. Patients who are coinfected with HIV-1 and HBV should be closely monitored with both clinical and laboratory follow-up for at least several months after stopping treatment with STRIBILD. If appropriate, initiation of anti-hepatitis B therapy may be warranted, especially in patients with advanced liver disease or cirrhosis, since post-treatment exacerbation of hepatitis may lead to hepatic decompensation and liver failure.
5.3 New Onset or Worsening Renal Impairment
Renal impairment, including cases of acute renal failure and Fanconi syndrome (renal tubular injury with severe hypophosphatemia), has been reported with the use of tenofovir DF, a component of STRIBILD, and with the use of STRIBILD [see Adverse Reactions (6.2)].
In the clinical trials of STRIBILD over 144 weeks, 13 (1.9%) subjects in the STRIBILD group (N=701), 8 (2.3%) subjects in the atazanavir (ATV) + ritonavir (RTV) + TRUVADA® (emtricitabine 200 mg/tenofovir DF 300 mg) group (N=355), and no subjects in the ATRIPLA® (efavirenz 600 mg/emtricitabine 200 mg/tenofovir DF 300 mg) group (N=352) discontinued study drug due to a renal adverse reaction. Of these discontinuations, 8 in the STRIBILD group and 1 in the ATV+RTV+TRUVADA group occurred during the first 48 weeks. Four (0.6%) subjects who received STRIBILD developed laboratory findings consistent with proximal renal tubular dysfunction, leading to discontinuation of STRIBILD during the first 48 weeks of treatment. Two of the four subjects had renal impairment (i.e., estimated creatinine clearance less than 70 mL per minute) at baseline. The laboratory findings in these 4 subjects improved but did not completely resolve in all subjects upon discontinuation of STRIBILD. Renal replacement therapy was not required for these subjects. One (0.3%) subject who received ATV+RTV+TRUVADA developed laboratory findings consistent with proximal renal tubular dysfunction, leading to discontinuation of ATV+RTV+TRUVADA after Week 96.
STRIBILD should be avoided with concurrent or recent use of a nephrotoxic agent (e.g., high-dose or multiple nonsteroidal anti-inflammatory drugs [NSAIDs]) [see Drug Interactions (7.4)]. Cases of acute renal failure after initiation of high-dose or multiple NSAIDs have been reported in HIV-infected patients with risk factors for renal dysfunction who appeared stable on tenofovir DF. Some patients required hospitalization and renal replacement therapy. Alternatives to NSAIDs should be considered, if needed, in patients at risk for renal dysfunction.
Persistent or worsening bone pain, pain in extremities, fractures, and/or muscular pain or weakness may be manifestations of proximal renal tubulopathy and should prompt an evaluation of renal function in at-risk patients.
It is recommended that serum creatinine, serum phosphorus, estimated creatinine clearance, urine glucose, and urine protein be assessed before initiating STRIBILD and during therapy in all patients as clinically appropriate. Initiation of STRIBILD in patients with estimated creatinine clearance below 70 mL per minute is not recommended [see Dosage and Administration (2.1)].
Although cobicistat (a component of STRIBILD) may cause modest increases in serum creatinine and modest declines in estimated creatinine clearance without affecting renal glomerular function [see Adverse Reactions (6.1)], patients who experience a confirmed increase in serum creatinine of greater than 0.4 mg per dL from baseline should be closely monitored for renal safety.
The emtricitabine and tenofovir DF components of STRIBILD are primarily excreted by the kidney. STRIBILD should be discontinued if estimated creatinine clearance declines below 50 mL per minute as dose interval adjustment required for emtricitabine and tenofovir DF cannot be achieved with the fixed-dose combination tablet [see Use in Specific Populations (8.6)].
5.4 Risk of Adverse Reactions or Loss of Virologic Response Due to Drug Interactions
The concomitant use of STRIBILD and other drugs may result in known or potentially significant drug interactions, some of which may lead to [see Contraindications (4) and Drug Interactions (7.5)]:
- Loss of therapeutic effect of STRIBILD and possible development of resistance.
- Possible clinically significant adverse reactions from greater exposures of concomitant drugs.
See Table 6 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations [see Drug Interactions (7.5)]. Consider the potential for drug interactions prior to and during STRIBILD therapy; review concomitant medications during STRIBILD therapy; and monitor for the adverse reactions associated with the concomitant drugs.
5.5 Bone Loss and Mineralization Defects
Bone Mineral Density
In clinical trials in HIV-1-infected adults, tenofovir DF (a component of STRIBILD) was associated with slightly greater decreases in bone mineral density (BMD) and increases in biochemical markers of bone metabolism, suggesting increased bone turnover relative to comparators. Serum parathyroid hormone levels and 1,25 Vitamin D levels were also higher in subjects receiving tenofovir DF. For additional information, [see Adverse Reactions (6.1)] and consult the tenofovir DF prescribing information.
Clinical trials evaluating tenofovir DF in pediatric and adolescent subjects were conducted. Under normal circumstances, BMD increases rapidly in pediatric patients. In HIV-1-infected subjects aged 2 years to less than 18 years, bone effects were similar to those observed in adult subjects and suggest increased bone turnover. Total body BMD gain was less in the tenofovir DF-treated HIV-1-infected pediatric subjects as compared to the control groups. In all pediatric trials, skeletal growth (height) appeared to be unaffected. For more information, please consult the tenofovir DF prescribing information.
The effects of tenofovir DF-associated changes in BMD and biochemical markers on long-term bone health and future fracture risk are unknown. Assessment of BMD should be considered for HIV-1-infected adult and pediatric patients who have a history of pathologic bone fracture or other risk factors for osteoporosis or bone loss. Although the effect of supplementation with calcium and vitamin D was not studied, such supplementation may be beneficial in all patients. If bone abnormalities are suspected, then appropriate consultation should be obtained.
Mineralization Defects
Cases of osteomalacia associated with proximal renal tubulopathy, manifested as bone pain or pain in extremities and which may contribute to fractures, have been reported in association with the use of tenofovir DF [see Adverse Reactions (6.2)]. Arthralgias and muscle pain or weakness have also been reported in cases of proximal renal tubulopathy. Hypophosphatemia and osteomalacia secondary to proximal renal tubulopathy should be considered in patients at risk of renal dysfunction who present with persistent or worsening bone or muscle symptoms while receiving products containing tenofovir DF [see Warnings and Precautions (5.3)].
5.6 Fat Redistribution
Redistribution/accumulation of body fat including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
5.7 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including STRIBILD. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia (PCP), or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable and can occur many months after initiation of treatment.
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in other sections of the labeling:
- Lactic Acidosis/Severe Hepatomegaly with Steatosis [see Boxed Warning and Warnings and Precautions (5.1)].
- Severe Acute Exacerbations of Hepatitis B in Patients Coinfected with HIV-1 and HBV [see Boxed Warning and Warnings and Precautions (5.2)].
- New Onset or Worsening Renal Impairment [see Warnings and Precautions (5.3)].
- Bone Loss and Mineralization Defects [see Warnings and Precautions (5.5)].
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.7)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical Trials in HIV-1-Infected Adult Subjects with No Antiretroviral Treatment History
The safety assessment of STRIBILD is based on the Week-144 pooled data from 1408 subjects in two randomized, double-blind, active-controlled clinical trials, Study 102 and Study 103, in antiretroviral treatment-naïve HIV-1-infected adult subjects [see Clinical Studies (14)]. A total of 701 subjects received STRIBILD once daily in these two studies.
The proportion of subjects who discontinued treatment with STRIBILD, ATRIPLA, or ATV+RTV+TRUVADA due to adverse events, regardless of severity, was 6.0%, 7.4%, and 8.5%, respectively. Table 2 displays the frequency of adverse reactions greater than or equal to 5% of subjects in any treatment arm.
Table 2 Adverse Reactions* (All Grades) Reported in ≥5% of Adult Subjects in Any Treatment Arm in Studies 102 and 103 (Week-144 Analysis) STRIBILD
N=701ATRIPLA
N=352ATV+RTV+TRUVADA
N=355- * Frequencies of adverse reactions are based on all treatment-emergent adverse events attributed to study drugs.
- † Rash event includes dermatitis, drug eruption, eczema, pruritus, pruritus generalized, rash, rash erythematous, rash generalized, rash macular, rash maculo-papular, rash morbilliform, rash papular, rash pruritic, and urticaria.
EYE DISORDERS Ocular icterus <1% 0% 13% GASTROINTESTINAL DISORDERS Diarrhea 12% 11% 17% Flatulence 2% <1% 8% Nausea 16% 9% 14% GENERAL DISORDERS AND ADMINISTRATION SITE CONDITIONS Fatigue 4% 8% 6% HEPATOBILIARY DISORDERS Jaundice 0% <1% 9% NERVOUS SYSTEM DISORDERS Somnolence 1% 7% 1% Headache 7% 4% 6% Dizziness 3% 21% 5% PSYCHIATRIC DISORDERS Insomnia 3% 9% 1% Abnormal dreams 9% 27% 4% SKIN AND SUBCUTANEOUS TISSUE DISORDERS Rash† 4% 15% 6% See Warnings and Precautions (5.3) for a discussion of renal adverse reactions from clinical trials experience with STRIBILD.
Additional adverse reactions observed with STRIBILD included suicidal ideation and suicide attempt (0.3%), all in subjects with a preexisting history of depression or psychiatric illness.
Clinical Trials in Virologically Suppressed HIV-1-Infected Adult Subjects
No new adverse reactions to STRIBILD through Week 48 were identified in 584 virologically stably suppressed adult subjects switching to STRIBILD from a regimen containing a RTV-boosted protease inhibitor (PI) or a non-nucleoside reverse transcriptase inhibitor (NNRTI). In a combined analysis of studies 115 and 121, the frequency of adverse reactions (all grades) was 24% in subjects switching to STRIBILD compared to 6% of subjects in either group who stayed on their baseline antiretroviral regimen, RTV-boosted PI+TRUVADA or NNRTI+TRUVADA. Common adverse reactions that occurred in greater than or equal to 2% of subjects switching to STRIBILD were nausea (4%), flatulence (2%), and headache (2%). The proportion of subjects who discontinued treatment with STRIBILD, the RTV-boosted PI, or the NNRTI due to adverse events was 2%, 3%, and 1%, respectively.
Clinical Trials of the Components of STRIBILD in Adult Subjects
Emtricitabine and Tenofovir Disoproxil Fumarate: In addition to the adverse reactions observed with STRIBILD, the following adverse reactions occurred in at least 5% of treatment-experienced or treatment-naïve subjects receiving emtricitabine or tenofovir DF with other antiretroviral agents in other clinical trials: depression, abdominal pain, dyspepsia, vomiting, fever, pain, nasopharyngitis, pneumonia, sinusitis, upper respiratory tract infection, arthralgia, back pain, myalgia, paresthesia, peripheral neuropathy (including peripheral neuritis and neuropathy), anxiety, increased cough, and rhinitis.
Skin discoloration has been reported with higher frequency among emtricitabine-treated subjects; it was manifested by hyperpigmentation on the palms and/or soles and was generally mild and asymptomatic. The mechanism and clinical significance are unknown.
Laboratory Abnormalities:
The frequency of laboratory abnormalities (Grades 3–4) occurring in at least 2% of subjects receiving STRIBILD in studies 102 and 103 are presented in Table 3.
Table 3 Laboratory Abnormalities (Grades 3–4) Reported in ≥2% of Adult Subjects Receiving STRIBILD in Studies 102 and 103 (Week-144 Analysis) Laboratory Parameter Abnormality*,† STRIBILD
N=701ATRIPLA
N=352ATV+RTV+TRUVADA
N=355- * Frequencies are based on treatment-emergent laboratory abnormalities.
- † For subjects with serum amylase >1.5 × upper limit of normal (ULN), lipase test was also performed. The frequency of increased lipase (Grades 3–4) occurring in STRIBILD (N=69), ATRIPLA (N=40), and ATV+RTV+TRUVADA (N=38) was 17%, 15%, and 24%, respectively.
AST (>5.0 × ULN) 3% 6% 6% ALT (>3.0 × ULN) 2% 5% 4% Amylase* (>2.0 × ULN) 3% 3% 5% Creatine Kinase (≥10.0 × ULN) 8% 15% 11% Urine RBC (Hematuria) (>75 RBC/HPF) 4% 2% 4% In Study 103, BMD was assessed by DEXA in a nonrandom subset of 120 subjects (STRIBILD group, N=54; ATV+RTV+TRUVADA group, N=66). Mean percentage decreases in BMD from baseline to Week 144 in the STRIBILD group were comparable to that in the ATV+RTV+TRUVADA group at the lumbar spine (–1.43% versus –3.68%, respectively) and at the hip (–2.83% versus –3.77%, respectively). In studies 102 and 103, bone fractures occurred in 27 subjects (3.9%) in the STRIBILD group, 8 subjects (2.3%) in the ATRIPLA group, and 19 subjects (5.4%) in the ATV+RTV+TRUVADA group. These findings were consistent with data from an earlier 144-week trial of treatment-naïve subjects receiving tenofovir DF + lamivudine + efavirenz.
Proteinuria (all grades) occurred in 52% of subjects receiving STRIBILD, 41% of subjects receiving ATRIPLA, and 42% of subjects receiving ATV+RTV+TRUVADA.
The cobicistat component of STRIBILD has been shown to increase serum creatinine and decrease estimated creatinine clearance due to inhibition of tubular secretion of creatinine without affecting renal glomerular function. In studies 102 and 103, increases in serum creatinine and decreases in estimated creatinine clearance occurred early in treatment with STRIBILD, after which levels stabilized. Table 4 displays the mean changes in serum creatinine and eGFR levels at Week 144 and the percentage of subjects with elevations in serum creatinine (all grades).
Table 4 Change from Baseline in Serum Creatinine and eGFR and Incidence of Elevated Serum Creatinine (All Grades) in Studies 102 and 103 at Week 144 STRIBILD
N=701ATRIPLA
N=352ATV+RTV+TRUVADA
N=355- * Mean change ± standard deviation
Serum Creatinine (mg/dL)* 0.14 (±0.14) 0.01 (±0.12) 0.09 (±0.15) eGFR by Cockcroft-Gault (mL/minute)* −14.0 (±16.6) −1.9 (±17.9) −9.8 (±19.4) Subjects with Elevations in Serum Creatinine (All Grades) (%) 12 2 6 Emtricitabine or Tenofovir Disoproxil Fumarate: In addition to the laboratory abnormalities observed with STRIBILD, the following laboratory abnormalities have been previously reported in subjects treated with emtricitabine or tenofovir DF with other antiretroviral agents in other clinical trials: Grade 3 or 4 laboratory abnormalities of ALT (M: greater than 215 U per L; F: greater than 170 U per L), alkaline phosphatase (greater than 550 U per L), bilirubin (greater than 2.5 × ULN), serum glucose (less than 40 or greater than 250 mg per dL), glycosuria (greater than or equal to 3+), neutrophils (less than 750 per mm3), fasting cholesterol (greater than 240 mg per dL), and fasting triglycerides (greater than 750 mg per dL).
Serum Lipids: In the clinical trials of STRIBILD, a similar percentage of subjects receiving STRIBILD, ATRIPLA, and ATV+RTV+TRUVADA were on lipid-lowering agents at baseline (12%, 12%, and 13%, respectively). While receiving study drug through Week 144, an additional 11% of STRIBILD subjects were started on lipid-lowering agents, compared to 13% of ATRIPLA and 12% of ATV+RTV+TRUVADA subjects.
Changes from baseline in total cholesterol, HDL-cholesterol, LDL-cholesterol, and triglycerides are presented in Table 5.
Table 5 Lipid Values, Mean Change from Baseline at Week 144 in Adult Subjects Receiving STRIBILD or Comparator in Studies 102 and 103 STRIBILD
N=701ATRIPLA
N=352ATV+RTV+TRUVADA
N=355Baseline Week 144 Baseline Week 144 Baseline Week 144 mg/dL Change* mg/dL Change* mg/dL Change* - * The change from baseline is the mean of within-patient changes from baseline for patients with both baseline and Week 144 values.
Total Cholesterol (fasted) 166
[N=675]+17
[N=535]161
[N=343]+22
[N=262]168
[N=337]+16
[N=243]HDL-cholesterol (fasted) 43
[N=675]+7
[N=535]43
[N=343]+9
[N=262]42
[N=335]+7
[N=242]LDL-cholesterol (fasted) 100
[N=675]+15
[N=535]97
[N=343]+19
[N=262]101
[N=337]+18
[N=242]Triglycerides (fasted) 122
[N=675]+12
[N=535]121
[N=343]+5
[N=262]132
[N=337]+22
[N=242]Clinical Trials in Pediatric Subjects
The safety of STRIBILD in 50 HIV-1-infected, treatment-naïve pediatric subjects aged 12 to less than 18 years and weighing at least 35 kg (77 lbs) was evaluated through 48 weeks in an open-label clinical trial (Study 112) [see Clinical Studies (14.4)]. In this study, the safety profile of STRIBILD was similar to that in adults. Twenty-two subjects (44%) had treatment-emergent proteinuria (Grades 1–2). One subject met laboratory criteria for proximal renal tubulopathy, evidenced by sustained proteinuria and normoglycemic glycosuria beginning at Week 32. The subject continued to receive STRIBILD and was ultimately lost to follow-up.
Among the 50 pediatric subjects receiving STRIBILD for 48 weeks, mean BMD increased from baseline to Week 48, +0.68% at the lumbar spine and +0.77% for total body less head. Mean changes from baseline BMD Z-scores (height-age adjusted) to Week 48 were –0.09 for lumbar spine and –0.12 for total body less head. At Week 48, 7 STRIBILD subjects had significant (greater than or equal to 4%) lumbar spine BMD loss and 2 had significant total body less head BMD loss.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of tenofovir DF. Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. No additional postmarketing adverse reactions specific for emtricitabine have been identified.
Immune System Disorders
allergic reaction, including angioedemaMetabolism and Nutrition Disorders
lactic acidosis, hypokalemia, hypophosphatemiaRespiratory, Thoracic, and Mediastinal Disorders
dyspneaGastrointestinal Disorders
pancreatitis, increased amylase, abdominal painHepatobiliary Disorders
hepatic steatosis, hepatitis, increased liver enzymes (most commonly AST, ALT, gamma GT)Skin and Subcutaneous Tissue Disorders
rashMusculoskeletal and Connective Tissue Disorders
rhabdomyolysis, osteomalacia (manifested as bone pain and which may contribute to fractures), muscular weakness, myopathyRenal and Urinary Disorders
acute renal failure, renal failure, acute tubular necrosis, Fanconi syndrome, proximal renal tubulopathy, interstitial nephritis (including acute cases), nephrogenic diabetes insipidus, renal insufficiency, increased creatinine, proteinuria, polyuriaGeneral Disorders and Administration Site Conditions
astheniaThe following adverse reactions, listed under the body system headings above, may occur as a consequence of proximal renal tubulopathy: rhabdomyolysis, osteomalacia, hypokalemia, muscular weakness, myopathy, hypophosphatemia.
-
7 DRUG INTERACTIONS
7.1 Not Recommended with Other Antiretroviral Medications
STRIBILD is a complete regimen for the treatment of HIV-1 infection; therefore, STRIBILD should not be administered with other antiretroviral medications for treatment of HIV-1 infection. Complete information regarding potential drug-drug interactions with other antiretroviral medications is not provided [see Contraindications (4), Warnings and Precautions (5.4) and Clinical Pharmacology (12.3)].
7.2 Potential for STRIBILD to Affect Other Drugs
Cobicistat, a component of STRIBILD, is an inhibitor of CYP3A and CYP2D6 and an inhibitor of the following transporters: P-glycoprotein (P-gp), BCRP, OATP1B1, and OATP1B3. Thus, coadministration of STRIBILD with drugs that are primarily metabolized by CYP3A or CYP2D6, or are substrates of P-gp, BCRP, OATP1B1, or OATP1B3, may result in increased plasma concentrations of such drugs. Elvitegravir is a modest inducer of CYP2C9 and may decrease the plasma concentrations of CYP2C9 substrates.
7.3 Potential for Other Drugs to Affect One or More Components of STRIBILD
Elvitegravir and cobicistat, components of STRIBILD, are metabolized by CYP3A. Cobicistat is also metabolized, to a minor extent, by CYP2D6.
Drugs that induce CYP3A activity are expected to increase the clearance of elvitegravir and cobicistat, resulting in decreased plasma concentration of cobicistat and elvitegravir, which may lead to loss of therapeutic effect of STRIBILD and development of resistance (See Table 6).
Coadministration of STRIBILD with other drugs that inhibit CYP3A may decrease the clearance and increase the plasma concentration of cobicistat (See Table 6).
7.4 Drugs Affecting Renal Function
Because emtricitabine and tenofovir, components of STRIBILD, are primarily excreted by the kidneys by a combination of glomerular filtration and active tubular secretion, coadministration of STRIBILD with drugs that reduce renal function or compete for active tubular secretion may increase concentrations of emtricitabine, tenofovir, and other renally eliminated drugs and this may increase the risk of adverse reactions. Some examples of drugs that are eliminated by active tubular secretion include, but are not limited to, acyclovir, cidofovir, ganciclovir, valacyclovir, valganciclovir, aminoglycosides (e.g., gentamicin), and high-dose or multiple NSAIDs [see Warnings and Precautions (5.3)].
7.5 Established and Other Potentially Significant Interactions
Table 6 provides a listing of established or potentially clinically significant drug interactions. The drug interactions described are based on studies conducted with either STRIBILD or the components of STRIBILD (elvitegravir, cobicistat, emtricitabine, and tenofovir DF) as individual agents and/or in combination, or are predicted drug interactions that may occur with STRIBILD [for magnitude of interaction see Clinical Pharmacology (12.3)]. The table includes potentially significant interactions but is not all inclusive [see Contraindications (4) and Clinical Pharmacology (12.3)].
Table 6 Established and Other Potentially Significant* Drug Interactions: Alteration in Dose or Regimen May Be Recommended Based on Drug Interaction Studies or Predicted Interaction Concomitant Drug Class: Drug Name Effect on Concentration† Clinical Comment - * This table is not all inclusive.
- † ↑=Increase, ↓=Decrease
- ‡ Indicates that a drug-drug interaction trial was conducted.
Acid Reducing Agents:
Antacids‡ e.g., aluminum and magnesium hydroxide↓ elvitegravir Separate STRIBILD and antacid administration by at least 2 hours. Antiarrhythmics:
e.g.,
amiodarone
bepridil
digoxin‡
disopyramide
flecainide
systemic lidocaine
mexiletine
propafenone
quinidine↑ antiarrhythmics
↑ digoxinTherapeutic concentration monitoring, if available, is recommended for antiarrhythmics when coadministered with STRIBILD. Antibacterials:
clarithromycin↑ clarithromycin
↑ cobicistatPatients with CLcr greater than or equal to 60 mL/minute:
No dose adjustment of clarithromycin is required.
Patients with CLcr between 50 mL/minute and 60 mL/minute:
The dose of clarithromycin should be reduced by 50%.Anticoagulants:
warfarinEffect on warfarin unknown Monitor international normalized ratio (INR) upon coadministration of warfarin with STRIBILD. Anticonvulsants:
oxcarbazepine↓ elvitegravir
↓ cobicistatContraindicated anticonvulsants [see Contraindications (4)] clonazepam
ethosuximide↑ clonazepam
↑ ethosuximideAlternative anticonvulsants should be considered when STRIBILD is coadministered with oxcarbazepine.
Clinical monitoring is recommended upon coadministration of clonazepam or ethosuximide with STRIBILD.Antidepressants:
Selective Serotonin Reuptake Inhibitors (SSRIs)
e.g.,
paroxetine
Tricyclic Antidepressants (TCAs)
e.g.,
amitriptyline
desipramine
imipramine
nortriptyline
bupropion
trazodone
↑ SSRIs
↑ TCAs
↑ trazodoneCareful dose titration of the antidepressant and monitoring for antidepressant response are recommended when coadministered with STRIBILD. Antifungals:
itraconazole
ketoconazole‡
voriconazole↑ elvitegravir
↑ cobicistat
↑ itraconazole
↑ ketoconazole
↑ voriconazoleWhen coadministered with STRIBILD, the maximum daily dose of ketoconazole or itraconazole should not exceed 200 mg per day.
An assessment of benefit/risk ratio is recommended to justify use of voriconazole with STRIBILD.Anti-gout:
colchicine↑ colchicine STRIBILD is not recommended to be coadministered with colchicine to patients with renal or hepatic impairment.
Treatment of gout-flares – coadministration of colchicine in patients receiving STRIBILD:
0.6 mg (1 tablet) × 1 dose, followed by 0.3 mg (half tablet) 1 hour later. Treatment course to be repeated no earlier than 3 days.
Prophylaxis of gout-flares – coadministration of colchicine in patients receiving STRIBILD:
If the original regimen was 0.6 mg twice a day, the regimen should be adjusted to 0.3 mg once a day. If the original regimen was 0.6 mg once a day, the regimen should be adjusted to 0.3 mg once every other day.
Treatment of familial Mediterranean fever – coadministration of colchicine in patients receiving STRIBILD:
Maximum daily dose of 0.6 mg (may be given as 0.3 mg twice a day).Antimycobacterial:
rifabutin‡
rifapentine↓ elvitegravir
↓ cobicistatContraindicated antimycobacterials [see Contraindications (4)]
Coadministration of STRIBILD with rifabutin or rifapentine is not recommended.Antipsychotics:
e.g.,
perphenazine
risperidone
thioridazine
quetiapine↑ antipsychotic
↑ quetiapineContraindicated antipsychotics [see Contraindications (4)]
A decrease in the dose of antipsychotics that are metabolized by CYP3A4 or CYP2D6 may be needed when coadministered with STRIBILD.
Initiation of STRIBILD in patients taking quetiapine:
Consider alternative antiretroviral therapy to avoid increases in quetiapine exposure. If coadministration is necessary, reduce the quetiapine dose to 1/6 of the current dose and monitor for quetiapine-associated adverse reactions. Refer to the quetiapine prescribing information for recommendations on adverse reaction monitoring.
Initiation of quetiapine in patients taking STRIBILD:
Refer to the quetiapine prescribing information for initial dosing and titration of quetiapine.Beta-Blockers:
e.g.,
metoprolol
timolol↑ beta-blockers Clinical monitoring is recommended and a dose decrease of the beta-blocker may be necessary when these agents are coadministered with STRIBILD. Calcium Channel Blockers:
e.g.,
amlodipine
diltiazem
felodipine
nicardipine
nifedipine
verapamil↑ calcium channel blockers Clinical monitoring is recommended upon coadministration of calcium channel blockers with STRIBILD. Corticosteroid:
Systemic:
dexamethasone↓ elvitegravir
↓ cobicistatAlternative corticosteroids should be considered. Corticosteroid:
Inhaled/Nasal:
fluticasone↑ fluticasone Concomitant use of STRIBILD with inhaled or nasal fluticasone may reduce serum cortisol concentrations. Alternative corticosteroids should be considered, particularly for long-term use. Endothelin Receptor Antagonists:
bosentan↑ bosentan Coadministration of bosentan in patients on STRIBILD:
In patients who have been receiving STRIBILD for at least 10 days, start bosentan at 62.5 mg once daily or every other day based upon individual tolerability.
Coadministration of STRIBILD in patients on bosentan:
Discontinue use of bosentan at least 36 hours prior to initiation of STRIBILD. After at least 10 days following the initiation of STRIBILD, resume bosentan at 62.5 mg once daily or every other day based upon individual tolerability.Hepatitis C Antiviral Agents:
ledipasvir/sofosbuvir↑ tenofovir The safety of increased tenofovir concentrations in the setting of HARVONI® and STRIBILD has not been established. Coadministration is not recommended. HMG-CoA Reductase Inhibitors:
atorvastatin↑ atorvastatin Contraindicated HMG-CoA Reductase Inhibitors [see Contraindications (4)]
Initiate with the lowest starting dose of atorvastatin and titrate carefully while monitoring for safety.Hormonal Contraceptives:
norgestimate/ethinyl estradiol‡↑ norgestimate
↓ ethinyl estradiolThe effects of increases in the concentration of the progestational component norgestimate are not fully known and can include increased risk of insulin resistance, dyslipidemia, acne, and venous thrombosis. The potential risks and benefits associated with coadministration of norgestimate/ethinyl estradiol with STRIBILD should be considered, particularly in women who have risk factors for these events. Coadministration of STRIBILD with other hormonal contraceptives (e.g., contraceptive patch, contraceptive vaginal ring, or injectable contraceptives) or oral contraceptives containing progestogens other than norgestimate has not been studied; therefore, alternative (nonhormonal) methods of contraception can be considered.
Immuno-suppressants:
e.g.,
cyclosporine
sirolimus
tacrolimus↑ immuno-suppressants Therapeutic monitoring of the immunosuppressive agents is recommended upon coadministration with STRIBILD. Narcotic Analgesics:
buprenorphine/naloxone‡↑ buprenorphine
↑ norbuprenorphine
↓ naloxonePatients should be closely monitored for sedation and cognitive effects. Inhaled Beta Agonist:
salmeterol↑ salmeterol Coadministration of salmeterol and STRIBILD is not recommended because it may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations, and sinus tachycardia. Phosphodiesterase-5 (PDE-5) Inhibitors:
sildenafil
tadalafil
vardenafil↑ PDE-5 inhibitors Contraindicated PDE-5 Inhibitors [see Contraindications (4)] Coadministration with STRIBILD may result in an increase in PDE-5 inhibitor associated adverse reactions, including hypotension, syncope, visual disturbances, and priapism.
Use of tadalafil for pulmonary arterial hypertension (PAH):
-
Coadministration of tadalafil in patients on STRIBILD:
- In patients receiving STRIBILD for at least 1 week, start tadalafil at 20 mg once daily. Increase tadalafil dose to 40 mg once daily based upon individual tolerability.
-
Coadministration of STRIBILD in patients on tadalafil:
- Avoid use of tadalafil during the initiation of STRIBILD. Stop tadalafil at least 24 hours prior to starting STRIBILD. After at least one week following initiation of STRIBILD, resume tadalafil at 20 mg once daily. Increase tadalafil dose to 40 mg once daily based upon individual tolerability.
Use of PDE-5 inhibitors for erectile dysfunction:
The below PDE-5 inhibitors can be used along with increased monitoring for PDE-5-inhibitor associated adverse events:
- Sildenafil at a single dose not exceeding 25 mg in 48 hours, or
- Tadalafil at a single dose not exceeding 10 mg in 72 hours, or
- Vardenafil at a single dose not exceeding 2.5 mg in 72 hours
Sedative/hypnotics:
Benzodiazepines:
e.g.,
parenterally administered midazolam
clorazepate
diazepam
estazolam
flurazepam
buspirone
zolpidem↑ sedatives/hypnotics Contraindicated sedative/hypnotics [see Contraindications (4)] Coadministration of parenteral midazolam with STRIBILD should be done in a setting that ensures close clinical monitoring and appropriate medical management in case of respiratory depression and/or prolonged sedation. Dosage reduction for midazolam should be considered, especially if more than a single dose of midazolam is administered.
With other sedative/hypnotics, dose reduction may be necessary and clinical monitoring is recommended.
7.6 Drugs without Clinically Significant Interactions with STRIBILD
Based on drug interaction studies conducted with the components of STRIBILD, no clinically significant drug interactions have been either observed or are expected when STRIBILD is combined with the following drugs: entecavir, famciclovir, H2 receptor antagonists, methadone, proton pump inhibitors, and ribavirin.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to STRIBILD during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Risk Summary
Prospective pregnancy data from the Antiviral Pregnancy Registry (APR) are not sufficient to adequately assess the risk of birth defects of miscarriage. However, elvitegravir, cobicistat, emtricitabine, and tenofovir DF use during pregnancy has been evaluated in a limited number of women as reported to the APR. Available data from the APR through January 2016 show no birth defects reported for elvitegravir or cobicistat, and no difference in the overall risk of major birth defects for emtricitabine or tenofovir DF compared with the background rate for major birth defects of 2.7% in a U.S. reference population of the Metropolitan Atlanta Congenital Defects Program (MACDP) [see Data].
In animal studies, no adverse developmental effects were observed when the components of STRIBILD were administered separately during the period of organogenesis at exposures up to 23 and 0.2 times (rats and rabbits, respectively, elvitegravir), 1.8 and 4.3 times (rats and rabbits, respectively, cobicistat), and 60 and 120 times (mice and rabbits, respectively, emtricitabine) the exposure at the recommended daily dose of these components in STRIBILD, and at 14 and 19 times (rats and rabbits, respectively, tenofovir DF) the human dose based on body surface area comparisons [see Data]. Likewise, no adverse developmental effects were seen when elvitegravir or cobicistat was administered to rats through lactation at exposures up to 18 times or 1.2 times, respectively, the exposure at the recommended daily therapeutic dose, and when emtricitabine was administered to mice through lactation at exposures up to approximately 60 times the exposure at the recommended daily therapeutic dose. No adverse effects were observed in the offspring of rats when tenofovir DF was administered through lactation at tenofovir exposures of approximately 2.7 times the exposure at the recommended daily dosage of STRIBILD.
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defects, loss, or other adverse outcomes. The rate of miscarriage is not reported in the APR. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Data
Human Data
Elvitegravir: Based on prospective reports from the APR through January 2016 of 73 exposures to elvitegravir-containing regimens during pregnancy resulting in live births (including 51 exposed in the first trimester), there have been no birth defects reported.
Cobicistat: Based on prospective reports from the APR through January 2016 of 77 exposures to cobicistat-containing regimens during pregnancy resulting in live births (including 54 exposed in the first trimester), there have been no birth defects reported.
Emtricitabine: Based on prospective reports to the APR through January 2016 of 3155 exposures to emtricitabine-containing regimens during pregnancy resulting in live births (including 2145 exposed in the first trimester and 1010 exposed in the second/third trimester), there was no difference between emtricitabine and overall birth defects compared with the background birth defect rate of 2.7% in the U.S. reference population of the MACDP. The prevalence of birth defects in live births was 2.2% (95% CI: 1.6% to 3.0%) with first trimester exposure to emtricitabine-containing regimens and 2.1% (95% CI: 1.3% to 3.2%) with the second/third trimester exposure to emtricitabine-containing regimens.
Tenofovir DF: Based on prospective reports from the APR through January 2016 of 4100 exposures to tenofovir DF-containing regimens during pregnancy resulting in live births (including 2779 exposed in the first trimester and 1321 exposed in the second/third trimester), there was no difference between tenofovir DF and overall birth defects compared with the background birth defect rate of 2.7% in the U.S. reference population of the MACDP. The prevalence of birth defects in live births was 2.2% (95% CI: 1.7% to 2.8%) with first trimester exposure, and 2.0% (95% CI: 1.3% to 3.0%) with the second/third trimester exposure to tenofovir DF-containing regimens.
Animal Data
Elvitegravir: Elvitegravir was administered orally to pregnant rats (at 0, 300, 1000, and 2000 mg/kg/day), and rabbits (at 0, 50, 150, and 450 mg/kg/day) through organogenesis (on gestation days 7 through 17 and days 7 through 19, respectively). No significant toxicological effects were observed in embryo-fetal toxicity studies performed with elvitegravir in rats at exposures (AUC) approximately 23 times higher and in rabbits at approximately 0.2 times higher than human exposures at the recommended daily dose. In a pre- and postnatal developmental study in rats, elvitegravir was administered orally at doses of 0, 300, 1000, and 2000 mg/kg from gestation day 7 to day 20 of lactation. At doses of 2000 mg/kg/day of elvitegravir, neither maternal nor developmental toxicity was noted. Systemic exposures (AUC) at this dose were 18 times the human exposures at the recommended daily dose.
Cobicistat: Cobicistat was administered orally to pregnant rats at doses of 0, 25, 50, and 125 mg/kg/day on gestation day 6 to 17. Increases in post-implantation loss and decreased fetal weights were observed at a maternal toxic dose of 125 mg/kg/day. No malformations were noted at doses up to 125 mg/kg/day. Systemic exposures (AUC) at 50 mg/kg/day in pregnant females were 1.8 times higher than human exposures at the recommended daily dose.
In pregnant rabbits, cobicistat was administered orally at doses of 0, 20, 50, and 100 mg/kg/day during the gestation days 7 to 20. No maternal or embryo/fetal effects were noted at the highest dose of 100 mg/kg/day. Systemic exposures (AUC) at 100 mg/kg/day were 4.3 times higher than human exposures at the recommended daily dose. In a pre- and postnatal developmental study in rats, cobicistat was administered orally at doses of 0, 10, 30, and 75 mg/kg from gestation day 6 to postnatal day 20, 21, or 22. At doses of 75 mg/kg/day of cobicistat, neither maternal nor developmental toxicity was noted. Systemic exposures (AUC) at this dose were 1.2 times the human exposures at the recommended daily dose.
Emtricitabine: Emtricitabine was administered orally to pregnant mice (at 0, 250, 500, or 1000 mg/kg/day), and rabbits (at 0, 100, 300, or 1000 mg/kg/day) through organogenesis (on gestation days 6 through 15, and 7 through 19, respectively). No significant toxicological effects were observed in embryo-fetal toxicity studies performed with emtricitabine in mice at exposures (AUC) approximately 60 times higher and in rabbits at approximately 120 times higher than human exposures at the recommended daily dose. In a pre/postnatal development study in mice, emtricitabine was administered orally at doses up to 1000 mg/kg/day; no significant adverse effects directly related to drug were observed in the offspring exposed daily from before birth (in utero) through sexual maturity at daily exposures (AUC) of approximately 60 times higher than human exposures at the recommended daily dose.
Tenofovir DF: Tenofovir DF was administered orally to pregnant rats (at 0, 50, 150, or 450 mg/kg/day) and rabbits (at 0, 30, 100, or 300 mg/kg/day) through organogenesis (on gestation days 7 through 17, and 6 through 18, respectively). No significant toxicological effects were observed in embryo-fetal toxicity studies performed with tenofovir DF in rats at doses up to 14 times the human dose based on body surface area comparisons and in rabbits at doses up to 19 times the human dose based on body surface area comparisons. In a pre/postnatal development study in rats, tenofovir DF was administered orally through lactation at doses up to 600 mg/kg/day; no adverse effects were observed in the offspring at tenofovir exposures of approximately 2.7 times higher than human exposures at the recommended daily dose of STRIBILD.
8.2 Lactation
Risk Summary
The Centers for Disease Control and Prevention recommend that HIV-infected mothers not breastfeed their infants to avoid risking postnatal transmission of HIV.
Based on limited published data, emtricitabine and tenofovir have been shown to be present in human breast milk. It is not known whether elvitegravir or cobicistat are present in human breast milk, while elvitegravir and cobicistat have been shown to be present in rat milk [see Data].
It is not known if the components of STRIBILD affect milk production or have effects on the breastfed child. Because of the potential for: (1) HIV transmission (in HIV-negative infants); (2) developing viral resistance (in HIV-positive infants); and (3) adverse reactions in a breastfed infant similar to those seen in adults, instruct mothers not to breastfeed if they are receiving STRIBILD [see Data].
8.4 Pediatric Use
The pharmacokinetics, safety, and virologic and immunologic responses were evaluated in 50 treatment-naïve, HIV-1-infected subjects aged 12 to less than 18 years weighing at least 35 kg (77 lbs) receiving STRIBILD through 48 weeks in an open-label trial (Study 112). The safety and efficacy of STRIBILD in these subjects was similar to that in antiretroviral treatment-naïve adults [see Dosage and Administration (2.2), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.4)].
Safety and effectiveness of STRIBILD in pediatric patients less than 12 years of age or weighing less than 35 kg (77 lbs) have not been established.
8.5 Geriatric Use
Clinical studies of STRIBILD did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, caution should be exercised in the administration of STRIBILD in elderly patients, keeping in mind the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Initiation of STRIBILD in patients with estimated creatinine clearance below 70 mL per min is not recommended. Because STRIBILD is a fixed-dose combination tablet, STRIBILD should be discontinued if estimated creatinine clearance declines below 50 mL per minute during treatment with STRIBILD as dose interval adjustment required for emtricitabine and tenofovir DF cannot be achieved [see Warnings and Precautions (5.3), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
No data are available to make dose recommendations for pediatric patients with renal impairment.
Clinical Trials in Adult Subjects with Mild to Moderate Renal Impairment
In Study 118, 33 HIV-1-infected treatment-naïve subjects with mild to moderate renal impairment (eGFR by Cockcroft-Gault method between 50 and 89 mL/minute) were studied in an open-label clinical trial evaluating the safety of 48 weeks of treatment with STRIBILD. After 48 weeks of treatment, the mean change in serum creatinine was 0.17 ± 0.14 mg/dL and the mean change in eGFR by Cockcroft-Gault method was –6.9 ± 9.0 mL/minute for subjects treated with STRIBILD.
Twelve of the 33 subjects studied had baseline eGFR between 50 and 70 mL/minute. Three subjects, all with baseline eGFR between 50–60 mL/minute, discontinued STRIBILD due to a renal adverse event. The safety of STRIBILD among 21 of the 33 subjects with baseline eGFR greater than or equal to 70 mL/minute was consistent with the safety profile in studies 102 and 103.
8.7 Hepatic Impairment
No dose adjustment of STRIBILD is required in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. No pharmacokinetic or safety data are available regarding the use of STRIBILD in patients with severe hepatic impairment (Child-Pugh Class C). Therefore, STRIBILD is not recommended for use in patients with severe hepatic impairment [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
No data are available on overdose of STRIBILD in patients. If overdose occurs the patient must be monitored for evidence of toxicity. Treatment of overdose with STRIBILD consists of general supportive measures, including monitoring of vital signs as well as observation of the clinical status of the patient.
Elvitegravir: Limited clinical experience is available at doses higher than the therapeutic dose of elvitegravir. In one study, boosted elvitegravir equivalent to 2 times the therapeutic dose of 150 mg once daily for 10 days was administered to 42 healthy subjects. No severe adverse reactions were reported. The effects of higher doses are not known. As elvitegravir is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis.
Cobicistat: Limited clinical experience is available at doses higher than the therapeutic dose of cobicistat. In two studies, a single dose of cobicistat 400 mg (2.7 times the dose in STRIBILD) was administered to a total of 60 healthy subjects. No severe adverse reactions were reported. The effects of higher doses are not known. As cobicistat is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis.
Emtricitabine: Limited clinical experience is available at doses higher than the therapeutic dose of EMTRIVA. In one clinical pharmacology study, single doses of emtricitabine 1200 mg (6 times the dose in STRIBILD) were administered to 11 subjects. No severe adverse reactions were reported. The effects of higher doses are not known.
Hemodialysis treatment removes approximately 30% of the emtricitabine dose over a 3-hour dialysis period starting within 1.5 hours of emtricitabine dosing (blood flow rate of 400 mL per minute and a dialysate flow rate of 600 mL per minute). It is not known whether emtricitabine can be removed by peritoneal dialysis.
Tenofovir Disoproxil Fumarate: Limited clinical experience at doses higher than the therapeutic dose of VIREAD 300 mg is available. In one study, 600 mg tenofovir DF (2 times the dosage in STRIBILD) was administered to 8 subjects orally for 28 days and no severe adverse reactions were reported. The effects of higher doses are not known. Tenofovir is efficiently removed by hemodialysis with an extraction coefficient of approximately 54%. Following a single 300 mg dose of VIREAD, a 4-hour hemodialysis session removed approximately 10% of the administered tenofovir dose.
-
11 DESCRIPTION
STRIBILD is a fixed-dose combination tablet containing elvitegravir, cobicistat, emtricitabine, and tenofovir DF for oral administration.
- Elvitegravir is an HIV-1 integrase strand transfer inhibitor.
- Cobicistat is a mechanism-based inhibitor of cytochrome P450 (CYP) enzymes of the CYP3A family.
- Emtricitabine is a synthetic nucleoside analog of cytidine. EMTRIVA is the brand name for emtricitabine.
- Tenofovir DF is converted in vivo to tenofovir, an acyclic nucleoside phosphonate (nucleotide) analog of adenosine 5′-monophosphate. VIREAD is the brand name for tenofovir DF.
Each tablet contains 150 mg of elvitegravir, 150 mg of cobicistat, 200 mg of emtricitabine, and 300 mg of tenofovir DF (equivalent to 245 mg of tenofovir disoproxil). The tablets include the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, silicon dioxide, croscarmellose sodium, hydroxypropyl cellulose, sodium lauryl sulfate, and magnesium stearate. The tablets are film coated with a coating material containing indigo carmine (FD&C Blue #2) aluminum lake, polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide, and yellow iron oxide.
Elvitegravir: The chemical name of elvitegravir is 6-(3-Chloro-2-fluorobenzyl)-1-[(2S)-1-hydroxy-3-methylbutan-2-yl]-7-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid.
It has a molecular formula of C23H23ClFNO5 and a molecular weight of 447.9. It has the following structural formula:
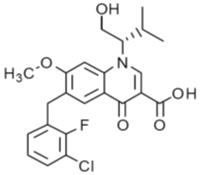
Elvitegravir is a white to pale-yellow powder with a solubility of less than 0.3 micrograms per mL in water at 20 °C.
Cobicistat: The chemical name for cobicistat is 1,3-thiazol-5-ylmethyl [(2R,5R)-5-{[(2S)-2-[(methyl{[2-(propan-2-yl)-1,3-thiazol-4-yl]methyl}carbamoyl)amino]-4-(morpholin-4-yl)butanoyl]amino}-1,6-diphenylhexan-2-yl]carbamate.
It has a molecular formula of C40H53N7O5S2 and a molecular weight of 776.0. It has the following structural formula:
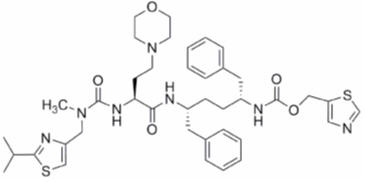
Cobicistat is adsorbed onto silicon dioxide. Cobicistat on silicon dioxide is a white to pale-yellow solid with a solubility of 0.1 mg per mL in water at 20 °C.
Emtricitabine: The chemical name of emtricitabine is 5-fluoro-1-[(2R,5S)-2-(hydroxymethyl)-1,3-oxathiolan-5-yl]cytosine. Emtricitabine is the (-)enantiomer of a thio analog of cytidine, which differs from other cytidine analogs in that it has a fluorine in the 5-position.
It has a molecular formula of C8H10FN3O3S and a molecular weight of 247.25. It has the following structural formula:
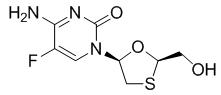
Emtricitabine is a white to off-white crystalline powder with a solubility of approximately 112 mg per mL in water at 25 °C.
Tenofovir Disoproxil Fumarate: Tenofovir DF is a fumaric acid salt of the bis-isopropoxycarbonyloxymethyl ester derivative of tenofovir. The chemical name of tenofovir DF is 9-[(R)-2-[[bis[[(isopropoxycarbonyl)oxy]-methoxy]phosphinyl]methoxy]propyl]adenine fumarate (1:1). It has a molecular formula of C19H30N5O10P ∙ C4H4O4 and a molecular weight of 635.51. It has the following structural formula:
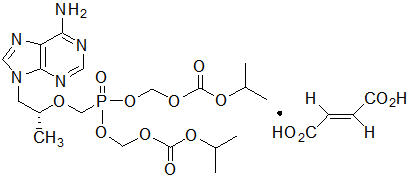
Tenofovir DF is a white to off-white crystalline powder with a solubility of 13.4 mg per mL in water at 25 °C. All dosages are expressed in terms of tenofovir DF except where otherwise noted.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
STRIBILD is a fixed-dose combination of antiretroviral drugs elvitegravir (boosted by the CYP3A inhibitor cobicistat), emtricitabine, and tenofovir DF [see Microbiology (12.4)].
12.2 Pharmacodynamics
Effects on Electrocardiogram
Thorough QT studies have been conducted for elvitegravir and cobicistat. The effect of the other two components, tenofovir and emtricitabine, or the combination regimen STRIBILD on the QT interval is not known.
Elvitegravir: In a thorough QT/QTc study in 126 healthy subjects, elvitegravir (coadministered with 100 mg ritonavir) 125 mg and 250 mg (0.83 and 1.67 times the dose in STRIBILD) did not affect the QT/QTc interval and did not prolong the PR interval.
Cobicistat: In a thorough QT/QTc study in 48 healthy subjects, a single dose of cobicistat 250 mg and 400 mg (1.67 and 2.67 times the dose in STRIBILD) did not affect the QT/QTc interval. Prolongation of the PR interval was noted in subjects receiving cobicistat. The maximum mean (95% upper confidence bound) difference in PR from placebo after baseline-correction was 9.5 (12.1) msec for the 250 mg cobicistat dose and 20.2 (22.8) for the 400 mg cobicistat dose. Because the 150 mg cobicistat dose used in the STRIBILD fixed-dose combination tablet is lower than the lowest dose studied in the thorough QT study, it is unlikely that treatment with STRIBILD will result in clinically relevant PR prolongation.
Effects on Serum Creatinine
The effect of cobicistat on serum creatinine was investigated in a Phase 1 study in subjects with an eGFR of at least 80 mL per minute (N=18) and with an eGFR of 50 to 79 mL per minute (N=12). A statistically significant change of eGFRCG from baseline was observed after 7 days of treatment with cobicistat 150 mg among subjects with an eGFR of at least 80 mL per minute (-9.9 ± 13.1 mL/min) and subjects with an eGFR of 50 to 79 mL per minute (-11.9 ± 7.0 mL per minute). These decreases in eGFRCG were reversible after cobicistat was discontinued. The actual glomerular filtration rate, as determined by the clearance of probe drug iohexol, was not altered from baseline following treatment of cobicistat among subjects with an eGFR of at least 50 mL per minute, indicating cobicistat inhibits tubular secretion of creatinine, reflected as a reduction in eGFRCG, without affecting the actual glomerular filtration rate.
12.3 Pharmacokinetics
The pharmacokinetic properties of the components of STRIBILD are provided in Table 7. The multiple dose pharmacokinetic parameters of elvitegravir, cobicistat, emtricitabine, and tenofovir are provided in Table 8.
Table 7 Pharmacokinetic Properties of the Components of STRIBILD Elvitegravir Cobicistat Emtricitabine Tenofovir NC=Not Calculated - * Values refer to mean systemic exposure (90% confidence interval). STRIBILD light meal=~373 kcal, 20% fat; STRIBILD high fat meal=~800 kcal, 50% fat. Increase = ↑; Decrease = ↓
- † t1/2 values refer to median terminal plasma half-life.
- ‡ Dosing in mass balance studies: elvitegravir (single dose administration of [14C] elvitegravir,coadministered with 100 mg RTV); cobicistat (single-dose administration of [14C] cobicistat after multiple dosing of cobicistat for six days); emtricitabine (single dose administration of [14C] emtricitabine after multiple dosing of emtricitabine for ten days); mass balance study not conducted for tenofovir.
Absorption Tmax (h) 4 3 3 2 Effect of light meal (relative to fasting)* ↑34%
(↑19, ↑51)↑3%
(↓10, ↑17)↓5%
(↓9, 0)↑24%
(↑18, ↑30)Effect of high fat meal (relative to fasting)* ↑87%
(↑66, ↑110)↓17%
(↓27, ↓5)↓4%
(↓8, 0)↑23%
(↑17, ↑29)Distribution % Bound to human plasma proteins ~99 ~98 <4 <0.7 Source of protein binding data Ex vivo In vitro In vitro In vitro Blood-to-plasma ratio 0.73 0.5 0.6 NC Metabolism Metabolism CYP3A (major)
UGT1A1/3 (minor)CYP3A (major)
CYP2D6 (minor)Not significantly metabolized Elimination Major route of elimination Metabolism Glomerular filtration and active tubular secretion T1/2 (h)† 12.9 3.5 10 12–18 % Of dose excreted in urine‡ 6.7 8.2 70 70–80 % Of dose excreted in feces‡ 94.8 86.2 13.7 NC Table 8 Pharmacokinetic Parameters of Elvitegravir, Cobicistat, Emtricitabine, and Tenofovir Exposure Following Oral Administration of STRIBILD in HIV-Infected Subjects Parameter
Mean ± SD
[range, min:max]Elvitegravir* Cobicistat† Emtricitabine† Tenofovir† SD=Standard Deviation - * From Population Pharmacokinetic analysis, N=419.
- † From Intensive Pharmacokinetic analysis, N=61−62, except cobicistat Ctrough N=53.
Cmax
(microgram per mL)1.7 ± 0.4
[0.4:3.7]1.1 ± 0.4
[0.1:2.1]1.9 ± 0.5
[0.6:3.6]0.45 ± 0.2
[0.2:1.2]AUCtau
(microgram∙hour per mL)23.0 ± 7.5
[4.4:69.8]8.3 ± 3.8
[0.5:18.3]12.7 ± 4.5
[5.2:34.1]4.4 ± 2.2
[2.1:18.2]Ctrough
(microgram per mL)0.45 ± 0.26
[0.05:2.34]0.05 ± 0.13
[0.01:0.92]0.14 ± 0.25
[0.04:1.94]0.10 ± 0.08
[0.04:0.58]Specific Populations
Patients with Renal Impairment
Elvitegravir and Cobicistat: A study of the pharmacokinetics of cobicistat+elvitegravir was performed in healthy subjects and subjects with severe renal impairment (estimated creatinine clearance less than 30 mL per minute). No clinically relevant differences in elvitegravir or cobicistat pharmacokinetics were observed between healthy subjects and subjects with severe renal impairment.
Emtricitabine and Tenofovir Disoproxil Fumarate: The pharmacokinetics of emtricitabine and tenofovir are altered in subjects with estimated creatinine clearance below 50 mL per minute or with end-stage renal disease requiring dialysis [see Warnings and Precautions (5.3) and Use in Specific Populations (8.6)].
Patients with Hepatic Impairment
Elvitegravir and Cobicistat: A study of the pharmacokinetics of cobicistat+elvitegravir was performed in healthy subjects and subjects with moderate hepatic impairment. No clinically relevant differences in elvitegravir or cobicistat pharmacokinetics were observed between subjects with moderate hepatic impairment (Child-Pugh Class B) and healthy subjects. The effect of severe hepatic impairment (Child-Pugh Class C) on the pharmacokinetics of elvitegravir or cobicistat has not been studied [see Use in Specific Populations (8.7)].
Emtricitabine: The pharmacokinetics of emtricitabine have not been studied in subjects with hepatic impairment; however, emtricitabine is not significantly metabolized by liver enzymes, so the impact of liver impairment should be limited.
Tenofovir Disoproxil Fumarate: The pharmacokinetics of tenofovir following a 300 mg dose of VIREAD have been studied in healthy subjects with moderate to severe hepatic impairment. No clinically relevant differences in tenofovir pharmacokinetics were observed between subjects with hepatic impairment and healthy subjects.
Hepatitis B and/or Hepatitis C Virus Coinfection
Elvitegravir: Limited data from population pharmacokinetic analysis (N=24) indicated that hepatitis B and/or C virus infection had no clinically relevant effect on the exposure of cobicistat-boosted elvitegravir.
Race
Elvitegravir: Population pharmacokinetic analysis of elvitegravir in HIV-1-infected subjects indicated that race had no clinically relevant effect on the exposure of cobicistat-boosted elvitegravir.
Cobicistat: Population pharmacokinetics analysis of cobicistat in HIV-1-infected subjects indicated that race had no clinically relevant effect on the exposure of cobicistat.
Gender
No clinically relevant pharmacokinetic differences have been observed between men and women for cobicistat-boosted elvitegravir, emtricitabine, and tenofovir DF.
Pediatric Patients
Exposures (AUC) of elvitegravir and tenofovir in 14 pediatric subjects aged 12 to less than 18 years who received STRIBILD in Study 112 were increased by 30% and 37%, respectively, compared with exposures achieved in adults following administration of STRIBILD, but were deemed acceptable based on the overall safety profile of these agents and exposure-safety assessments. The other components of STRIBILD had similar exposures in adolescents compared with adults [see Use in Specific Populations (8.4)].
Emtricitabine has been studied in pediatric subjects from 3 months to 17 years of age. Tenofovir DF has been studied in pediatric subjects from 2 years to less than 18 years of age. The pharmacokinetics of elvitegravir or cobicistat in pediatric subjects less than 12 years of age have not been established [see Use in Specific Populations (8.4)].
Geriatric Patients
The pharmacokinetics of elvitegravir, cobicistat, emtricitabine, and tenofovir have not been fully evaluated in elderly (65 years of age and older) patients [see Use in Specific Populations (8.5)].
Assessment of Drug Interactions
[see Contraindications (4) and Drug Interactions (7)]
The drug-drug interaction studies described in Tables 9 and 10 were conducted with STRIBILD, elvitegravir (coadministered with cobicistat or RTV), or cobicistat administered alone.
As STRIBILD is indicated for use as a complete regimen for the treatment of HIV-1 infection and should not be administered with other antiretroviral medications, information regarding drug-drug interactions with other antiretroviral agents is not provided.
The effects of coadministered drugs on the exposure of elvitegravir are shown in Table 9. The effects of elvitegravir or cobicistat on the exposure of coadministered drugs are shown in Table 10. For information regarding clinical recommendations, [see Drug Interactions (7)].
Table 9 Drug Interactions: Changes in Pharmacokinetic Parameters for Elvitegravir in the Presence of the Coadministered Drug* Coadministered Drug Dose of Coadministered Drug Elvitegravir Dose (mg) Cobicistat or RTV Booster Dose (mg) N Mean Ratio of Elvitegravir Pharmacokinetic Parameters (90% CI);
No Effect=1.00Cmax AUC Cmin - * All interaction studies conducted in healthy volunteers.
- † Percent change of cobicistat PK parameters (90% CI) was 1.25 (1.18 to 1.32) for Cmax, 1.59 (1.49 to 1.70) for AUC, and 4.25 (3.47 to 5.22) for Cmin.
Antacids 20 mL single dose given 4 hours before elvitegravir 50 single dose RTV 100 single dose 8 0.95
(0.84, 1.07)0.96
(0.88, 1.04)1.04
(0.93, 1.17)20 mL single dose given 4 hours after elvitegravir 10 0.98
(0.88, 1.10)0.98
(0.91, 1.06)1.00
(0.90, 1.11)20 mL single dose given 2 hours before elvitegravir 11 0.82
(0.74, 0.91)0.85
(0.79, 0.91)0.90
(0.82, 0.99)20 mL single dose given 2 hours after elvitegravir 10 0.79
(0.71, 0.88)0.80
(0.75, 0.86)0.80
(0.73, 0.89)Carbamazepine 200 mg twice daily 150 once daily Cobicistat 150 once daily 12 0.55
(0.49, 0.61)0.31
(0.28, 0.33)0.03
(0.02, 0.04)Famotidine 40 mg once daily given 12 hours after elvitegravir 150 once daily Cobicistat 150 once daily 10 1.02
(0.89, 1.17)1.03
(0.95, 1.13)1.18
(1.05, 1.32)40 mg once daily given simultaneously with elvitegravir 16 1.00
(0.92, 1.10)1.03
(0.98, 1.08)1.07
(0.98, 1.17)Ketoconazole 200 mg twice daily 150 once daily RTV 100 once daily 18 1.17
(1.04, 1.33)1.48
(1.36, 1.62)1.67
(1.48, 1.88)Ledipasvir/Sofosbuvir 90/400 mg once daily 150 once daily Cobicistat 150 once daily† 29 0.88
(0.82, 0.95)1.02
(0.95, 1.09)1.36
(1.23, 1.49)Omeprazole 40 mg once daily given 2 hours before elvitegravir 50 once daily RTV 100 once daily 9 0.93
(0.83, 1.04)0.99
(0.91, 1.07)0.94
(0.85, 1.04)20 mg once daily given 2 hours before elvitegravir 150 once daily Cobicistat 150 once daily 11 1.16
(1.04, 1.30)1.10
(1.02, 1.19)1.13
(0.96, 1.34)20 mg once daily given 12 hours after elvitegravir 11 1.03
(0.92, 1.15)1.05
(0.93, 1.18)1.10
(0.92, 1.32)Rifabutin 150 mg once every other day 150 once daily Cobicistat 150 once daily 12 0.91
(0.84, 0.99)0.79
(0.74, 0.85)0.33
(0.27, 0.40)Rosuvastatin 10 mg single dose 150 once daily Cobicistat 150 once daily 10 0.94
(0.83, 1.07)1.02
(0.91, 1.14)0.98
(0.83, 1.16)Table 10 Drug Interactions: Changes in Pharmacokinetic Parameters for Coadministered Drug in the Presence of Elvitegravir plus Cobicistat, Cobicistat, or STRIBILD* Coadministered Drug Dose of Coadministered Drug (mg) Elvitegravir Dose† (mg) Cobicistat Booster Dose (mg) N Mean Ratio of Coadministered Drug Pharmacokinetic Parameters‡ (90% CI);
No Effect=1.00Cmax AUC Cmin - * All interaction studies conducted in healthy volunteers.
- † NA=Not Applicable
- ‡ NC=Not Calculated
- § The predominant circulating nucleoside metabolite of sofosbuvir.
- ¶ Study conducted with STRIBILD.
- # Comparison based on rifabutin 300 mg once daily.
Buprenorphine 16–24 once daily 150 once daily 150 once daily 17 1.12
(0.98, 1.27)1.35
(1.18, 1.55)1.66
(1.43, 1.93)Norbuprenorphine 1.24
(1.03, 1.49)1.42
(1.22, 1.67)1.57
(1.31, 1.88)Carbamazepine 200 twice daily 150 once daily 150 once daily 12 1.40
(1.32, 1.49)1.43
(1.36, 1.52)1.51
(1.41, 1.62)Carbamazepine-10,11-epoxide 0.73
(0.70, 0.78)0.65
(0.63, 0.66)0.59
(0.57, 0.61)Desipramine 50 single dose NA 150 once daily 8 1.24
(1.08, 1.44)1.65
(1.36, 2.02)NC Digoxin 0.5 single dose NA 150 once daily 22 1.41
(1.29, 1.55)1.08
(1.00, 1.17)NC Ledipasvir 90/400 once daily 150 once daily Cobicistat 150 once daily 29 1.63
(1.51, 1.75)1.78
(1.64, 1.94)1.91
(1.76, 2.08)Sofosbuvir 1.33
(1.14, 1.56)1.36
(1.21, 1.52)NA GS-331007§ 1.33
(1.22, 1.44)1.44
(1.41, 1.48)1.53
(1.47, 1.59)Naloxone 4–6 once daily 150 once daily 150 once daily 17 0.72
(0.61, 0.85)0.72
(0.59, 0.87)NA Norgestimate/ethinyl estradiol 0.180/0.215/0.250 norgestimate once daily 150 once daily¶ 150 once daily¶ 13 2.08
(2.00, 2.17)2.26
(2.15, 2.37)2.67
(2.43, 2.92)0.025 ethinyl estradiol once daily 0.94
(0.86, 1.04)0.75
(0.69, 0.81)0.56
(0.52, 0.61)R-Methadone 80−120 daily 150 once daily 150 once daily 11 1.01
(0.91, 1.13)1.07
(0.96, 1.19)1.10
(0.95, 1.28)S-Methadone 0.96
(0.87, 1.06)1.00
(0.89, 1.12)1.02
(0.89, 1.17)Rifabutin 150 once every other day 150 once daily 150 once daily 12 1.09
(0.98, 1.20)#0.92
(0.83, 1.03)#0.94
(0.85, 1.04)#25-O-desacetyl-rifabutin 12 4.84
(4.09, 5.74)#6.25
(5.08, 7.69)#4.94
(4.04, 6.04)#Rosuvastatin 10 single dose 150 once daily 150 single dose 10 1.89
(1.48, 2.42)1.38
(1.14, 1.67)NC 12.4 Microbiology
Mechanism of Action
Elvitegravir: Elvitegravir inhibits the strand transfer activity of HIV-1 integrase (integrase strand transfer inhibitor; INSTI), an HIV-1 encoded enzyme that is required for viral replication. Inhibition of integrase prevents the integration of HIV-1 DNA into host genomic DNA, blocking the formation of the HIV-1 provirus and propagation of the viral infection. Elvitegravir does not inhibit human topoisomerases I or II.
Cobicistat: Cobicistat is a selective, mechanism-based inhibitor of cytochromes P450 of the CYP3A subfamily. Inhibition of CYP3A-mediated metabolism by cobicistat enhances the systemic exposure of CYP3A substrates, such as elvitegravir, where bioavailability is limited and half-life is shortened by CYP3A-dependent metabolism.
Emtricitabine: Emtricitabine, a synthetic nucleoside analog of cytidine, is phosphorylated by cellular enzymes to form emtricitabine 5'-triphosphate. Emtricitabine 5'-triphosphate inhibits the activity of the HIV-1 RT by competing with the natural substrate deoxycytidine 5'-triphosphate and by being incorporated into nascent viral DNA, which results in chain termination. Emtricitabine 5'-triphosphate is a weak inhibitor of mammalian DNA polymerases α, β, ε, and mitochondrial DNA polymerase γ.
Tenofovir Disoproxil Fumarate: Tenofovir DF is an acyclic nucleoside phosphonate diester analog of adenosine monophosphate. Tenofovir DF requires initial diester hydrolysis for conversion to tenofovir and subsequent phosphorylations by cellular enzymes to form tenofovir diphosphate. Tenofovir diphosphate inhibits the activity of HIV-1 RT by competing with the natural substrate deoxyadenosine 5′-triphosphate and, after incorporation into DNA, by DNA chain termination. Tenofovir diphosphate is a weak inhibitor of mammalian DNA polymerases α, β, and mitochondrial DNA polymerase γ.
Antiviral Activity in Cell Culture
Elvitegravir, Cobicistat, Emtricitabine, and Tenofovir Disoproxil Fumarate: The triple combination of elvitegravir, emtricitabine, and tenofovir was not antagonistic in cell culture combination antiviral activity assays and was not affected by the addition of cobicistat.
Elvitegravir: The antiviral activity of elvitegravir against laboratory and clinical isolates of HIV-1 was assessed in T lymphoblastoid cell lines, monocyte/macrophage cells, and primary peripheral blood lymphocytes. The 50% effective concentrations (EC50) ranged from 0.02 to 1.7 nM. Elvitegravir displayed antiviral activity in cell culture against HIV-1 clades A, B, C, D, E, F, G, and O (EC50 values ranged from 0.1 to 1.3 nM) and activity against HIV-2 (EC50 value of 0.53 nM). Elvitegravir did not show inhibition of replication of HBV or HCV in cell culture.
Cobicistat: Cobicistat has no detectable antiviral activity in cell culture against HIV-1, HBV, or HCV and does not antagonize the antiviral activity of elvitegravir, emtricitabine, or tenofovir.
Emtricitabine: The antiviral activity of emtricitabine against laboratory and clinical isolates of HIV-1 was assessed in T lymphoblastoid cell lines, the MAGI-CCR5 cell line, and primary peripheral blood mononuclear cells. The EC50 values for emtricitabine were in the range of 0.0013–0.64 micromolar. Emtricitabine displayed antiviral activity in cell culture against HIV-1 clades A, B, C, D, E, F, and G (EC50 values ranged from 0.007–0.075 micromolar) and showed strain-specific activity against HIV-2 (EC50 values ranged from 0.007–1.5 micromolar).
Tenofovir Disoproxil Fumarate: The antiviral activity of tenofovir against laboratory and clinical isolates of HIV-1 was assessed in T lymphoblastoid cell lines, primary monocyte/macrophage cells and peripheral blood lymphocytes. The EC50 values for tenofovir were in the range of 0.04–8.5 micromolar. Tenofovir displayed antiviral activity in cell culture against HIV-1 clades A, B, C, D, E, F, G, and O (EC50 values ranged from 0.5–2.2 micromolar) and showed strain-specific activity against HIV-2 (EC50 values ranged from 1.6–5.5 micromolar).
Resistance
In Cell Culture
Elvitegravir: HIV-1 isolates with reduced susceptibility to elvitegravir have been selected in cell culture. Reduced susceptibility to elvitegravir was associated with the primary integrase substitutions T66A/I, E92G/Q, S147G, and Q148R. Additional integrase substitutions observed in cell-culture selection included D10E, S17N, H51Y, F121Y, S153F/Y, E157Q, D232N, R263K, and V281M.
Emtricitabine and Tenofovir Disoproxil Fumarate: HIV-1 isolates with reduced susceptibility to emtricitabine or tenofovir have been selected in cell culture. Reduced susceptibility to emtricitabine was associated with M184V/I substitutions in HIV-1 RT. HIV-1 isolates selected by tenofovir expressed a K65R substitution in HIV-1 RT and showed a 2–4 fold reduction in susceptibility to tenofovir.
In Clinical Studies
Elvitegravir: Development of substitutions T66A/I/K, E92G/Q, T97A, S147G, Q148H/K/R, and N155H in the HIV-1 integrase protein was primarily associated with resistance to elvitegravir. In addition to these primary elvitegravir resistance-associated substitutions, E92A, F121C/Y, P145S, Q146I/L/R, and N155S were also occasionally observed and were shown to confer reduced susceptibility to elvitegravir. In virus isolates harboring the observed primary elvitegravir resistance-associated substitutions, additional substitutions in integrase were detected including H51Y, L68I/V, G70R, V72A/N, I73V, Q95K/R, S119R, E138A/K, G140A/C/S, E157Q, K160N, E170A, S230R, and D232N.
Emtricitabine and Tenofovir Disoproxil Fumarate: HIV-1 isolates with reduced susceptibility to emtricitabine or tenofovir have been selected in subjects experiencing virologic failure in clinical trials. Genotypic analysis of these isolates identified the M184V/I and K65R amino acid substitutions in the viral RT, respectively.
Elvitegravir, Cobicistat, Emtricitabine, and Tenofovir Disoproxil Fumarate: In clinical trials of HIV-1-infected subjects with no antiretroviral treatment history, Studies 102 and 103 [see Clinical Studies (14)], by Week 144, the development of one or more primary substitutions associated with resistance to elvitegravir, emtricitabine, and/or tenofovir was observed in viruses from 51% (18/35) of the STRIBILD-treatment failure subjects with evaluable genotypic resistance data who received at least 8 weeks of STRIBILD and had HIV-1 RNA greater than or equal to 400 copies per mL at confirmed virologic failure, the end of each study year, or the time of early study drug discontinuation. The most common substitutions that emerged were M184V/I (N=17) in HIV-1 RT and the primary elvitegravir resistance-associated substitutions, E92Q (N=9), N155H (N=5), Q148R (N=3), T66I (N=2), and T97A (N=1) in integrase; K65R in RT was also detected (N=5). In virus isolates harboring the observed primary elvitegravir resistance substitutions, additional substitutions in integrase were detected including H51Y, L68I/V, G70R, I73V, G140C, S153A, E157Q, and G163R. The virus in all subjects with evaluable data for RT and IN and whose virus developed integrase substitutions associated with elvitegravir resistance (N=14) also developed the M184I/V RT substitutions, and had reduced susceptibility to both elvitegravir and emtricitabine. In phenotypic analyses, HIV-1 isolates expressing M184V/I RT substitutions showed reduced susceptibility to emtricitabine (42- to greater than 152-fold); those expressing the primary elvitegravir resistance-associated integrase substitutions showed reduced susceptibility to elvitegravir (4- to greater than 198-fold); and those expressing the K65R RT substitution showed reduced susceptibility to tenofovir (0.8- to 1.6-fold), compared to wild-type reference HIV-1.
There was an insufficient number of virologic failures with evaluable data (N=1) in clinical trials of virologically suppressed HIV-1-infected subjects with no history of virologic failure, studies 115 and 121, [see Clinical Studies (14)] to draw conclusions about the development of resistance.
Cross-Resistance
STRIBILD-treatment failure subject isolates exhibited varying degrees of cross-resistance within the INSTI and NRTI drug classes depending on the specific substitutions observed. These isolates remained susceptible to all NNRTIs and protease inhibitors.
Elvitegravir: Cross-resistance has been observed among INSTIs. Elvitegravir-resistant viruses showed varying degrees of cross-resistance in cell culture to raltegravir depending on the type and number of substitutions in HIV-1 integrase. Of the primary elvitegravir resistance-associated substitutions tested (T66A/I/K, E92G/Q, T97A, S147G, Q148H/K/R, and N155H), all but three (T66I, E92G, and S147G) conferred greater than 1.5-fold reduced susceptibility to raltegravir (above the biological cutoff for raltegravir) when introduced individually into a wild-type virus by site-directed mutagenesis. Of the primary raltegravir resistance-associated substitutions (Y143C/H/R, Q148H/K/R, and N155H), all but Y143C/H conferred greater than 2.5-fold reductions in susceptibility to elvitegravir (above the biological cutoff for elvitegravir).
Emtricitabine: Cross-resistance has been observed among NRTIs. Emtricitabine-resistant isolates harboring an M184V/I substitution in HIV-1 RT were cross-resistant to lamivudine. HIV-1 isolates containing the K65R RT substitution, selected in vivo by abacavir, didanosine, and tenofovir, demonstrated reduced susceptibility to inhibition by emtricitabine.
Tenofovir Disoproxil Fumarate: Cross-resistance has been observed among NRTIs. The K65R substitution in HIV-1 RT selected by tenofovir is also selected in some HIV-1-infected patients treated with abacavir or didanosine. HIV-1 isolates with the K65R substitution also showed reduced susceptibility to emtricitabine and lamivudine. Therefore, cross-resistance among these NRTIs may occur in patients whose virus harbors the K65R substitution. The K70E substitution selected clinically by tenofovir DF results in reduced susceptibility to abacavir, didanosine, emtricitabine, lamivudine, and tenofovir. HIV-1 isolates from patients (N=20) whose HIV-1 expressed a mean of 3 zidovudine-associated RT amino acid substitutions (M41L, D67N, K70R, L210W, T215Y/F, or K219Q/E/N) showed a 3.1-fold decrease in the susceptibility to tenofovir. Subjects whose virus expressed an L74V RT substitution without zidovudine resistance-associated substitutions (N=8) had reduced response to tenofovir DF. Limited data are available for patients whose virus expressed a Y115F substitution (N=3), Q151M substitution (N=2), or T69 insertion (N=4) in HIV-1 RT, all of whom had a reduced response in clinical trials.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Elvitegravir: Long-term carcinogenicity studies of elvitegravir were carried out in mice (104 weeks) and in rats for up to 88 weeks (males) and 90 weeks (females). No drug-related increases in tumor incidence were found in mice at doses up to 2000 mg per kg per day alone or in combination with 25 mg per kg per day RTV at exposures 3- and 14-fold, respectively, the human systemic exposure at the recommended daily dose of 150 mg. No drug-related increases in tumor incidence were found in rats at doses up to 2000 mg per kg per day at exposures 12- to 27-fold, respectively in male and female, the human systemic exposure.
Elvitegravir was not genotoxic in the reverse mutation bacterial test (Ames test) and the rat micronucleus assay. In an in vitro chromosomal aberration test, elvitegravir was negative with metabolic activation; however, an equivocal response was observed without activation.
Elvitegravir did not affect fertility in male and female rats at approximately 16- and 30-fold higher exposures (AUC), respectively, than in humans at the therapeutic 150 mg daily dose.
Fertility was normal in the offspring of rats exposed daily from before birth (in utero) through sexual maturity at daily exposures (AUC) of approximately 18-fold higher than human exposures at the recommended 150 mg daily dose.
Cobicistat: In a long-term carcinogenicity study in mice, no drug-related increases in tumor incidence were observed at doses up to 50 and 100 mg/kg/day (males and females, respectively). Cobicistat exposures at these doses were approximately 7 (male) and 16 (females) times, respectively, the human systemic exposure at the therapeutic daily dose. In a long-term carcinogenicity study of cobicistat in rats, an increased incidence of follicular cell adenomas and/or carcinomas in the thyroid gland was observed at doses of 25 and 50 mg/kg/day in males, and at 30 mg/kg/day in females. The follicular cell findings are considered to be rat-specific, secondary to hepatic microsomal enzyme induction and thyroid hormone imbalance, and are not relevant for humans. At the highest doses tested in the rat carcinogenicity study, systemic exposures were approximately 2 times the human systemic exposure at the therapeutic daily dose.
Cobicistat was not genotoxic in the reverse mutation bacterial test (Ames test), or the mouse lymphoma or rat micronucleus assays.
Cobicistat did not affect fertility in male or female rats at daily exposures (AUC) approximately 4-fold higher than human exposures at the recommended 150 mg daily dose.
Fertility was normal in the offspring of rats exposed daily from before birth (in utero) through sexual maturity at daily exposures (AUC) of approximately 1.2-fold higher than human exposures at the recommended 150 mg daily dose.
Emtricitabine: In long-term carcinogenicity studies of emtricitabine, no drug-related increases in tumor incidence were found in mice at doses up to 750 mg per kg per day (23 times the human systemic exposure at the therapeutic dose of 200 mg per day) or in rats at doses up to 600 mg per kg per day (28 times the human systemic exposure at the therapeutic dose).
Emtricitabine was not genotoxic in the reverse mutation bacterial test (Ames test), or the mouse lymphoma or mouse micronucleus assays.
Emtricitabine did not affect fertility in male rats at approximately 140-fold or in male and female mice at approximately 60-fold higher exposures (AUC) than in humans given the recommended 200 mg daily dose. Fertility was normal in the offspring of mice exposed daily from before birth (in utero) through sexual maturity at daily exposures (AUC) of approximately 60-fold higher than human exposures at the recommended 200 mg daily dose.
Tenofovir Disoproxil Fumarate: Long-term oral carcinogenicity studies of tenofovir DF in mice and rats were carried out at exposures up to approximately 10 times (mice) and 4 times (rats) those observed in humans at the therapeutic dose for HIV-1 infection. At the high dose in female mice, liver adenomas were increased at exposures 10 times of that in humans. In rats, the study was negative for carcinogenic findings at exposures up to 4 times that observed in humans at the therapeutic dose.
Tenofovir DF was mutagenic in the in vitro mouse lymphoma assay and negative in an in vitro bacterial mutagenicity test (Ames test). In an in vivo mouse micronucleus assay, tenofovir DF was negative when administered to male mice.
There were no effects on fertility, mating performance or early embryonic development when tenofovir DF was administered to male rats at a dose equivalent to 10 times the human dose based on body-surface-area comparisons for 28 days prior to mating and to female rats for 15 days prior to mating through day seven of gestation. There was, however, an alteration of the estrous cycle in female rats.
-
14 CLINICAL STUDIES
14.1 Description of Clinical Trials
The efficacy and safety of STRIBILD were evaluated in the studies summarized in Table 11.
Table 11 Trials Conducted with STRIBILD in Subjects with HIV-1 Infection Trial Population Study Arms (N)* Timepoint (Week) - * Randomized and dosed.
- † Randomized, double blind, active-controlled trial.
- ‡ Patients had estimated creatinine clearance greater than or equal to 70 mL/min at screening.
- § Randomized, open label, active-controlled trial.
- ¶ HIV-1 RNA less than 50 copies per mL.
- # Open label trial.
Study 102 †,‡ Adults with no antiretroviral treatment history STRIBILD (348)
ATRIPLA (352)144 Study 103 †,‡ STRIBILD (353)
TRUVADA+atazanavir+ritonavir (355)Study 115‡,§ Virologically suppressed adults without a history of virologic failure¶ STRIBILD (293)
TRUVADA+PI+ritonavir (140)48 Study 121‡,§ STRIBILD (291)
TRUVADA+NNRTI (143)Study 112# Treatment-naïve adolescents between the ages of 12 to less than 18 years STRIBILD (50) 48 14.2 Clinical Trial Results in HIV-1-Infected Adult Subjects with No Antiretroviral Treatment History
In Study 102, subjects were randomized in a 1:1 ratio to receive either STRIBILD (N=348) once daily or ATRIPLA (N=352) once daily. The mean age was 38 years (range, 18–67), 89% were male, 63% were White, 28% were Black, and 2% were Asian. Twenty-four percent of subjects identified as Hispanic/Latino. The mean baseline plasma HIV-1 RNA was 4.8 log10 copies per mL (range, 2.6–6.5). The mean baseline CD4+ cell count was 386 cells per mm3 (range, 3–1348), and 13% had CD4+ cell counts less than 200 cells per mm3. Thirty-three percent of subjects had baseline viral loads greater than 100,000 copies per mL.
In Study 103, subjects were randomized in a 1:1 ratio to receive either STRIBILD (N=353) once daily or ATV 300 mg + RTV 100 mg + TRUVADA (N=355) once daily. The mean age was 38 years (range, 19–72), 90% were male, 74% were White, 17% were Black, and 5% were Asian. Sixteen percent of subjects identified as Hispanic/Latino. The mean baseline plasma HIV-1 RNA was 4.8 log10 copies per mL (range, 1.7–6.6). The mean baseline CD4+ cell count was 370 cells per mm3 (range, 5–1132), and 13% had CD4+ cell count less than 200 cells per mm3. Forty-one percent of subjects had baseline viral loads greater than 100,000 copies per mL.
In both studies, subjects were stratified by baseline HIV-1 RNA (less than or equal to 100,000 copies per mL or greater than 100,000 copies per mL).
Treatment outcomes of Study 102 and Study 103 through 144 weeks are presented in Table 12.
Table 12 Virologic Outcome of Randomized Treatment of Study 102 and Study 103 at Week 144* Study 102 Study 103 STRIBILD
N=348ATRIPLA
N=352STRIBILD
N=353ATV+RTV+TRUVADA
N=355- * Week-144 window is between Day 967 and 1050 (inclusive).
- † Includes subjects who had ≥50 copies/mL in the Week-144 window, subjects who discontinued early due to lack or loss of efficacy, subjects who discontinued for reasons other than an adverse event, death, or lack or loss of efficacy and at the time of discontinuation had a viral value of ≥50 copies/mL.
- ‡ Includes patients who discontinued due to an adverse event or death at any time point from Day 1 through the time window if this resulted in no virologic data on treatment during the specified window.
- § Includes subjects who discontinued for reasons other than an adverse event, death, or lack or loss of efficacy, e.g., withdrew consent, loss to follow-up, etc.
Virologic Success
HIV-1 RNA <50 copies/mL80% 75% 78% 75% Treatment Difference 4.9% (95% CI = −1.3%, 11.1%) 3.1% (95% CI = −3.2%, 9.4%) Virologic Failure† 7% 10% 8% 7% No Virologic Data in Week 144 Window Discontinued Study Drug Due to AE or Death‡ 6% 8% 6% 8% Discontinued Study Drug Due to Other Reasons and Last Available HIV-1 RNA <50 copies/mL§ 5% 7% 8% 9% Missing Data During Window but on Study Drug 1% 0% 1% 1% In Study 102, the mean increase from baseline in CD4+ cell count at Week 144 was 298 cells per mm3 in the STRIBILD-treated subjects and 272 cells per mm3 in the ATRIPLA -treated subjects. In Study 103, the mean increase from baseline in CD4+ cell count at Week 144 was 261 cells per mm3 in the STRIBILD-treated subjects and 269 cells per mm3 in the ATV+RTV+TRUVADA-treated subjects.
14.3 Clinical Trial Results in Virologically Suppressed HIV-1-Infected Adult Subjects with No History of Virologic Failure
In Study 115, subjects had to be on either their first or second antiretroviral regimen with no history of virologic failure, with no current or past history of resistance to the antiretroviral components of STRIBILD, and must have been suppressed (HIV-1 RNA <50 copies/mL) on a ritonavir-boosted PI in combination with TRUVADA for at least 6 months prior to screening. Subjects were randomized in a 2:1 ratio to either switch to STRIBILD (STRIBILD arm, N=293; randomized and dosed) or stay on their baseline antiretroviral regimen for 48 weeks (PI+RTV+TRUVADA arm, N=140; randomized and dosed). Subjects had a mean age of 41 years (range, 21–76), 86% were male, 80% were White, and 15% were Black. The mean baseline CD4+ cell count was 610 cells per mm3 (range, 74–1919). At screening subjects were receiving atazanavir (40%), darunavir (40%), lopinavir (17%), fosamprenavir (3%), or saquinavir (<1%) as the PI in their regimen.
In Study 121, subjects had to be on either their first or second antiretroviral regimen with no history of virologic failure, with no current or past history of resistance to the antiretroviral components of STRIBILD, and must have been suppressed (HIV-1 RNA <50 copies/mL) on a NNRTI in combination with TRUVADA for at least 6 months prior to screening. Subjects were randomized in a 2:1 ratio to either switch to STRIBILD (STRIBILD arm, N=291; randomized and dosed) or stay on their baseline antiretroviral regimen for 48 weeks (NNRTI+TRUVADA arm, N=143; randomized and dosed). Subjects had a mean age of 41 years (range, 20–72), 93% were male, 78% were White, and 17% were Black. The mean baseline CD4+ cell count was 588 cells per mm3 (range, 100–1614). Randomization was stratified by use of efavirenz in the baseline regimen. At screening subjects were receiving efavirenz (78%) (predominantly as ATRIPLA [74%]), nevirapine (17%), rilpivirine (4%) (as COMPLERA [4%]), or etravirine (1%) as the NNRTI in their regimen.
Virologic outcomes of Study 115 and Study 121 are presented in Table 13. Five treated subjects were excluded from the efficacy analysis: in Study 115, three STRIBILD subjects had protocol-prohibited documented resistance and one PI+RTV+TRUVADA subject was not on a protease inhibitor-based regimen at screening; in Study 121, one STRIBILD subject had protocol-prohibited documented resistance.
Table 13 Virologic Outcomes of Randomized Treatment in Study 115 and Study 121 at Week 48 Study GS-US-236-0115* Study GS-US-236-0121* STRIBILD
N=290PI+RTV+TRUVADA
N=139STRIBILD
N=290NNRTI+TRUVADA
N=143- * Week-48 window is between Day 295 and 378 (inclusive).
- † Includes subjects who had ≥50 copies/mL in the Week-48 window, subjects who discontinued early due to lack or loss of efficacy, subjects who discontinued for reasons other than an adverse event, death, or lack or loss of efficacy and at the time of discontinuation had a viral value of ≥50 copies/mL.
- ‡ Includes subjects who discontinued due to an adverse event or death at any time point from Day 1 through the time window if this resulted in no virologic data on treatment during the specified window.
- § Includes subjects who discontinued for reasons other than an adverse event, death, or lack or loss of efficacy, e.g., withdrew consent, loss to follow-up, etc.
Virologic Success HIV-1 RNA <50 copies/mL 94% 87% 93% 88% Virologic Failure† 1% 1% 1% 1% No Virologic Data in Week 48 Window 6% 12% 6% 11% Discontinued Study Drug Due to AE or Death‡ 2% 1% 2% 1% Discontinued Study Drug Due to Other Reasons and Last Available HIV-1 RNA <50 copies/mL§ 4% 10% 4% 9% Missing Data During Window but on Study Drug 0% 0% 0% 1% 14.4 Clinical Trial Results in HIV-1Treatment-Naïve Adolescent Subjects Aged 12 to Less than 18 Years
In Study 112, the efficacy, safety, and pharmacokinetics of STRIBILD were evaluated in a single group, open-label trial in HIV-1-infected treatment-naïve adolescents aged 12 to less than 18 years of age and weighing at least 35 kg (77 lbs) (N=50). Mean age was 15 years (range, 12–17); 70% were male, 68% black, and 28% Asian. At baseline, mean plasma HIV-1 RNA was 4.60 log10 copies per mL (range, 3.18–5.73), mean CD4+ cell count was 399 cells per mm3 (range, 133–734), and mean CD4+ percentage was 20.9% (range, 4.5%–41.1%). Twenty percent had baseline plasma HIV-1 RNA >100,000 copies per mL.
At Week 48, 44 of 50 (88%) adolescent patients treated with STRIBILD achieved HIV-1 RNA <50 copies per mL and 4 had HIV-1 RNA ≥50 copies per mL; 1 patient discontinued study drug; 1 had no virologic data at Week 48. The mean decrease from baseline in HIV-1 RNA was –3.16 log10 copies per mL; mean increase from baseline in CD4+ cell count was 229 cells per mm3. No emergent resistance to STRIBILD was detected through Week 48.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
STRIBILD tablets are green, capsule shaped, film coated, and debossed with "GSI" on one side and the number "1" surrounded by a square box (
 ) on the other side. Each bottle contains 30 tablets (NDC: 61958-1201-1) and a silica gel desiccant, and is closed with a child-resistant closure.
) on the other side. Each bottle contains 30 tablets (NDC: 61958-1201-1) and a silica gel desiccant, and is closed with a child-resistant closure.NDC 69189-1201-1 single dose pack with 1 tablet as repackaged by Avera McKennan Hospital
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Drug Interactions
Advise patients that STRIBILD may interact with many drugs; therefore, advise patients to report to their healthcare provider the use of any other prescription or nonprescription medication or herbal products, including St. John's wort [see Contraindications (4), Warnings and Precautions (5.4) and Drug Interactions (7)].
Lactic Acidosis/Severe Hepatomegaly with Steatosis
Inform patients that lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported. Advise patients that treatment with STRIBILD should be suspended if they develop clinical symptoms suggestive of lactic acidosis or pronounced hepatotoxicity (including nausea, vomiting, unusual or unexpected stomach discomfort, and weakness) [see Warnings and Precautions (5.1)].
Severe Acute Exacerbation of Hepatitis B in Patients Coinfected with HIV-1 and HBV
Inform patients that hepatitis B testing is recommended prior to initiating antiretroviral therapy. Severe acute exacerbations of hepatitis B have been reported in patients who are coinfected with HBV and HIV-1 and have discontinued emtricitabine or tenofovir DF [see Warnings and Precautions (5.2)].
New Onset or Worsening Renal Impairment
Inform patients that renal impairment, including cases of acute renal failure and Fanconi syndrome, has been reported in association with the use of STRIBILD. Advise patients to avoid STRIBILD with concurrent or recent use of a nephrotoxic agent (e.g., high-dose or multiple NSAIDs) [see Warnings and Precautions (5.3)].
Bone Loss and Mineralization Defects
Inform patients that decreases in bone mineral density have been observed with the use of STRIBILD. Assessment of bone mineral density (BMD) should be considered in patients who have a history of pathologic bone fracture or other risk factors for osteoporosis or bone loss [see Warnings and Precautions (5.5)].
Fat Redistribution
Inform patients that redistribution or accumulation of body fat may occur in patients receiving antiretroviral therapy, and that the cause and long-term health effects of these conditions are not known [see Warnings and Precautions (5.6)].
Immune Reconstitution Syndrome
Inform patients that in some patients with advanced HIV infection (AIDS), signs and symptoms of inflammation from previous infections may occur soon after anti-HIV treatment is started. It is believed that these symptoms are due to an improvement in the body's immune response, enabling the body to fight infections that may have been present with no obvious symptoms. Advise patients to inform their healthcare provider immediately of any symptoms of infection [see Warnings and Precautions (5.7)].
Missed Dosage
Inform patients that it is important to take STRIBILD on a regular dosing schedule with food and to avoid missing doses as it can result in development of resistance [see Dosage and Administration (2.2)].
Pregnancy Registry
Inform patients that there is an antiretroviral pregnancy registry to monitor fetal outcomes of pregnant women exposed to STRIBILD [see Use in Specific Populations (8.1)].
Lactation
Instruct women with HIV-1 infection not to breastfeed because HIV-1 can be passed to the baby in breast milk [see Use in Specific Populations (8.2)].
-
SPL UNCLASSIFIED SECTION
© 2017 Gilead Sciences, Inc. All rights reserved.
COMPLERA, EMTRIVA, GSI, STRIBILD, TRUVADA, VITEKTA, and VIREAD are trademarks of Gilead Sciences, Inc., or its related companies. ATRIPLA is a trademark of Bristol-Myers Squibb & Gilead Sciences, LLC. All other trademarks referenced herein are the property of their respective owners.
-
Patient Information
STRIBILD® (STRY-bild)
(elvitegravir, cobicistat, emtricitabine,
and tenofovir disoproxil fumarate)
TabletsImportant: Ask your healthcare provider or pharmacist about medicines that should not be taken with STRIBILD. For more information, see the section "What should I tell my healthcare provider before taking STRIBILD?"
Read this Patient Information before you start taking STRIBILD and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment.
What is the most important information I should know about STRIBILD?
STRIBILD can cause serious side effects, including:
- 1.
Build-up of lactic acid in your blood (lactic acidosis). Lactic acidosis can happen in some people who take STRIBILD or similar (nucleoside analogs) medicines. Lactic acidosis is a serious medical emergency that can lead to death.
Lactic acidosis can be hard to identify early, because the symptoms could seem like symptoms of other health problems. Call your healthcare provider right away if you get any of the following symptoms which could be signs of lactic acidosis:
- feel very weak or tired
- have unusual (not normal) muscle pain
- have trouble breathing
- have stomach pain with
- nausea
- vomiting
- feel cold, especially in your arms and legs
- feel dizzy or lightheaded
- have a fast or irregular heartbeat
- 2. Severe liver problems. Severe liver problems can happen in people who take STRIBILD. In some cases, these liver problems can lead to death. Your liver may become large (hepatomegaly) and you may develop fat in your liver (steatosis).
Call your healthcare provider right away if you get any of the following symptoms of liver problems:
- your skin or the white part of your eyes turns yellow (jaundice)
- dark "tea-colored" urine
- light-colored bowel movements (stools)
- loss of appetite for several days or longer
- nausea
- stomach pain
You may be more likely to get lactic acidosis or severe liver problems if you are female, very overweight (obese), or have been taking STRIBILD for a long time.
- 3. Worsening of Hepatitis B infection. If you have hepatitis B virus (HBV) infection and take STRIBILD, your HBV may get worse (flare-up) if you stop taking STRIBILD. A "flare-up" is when your HBV infection suddenly returns in a worse way than before.
- Do not run out of STRIBILD. Refill your prescription or talk to your healthcare provider before your STRIBILD is all gone.
- Do not stop taking STRIBILD without first talking to your healthcare provider.
- If you stop taking STRIBILD, your healthcare provider will need to check your health often and do blood tests regularly for several months to check your HBV infection. Tell your healthcare provider about any new or unusual symptoms you may have after you stop taking STRIBILD.
For more information about side effects, see the section "What are the possible side effects of STRIBILD?"
What is STRIBILD?
STRIBILD is a prescription medicine that is used without other antiretroviral medicines to treat Human Immunodeficiency Virus-1 (HIV-1) in people 12 years of age and older:
- who have not received anti-HIV-1 medicines in the past, or
- to replace their current anti-HIV-1 medicines:
- in people who have been on the same anti-HIV-1 medicine regimen for at least 6 months, and
- who have an amount of HIV-1 in their blood (this is called "viral load") that is less than 50 copies/mL, and
- have never failed past HIV-1 treatment.
HIV-1 is the virus that causes AIDS (Acquired Immune Deficiency Syndrome).
STRIBILD contains the prescription medicines elvitegravir (VITEKTA®), cobicistat (TYBOST®), emtricitabine (EMTRIVA®) and tenofovir disoproxil fumarate (VIREAD®).
It is not known if STRIBILD is safe and effective in children under 12 years of age or who weigh less than 77 lbs.
Who should not take STRIBILD?
Do not take STRIBILD if you also take a medicine that contains:
- alfuzosin hydrochloride (UROXATRAL®)
- cisapride (PROPULSID®, PROPULSID QUICKSOLV®)
- carbamazepine (CARBATROL®, EPITOL®, EQUETRO®, TEGRETOL®, TEGRETOL-XR®, TERIL®)
- ergot-containing medicines, including:
- dihydroergotamine mesylate (D.H.E. 45®, MIGRANAL®)
- ergotamine tartrate (CAFERGOT®, MIGERGOT®, ERGOSTAT®, MEDIHALER ERGOTAMINE®, WIGRAINE®, WIGRETTES®)
- methylergonovine maleate (ERGOTRATE®, METHERGINE®)
- lovastatin (ADVICOR®, ALTOPREV®, MEVACOR®)
- lurasidone (LATUDA®)
- midazolam, when taken by mouth
- phenobarbital (LUMINAL®)
- phenytoin (DILANTIN®, PHENYTEK®)
- pimozide (ORAP®)
- rifampin (RIFADIN®, RIFAMATE®, RIFATER®, RIMACTANE®)
- sildenafil (REVATIO®), when used for treating the lung problem, pulmonary arterial hypertension (PAH)
- simvastatin (SIMCOR®, VYTORIN®, ZOCOR®)
- triazolam (HALCION®)
- St. John's wort (Hypericum perforatum) or a product that contains St. John's wort
What should I tell my healthcare provider before taking STRIBILD?
Before taking STRIBILD, tell your healthcare provider if you:
- have liver problems including hepatitis B infection
- have kidney problems
- have bone problems
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if STRIBILD can harm your unborn baby. Tell your healthcare provider if you become pregnant while taking STRIBILD.
Pregnancy Registry. There is a pregnancy registry for women who take antiviral medicines during pregnancy. The purpose of this registry is to collect information about the health of you and your baby. Talk with your healthcare provider about how you can take part in this registry.
- are breastfeeding or plan to breastfeed. Do not breastfeed if you take STRIBILD.
- You should not breastfeed if you have HIV-1 because of the risk of passing HIV-1 to your baby.
- At least two of the medicines in STRIBILD can pass to your baby in your breast milk. It is not known if the other medicines in STRIBILD can pass into your breast milk.
Talk with your healthcare provider about the best way to feed your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Some medicines may interact with STRIBILD. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.
- You can ask your healthcare provider or pharmacist for a list of medicines that interact with STRIBILD.
- Do not start a new medicine without telling your healthcare provider. Your healthcare provider can tell you if it is safe to take STRIBILD with other medicines.
How should I take STRIBILD?
- Take STRIBILD exactly as your healthcare provider tells you to take it. STRIBILD is taken by itself (not with other anti-HIV medicines) to treat HIV-1 infection.
- STRIBILD is usually taken 1 time each day.
- Take STRIBILD with food.
- Do not change your dose or stop taking STRIBILD without first talking with your healthcare provider. Stay under a healthcare provider's care when taking STRIBILD.
- If you need to take a medicine for indigestion (antacid) that contains aluminum and magnesium hydroxide or calcium carbonate during treatment with STRIBILD, take it at least 2 hours before or after you take STRIBILD.
- Do not miss a dose of STRIBILD. If you miss a dose of STRIBILD, take the missed dose as soon as you remember. If it is almost time for your next dose of STRIBILD, do not take the missed dose. Take the next dose of STRIBILD at your regular time. Do not take 2 doses at the same time to make up for a missed dose.
- If you take too much STRIBILD, call your healthcare provider or go to the nearest hospital emergency room right away.
- When your STRIBILD supply starts to run low, get more from your healthcare provider or pharmacy. This is very important because the amount of virus in your blood may increase if the medicine is stopped for even a short time. The virus may develop resistance to STRIBILD and become harder to treat.
What are the possible side effects of STRIBILD?
STRIBILD may cause the following serious side effects, including:
- See "What is the most important information I should know about STRIBILD?"
- New or worse kidney problems, including kidney failure. Your healthcare provider should do blood and urine tests to check your kidneys before you start and while you are taking STRIBILD. Your healthcare provider may tell you to stop taking STRIBILD if you develop new or worse kidney problems.
- Bone problems can happen in some people who take STRIBILD. Bone problems include bone pain, softening, or thinning (which may lead to fractures). Your healthcare provider may need to do tests to check your bones.
- Changes in body fat can happen in people who take HIV-1 medicine. These changes may include increased amount of fat in the upper back and neck ("buffalo hump"), breast, and around the middle of your body (trunk). Loss of fat from the legs, arms, and face may also happen. The exact cause and long-term health effects of these conditions are not known.
- Changes in your immune system (Immune Reconstitution Syndrome) can happen when you start taking HIV-1 medicines. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Tell your healthcare provider right away if you start having any new symptoms after starting your HIV-1 medicine.
The most common side effects of STRIBILD include:
- nausea
- diarrhea
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of STRIBILD. For more information, ask your healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store STRIBILD?
- Store STRIBILD at room temperature between 68 °F to 77 °F (20 °C to 25 °C).
- Keep STRIBILD in its original container.
- Keep the container tightly closed.
- Do not use STRIBILD if the seal over the bottle opening is broken or missing.
Keep STRIBILD and all medicines out of reach of children.
General information about STRIBILD.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use STRIBILD for a condition for which it was not prescribed. Do not give STRIBILD to other people, even if they have the same symptoms you have. It may harm them.
This leaflet summarizes the most important information about STRIBILD. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about STRIBILD that is written for health professionals.
For more information, call 1-800-445-3235 or go to www.STRIBILD.com.
What are the ingredients in STRIBILD?
Active ingredients: elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate
Inactive ingredients: lactose monohydrate, microcrystalline cellulose, silicon dioxide, croscarmellose sodium, hydroxypropyl cellulose, sodium lauryl sulfate, and magnesium stearate. The tablets are film coated with a coating material containing indigo carmine (FD&C blue #2) aluminum lake, polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide, and yellow iron oxide.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Manufactured and distributed by:
Gilead Sciences, Inc.
Foster City, CA 94404Revised: 01/2017
© 2017 Gilead Sciences, Inc. All rights reserved.
COMPLERA, EMTRIVA, STRIBILD, TRUVADA, TYBOST, VIREAD, and VITEKTA are trademarks of Gilead Sciences, Inc., or its related companies. ATRIPLA is a trademark of Bristol-Myers Squibb & Gilead Sciences, LLC. All other trademarks referenced herein are the property of their respective owners.
203100-GS-008
- 1.
Build-up of lactic acid in your blood (lactic acidosis). Lactic acidosis can happen in some people who take STRIBILD or similar (nucleoside analogs) medicines. Lactic acidosis is a serious medical emergency that can lead to death.
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
STRIBILD
elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69189-1201(NDC:61958-1201) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength elvitegravir (UNII: 4GDQ854U53) (elvitegravir - UNII:4GDQ854U53) elvitegravir 150 mg cobicistat (UNII: LW2E03M5PG) (cobicistat - UNII:LW2E03M5PG) cobicistat 150 mg emtricitabine (UNII: G70B4ETF4S) (emtricitabine - UNII:G70B4ETF4S) emtricitabine 200 mg tenofovir disoproxil fumarate (UNII: OTT9J7900I) (tenofovir anhydrous - UNII:W4HFE001U5) tenofovir disoproxil fumarate 300 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) HYDROXYPROPYL CELLULOSE (1200000 MW) (UNII: RFW2ET671P) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) ALUMINUM OXIDE (UNII: LMI26O6933) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color GREEN Score no score Shape OVAL (Capsule shape) Size 20mm Flavor Imprint Code GSI;1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69189-1201-1 1 in 1 DOSE PACK; Type 0: Not a Combination Product 10/28/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA203100 10/28/2015 Labeler - Avera McKennan Hospital (068647668) Establishment Name Address ID/FEI Business Operations Avera McKennan Hospital 068647668 relabel(69189-1201) , repack(69189-1201)
Trademark Results [Stribild]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 STRIBILD 85688699 4320307 Live/Registered |
GILEAD SCIENCES IRELAND UC 2012-07-27 |
 STRIBILD 85555217 4263613 Live/Registered |
GILEAD SCIENCES IRELAND UC 2012-02-28 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
