IMFINZI- durvalumab injection, solution
IMFINZI by
Drug Labeling and Warnings
IMFINZI by is a Prescription medication manufactured, distributed, or labeled by AstraZeneca Pharmaceuticals LP, AstraZeneca PLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use IMFINZI safely and effectively. See full prescribing information for IMFINZI.
IMFINZI® (durvalumab) injection, for intravenous use
Initial U.S. Approval: 2017RECENT MAJOR CHANGES
INDICATIONS AND USAGE
IMFINZI is a programmed death-ligand 1 (PD-L1) blocking antibody indicated:
- for the treatment of adult patients with locally advanced or metastatic urothelial carcinoma who:
This indication is approved under accelerated approval based on tumor response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials. (1.1)
- for the treatment of adult patients with unresectable, Stage III non-small cell lung cancer (NSCLC) whose disease has not progressed following concurrent platinum-based chemotherapy and radiation therapy. (1.2)
- in combination with etoposide and either carboplatin or cisplatin, as first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC). (1.3)
DOSAGE AND ADMINISTRATION
- Administer IMFINZI as an intravenous infusion over 60 minutes. (2.3)
- Urothelial Carcinoma: 10 mg/kg every 2 weeks. (2.1)
- Stage III NSCLC: 10 mg/kg every 2 weeks. (2.1)
- ES-SCLC: when administered with etoposide and either carboplatin or cisplatin, administer IMFINZI 1500 mg every 3 weeks prior to chemotherapy and then every 4 weeks as a single agent. (2.1)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Immune-Mediated Pneumonitis: Withhold for moderate and permanently discontinue for severe or life-threatening pneumonitis. (2.3, 5.1)
- Immune-Mediated Hepatitis: Monitor for changes in liver function. Withhold for moderate and permanently discontinue for severe or life-threatening transaminase or total bilirubin elevation. (2.3, 5.2)
- Immune-Mediated Colitis: Withhold for moderate and permanently discontinue for severe or life-threatening colitis. (2.3, 5.3)
- Immune-Mediated Endocrinopathies: Withhold for moderate, severe, or life-threatening. (2.3, 5.4)
- Immune-Mediated Nephritis: Monitor for changes in renal function. Withhold for moderate and permanently discontinue for severe or life-threatening nephritis. (2.3, 5.5)
- Immune-Mediated Dermatologic Reactions: Withhold for moderate and permanently discontinue for severe or life-threatening rash. (2.3, 5.6)
- Infection: Withhold for severe or life-threatening infection. (2.3, 5.8)
- Infusion-Related Reactions: Interrupt or slow the rate of infusion for mild or moderate and permanently discontinue for severe or life-threatening infusion-related reactions. (2.3, 5.9)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. (5.10, 8.1, 8.3)
ADVERSE REACTIONS
- Most common adverse reactions (≥15% of patients with urothelial carcinoma) were fatigue, musculoskeletal pain, constipation, decreased appetite, nausea, peripheral edema, and urinary tract infection. (6.1)
- Most common adverse reactions (≥20% of patients with unresectable, Stage III NSCLC) were cough, fatigue, pneumonitis/radiation pneumonitis, upper respiratory tract infections, dyspnea, and rash. (6.1)
- Most common adverse reactions (≥20% of patients with extensive-stage SCLC) were, nausea, fatigue/asthenia, alopecia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact AstraZeneca at 1-800-236-9933 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Lactation: Advise not to breastfeed. (8.2)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 3/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Urothelial Carcinoma
1.2 Non-Small Cell Lung Cancer
1.3 Small Cell Lung Cancer
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Dosage Modifications for Adverse Reactions
2.3 Preparation and Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Immune-Mediated Pneumonitis
5.2 Immune-Mediated Hepatitis
5.3 Immune-Mediated Colitis
5.4 Immune-Mediated Endocrinopathies
5.5 Immune-Mediated Nephritis
5.6 Immune-Mediated Dermatologic Reactions
5.7 Other Immune-Mediated Adverse Reactions
5.8 Infection
5.9 Infusion-Related Reactions
5.10 Embryo-Fetal Toxicity
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Immunogenicity
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Urothelial Carcinoma
14.2 Non-Small Cell Lung Cancer (NSCLC)
14.3 Small Cell Lung Cancer (SCLC)
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Urothelial Carcinoma
IMFINZI is indicated for the treatment of adult patients with locally advanced or metastatic urothelial carcinoma who:
- have disease progression during or following platinum-containing chemotherapy.
- have disease progression within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy.
This indication is approved under accelerated approval based on tumor response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials [see Clinical Studies (14.1)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dosages for IMFINZI as a single agent and IMFINZI in combination with chemotherapy are presented in Table 1 [see Clinical Studies (14)].
IMFINZI is administered as an intravenous infusion over 60 minutes.
Table 1. Recommended Dosages of IMFINZI - * Patients with a body weight of 30 kg or less must receive weight-based dosing, equivalent to IMFINZI 20 mg/kg in combination with chemotherapy every 3 weeks (21 days) for 4 cycles, followed by 20 mg/kg every 4 weeks as a single agent until weight increases to greater than 30 kg.
- † Administer IMFINZI prior to chemotherapy on the same day. When IMFINZI is administered in combination with chemotherapy, refer to the Prescribing Information for etoposide and carboplatin or cisplatin for dosing information.
Indication
Recommended IMFINZI dosage
Duration of Therapy
Urothelial Carcinoma
10 mg/kg every 2 weeks
Until disease progression or unacceptable toxicity
Unresectable stage III NSCLC
10 mg/kg every 2 weeks
Until disease progression, unacceptable toxicity, or a maximum of 12 months
ES-SCLC
1500 mg* in combination with chemotherapy† every 3 weeks (21 days) for 4 cycles, followed by 1500 mg every 4 weeks as a single agent
Until disease progression or unacceptable toxicity
2.2 Dosage Modifications for Adverse Reactions
No dose reductions are recommended. Withhold or discontinue IMFINZI to manage adverse reactions as described in Table 2.
Table 2. Recommended Dosage Modifications for Adverse Reactions - * National Cancer Institute Common Terminology Criteria for Adverse Events, version 4.03. ALT: alanine aminotransferase; AST: aspartate aminotransferase; ULN: upper limit of normal.
Adverse Reaction
Severity*
Dosage Modification
Pneumonitis [see Warnings and Precautions (5.1)]
Grade 2
Withhold dose until Grade 1 or resolved and corticosteroid dose is less than or equal to prednisone 10 mg per day (or equivalent).
Grade 3 or 4
Permanently discontinue
Hepatitis [see Warnings and Precautions (5.2)]
For ALT or AST greater than 3 but less than or equal to 8 times the ULN
or
Total bilirubin greater than 1.5 but less than or equal to 5 times the ULN
Withhold dose until Grade 1 or resolved and corticosteroid dose is less than or equal to prednisone 10 mg per day (or equivalent).
ALT or AST greater than 8 times the ULN or total bilirubin greater than 5 times the ULN
or
Concurrent ALT or AST greater than 3 times the ULN and total bilirubin greater than 2 times the ULN with no other cause
Permanently discontinue
Colitis or diarrhea [see Warnings and Precautions (5.3)]
Grade 2
Withhold dose until Grade 1 or resolved and corticosteroid dose is less than or equal to prednisone 10 mg per day (or equivalent).
Grade 3 or 4
Permanently discontinue
Hyperthyroidism or thyroiditis [see Warnings and Precautions (5.4)]
Grade 2-4
Withhold dose until clinically stable
Adrenal insufficiency or Hypophysitis/Hypopituitarism [see Warnings and Precautions (5.4)]
Grade 2-4
Withhold dose until clinically stable
Type 1 Diabetes
Mellitus [see Warnings and Precautions (5.4)]
Grade 2-4
Withhold dose until clinically stable
Nephritis [see Warnings and Precautions (5.5)]
For Creatinine greater than 1.5 to 3 times the ULN
Withhold dose until Grade 1 or resolved and corticosteroid dose is less than or equal to prednisone 10 mg per day (or equivalent).
For Creatinine greater than 3 times the ULN
Permanently discontinue
Rash or dermatitis [see Warnings and Precautions (5.6)]
Grade 2 for longer than 1 week or Grade 3
Withhold dose until Grade 1 or resolved and corticosteroid dose is less than or equal to prednisone 10 mg per day (or equivalent).
Grade 4
Permanently discontinue
Infection [see Warnings and Precautions (5.8)]
Grade 3 or 4
Withhold dose until clinically stable
Infusion-related reactions [see Warnings and Precautions (5.9)]
Grade 1 or 2
Interrupt or slow the rate of infusion
Grade 3 or 4
Permanently discontinue
Other immune-mediated adverse reactions [see Warnings and Precautions (5.7)]
Grade 3
Withhold dose until Grade 1 or resolved and corticosteroid dose is less than or equal to prednisone 10 mg per day (or equivalent).
Grade 4
Permanently discontinue
Persistent Grade 2 or 3 adverse reaction (excluding endocrinopathies)
Grade 2 or 3 adverse reaction that does not recover to Grade 0 or 1 within 12 weeks after last IMFINZI dose
Permanently discontinue
Inability to taper corticosteroid
Inability to reduce to less than or equal to prednisone 10 mg per day (or equivalent) within 12 weeks after the last IMFINZI dose
Permanently discontinue
Recurrent Grade 3 or 4 adverse reaction
Recurrent Grade 3 or 4 (severe or life-threatening) adverse reaction
Permanently discontinue
2.3 Preparation and Administration
Preparation
- Visually inspect drug product for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard the vial if the solution is cloudy, discolored, or visible particles are observed.
- Do not shake the vial.
- Withdraw the required volume from the vial(s) of IMFINZI and transfer into an intravenous bag containing 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. Mix diluted solution by gentle inversion. Do not shake the solution. The final concentration of the diluted solution should be between 1 mg/mL and 15 mg/mL.
- Discard partially used or empty vials of IMFINZI.
Storage of Infusion Solution
- IMFINZI does not contain a preservative.
-
Administer infusion solution immediately once prepared. If infusion solution is not administered immediately and needs to be stored, the total time from vial puncture to the start of the administration should not exceed:
- o 24 hours in a refrigerator at 2°C to 8°C (36°F to 46°F)
- o 8 hours at room temperature up to 25°C (77°F)
- Do not freeze.
- Do not shake.
Administration
- Administer infusion solution intravenously over 60 minutes through an intravenous line containing a sterile, low-protein binding 0.2 or 0.22 micron in-line filter.
- Do not co-administer other drugs through the same infusion line.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Immune-Mediated Pneumonitis
IMFINZI can cause immune-mediated pneumonitis, defined as requiring use of corticosteroids. Fatal cases have been reported.
Monitor patients for signs and symptoms of pneumonitis. Evaluate patients with suspected pneumonitis with radiographic imaging. Administer corticosteroids, prednisone 1 to 2 mg per kg per day or equivalent for moderate (Grade 2) pneumonitis or prednisone 1 to 4 mg per kg per day or equivalent for more severe (Grade 3-4) pneumonitis, followed by taper. Interrupt or permanently discontinue IMFINZI based on the severity [see Dosage and Administration (2.3)].
In clinical studies enrolling 1889 patients with various cancers who received IMFINZI [see Adverse Reactions (6.1)], pneumonitis occurred in 5% of patients, including Grade 3 (0.8%), Grade 4 (< 0.1%), and Grade 5 (0.3%). The median time to onset was 1.8 months (range: 1 day to 13.9 months) and the median time to resolution was 4.9 months (range: 0 days to 13.7 months).
Pneumonitis led to discontinuation of IMFINZI in 1.5% of the 1889 patients. Pneumonitis was resolved in 54% of patients. Systemic corticosteroids were required in 3.5% of the 1889 patients, with 2.5% requiring high-dose corticosteroids (prednisone ≥ 40 mg per day or equivalent) and 0.1% requiring infliximab.
The frequency and severity of immune-mediated pneumonitis were similar whether IMFINZI was given as a single agent in patients with various cancers or in combination with chemotherapy in patients with ES-SCLC.
The incidence of pneumonitis (including radiation pneumonitis) was higher in patients in the PACIFIC study who completed treatment with definitive chemoradiation within 42 days prior to initiation of IMFINZI (34%) compared to patients in other clinical studies (2.3%) in which radiation therapy was generally not administered immediately prior to initiation of IMFINZI.
In the PACIFIC study, the incidence of Grade 3 pneumonitis was 3.4% and the incidence of Grade 5 pneumonitis was 1.1% in the IMFINZI arm. The median time to onset of pneumonitis was 1.8 months and the median duration was 2.1 months (range: 3 days to 18.7 months). Pneumonitis led to discontinuation of IMFINZI in 6% of patients. Pneumonitis resolved in 47% of patients experiencing pneumonitis. Systemic corticosteroids were required in 21% of patients, with 12% requiring high-dose corticosteroids and 0.1% requiring infliximab.
5.2 Immune-Mediated Hepatitis
IMFINZI can cause immune-mediated hepatitis, defined as requiring use of corticosteroids. Fatal cases have been reported.
Monitor patients for signs and symptoms of hepatitis, during and after discontinuation of IMFINZI, including clinical chemistry monitoring. Administer corticosteroids, prednisone 1 to 2 mg per kg per day or equivalent, followed by taper for Grade 2 or higher elevations of ALT, AST, and/or total bilirubin. Interrupt or permanently discontinue IMFINZI based on the severity [see Dosage and Administration (2.3)].
In clinical studies enrolling 1889 patients with various cancers who received IMFINZI [see Adverse Reactions (6.1)], hepatitis occurred in 12% of patients, including Grade 3 (4.4%), Grade 4 (0.4%), and Grade 5 (0.2%) immune-mediated hepatitis. The median time to onset was 1.2 months (range: 1 day to 13.6 months). Hepatitis led to discontinuation of IMFINZI in 0.7% of the 1889 patients. Hepatitis resolved in 49% of patients. Systemic corticosteroids were required in 2.7% of patients, with 1.7% requiring high-dose corticosteroids and 0.1% requiring mycophenolate.
5.3 Immune-Mediated Colitis
IMFINZI can cause immune-mediated colitis, defined as requiring use of corticosteroids.
Monitor patients for signs and symptoms of diarrhea or colitis. Administer corticosteroids, prednisone 1 to 2 mg per kg per day or equivalent, for moderate (Grade 2) or more severe (Grade 3-4) colitis, followed by taper. Interrupt or permanently discontinue IMFINZI based on the severity [see Dosage and Administration (2.3)].
In clinical studies enrolling 1889 patients with various cancers who received IMFINZI [see Adverse Reactions (6.1)], diarrhea or colitis occurred in 18% of patients, including Grade 3 (1%) and Grade 4 (0.1%) immune-mediated colitis. The median time to onset was 1.4 months (range: 1 day to 14 months). Diarrhea or colitis lead to discontinuation of IMFINZI in 0.4% of the 1889 patients. Diarrhea or colitis resolved in 78% of the patients. Systemic corticosteroids were required in 1.9% of patients, with 1% requiring high-dose corticosteroids and 0.1% requiring other immunosuppressants (e.g., infliximab, mycophenolate).
5.4 Immune-Mediated Endocrinopathies
IMFINZI can cause immune-mediated endocrinopathies, including thyroid disorders, adrenal insufficiency, type 1 diabetes mellitus, and hypophysitis/hypopituitarism.
Thyroid Disorders: Monitor thyroid function prior to and periodically during treatment with IMFINZI. Initiate hormone replacement therapy or medical management of hyperthyroidism as clinically indicated. Continue IMFINZI for hypothyroidism and interrupt for hyperthyroidism based on the severity [see Dosage and Administration (2.3)].
In clinical studies enrolling 1889 patients who received IMFINZI [see Adverse Reactions (6.1)], hypothyroidism occurred in 11% of patients and hyperthyroidism occurred in 7% of patients. Thyroiditis occurred in 0.9% of patients, including Grade 3 (< 0.1%) thyroiditis. Hypothyroidism was preceded by thyroiditis or hyperthyroidism in 25% of patients.
Adrenal Insufficiency: Monitor patients for clinical signs and symptoms of adrenal insufficiency. For Grade 2 or higher adrenal insufficiency, initiate prednisone 1 to 2 mg per kg per day or equivalent, followed by corticosteroid taper and hormone replacement as clinically indicated. Interrupt IMFINZI based on the severity [see Dosage and Administration (2.3)].
In clinical studies enrolling 1889 patients who received IMFINZI, adrenal insufficiency occurred in 0.7% of patients, including Grade 3 (< 0.1%). Systemic corticosteroids were required in 0.4% of patients, including 0.1% of patients who required high-dose corticosteroids.
Type 1 Diabetes Mellitus: Monitor patients for hyperglycemia or other signs and symptoms of diabetes. Initiate treatment with insulin as clinically indicated. Interrupt IMFINZI based on the severity [see Dosage and Administration (2.3)].
In clinical studies enrolling 1889 patients who received IMFINZI, type 1 diabetes mellitus occurred in < 0.1% of patients. The median time to onset was 1.4 months.
Hypophysitis: For Grade 2 or higher hypophysitis, initiate prednisone 1 to 2 mg per kg per day or equivalent, followed by corticosteroid taper and hormone replacement therapy as clinically indicated. Interrupt IMFINZI based on the severity [see Dosage and Administration (2.3)].
Hypopituitarism leading to adrenal insufficiency and diabetes insipidus occurred in < 0.1% of 1889 patients who received IMFINZI in clinical studies.
5.5 Immune-Mediated Nephritis
IMFINZI can cause immune-mediated nephritis, defined as evidence of renal dysfunction requiring use of corticosteroids. Fatal cases have occurred.
Monitor patients for abnormal renal function tests prior to and periodically during treatment with IMFINZI. Initiate prednisone 1 to 2 mg per kg per day or equivalent, for moderate (Grade 2) or severe (Grade 3-4) nephritis, followed by taper. Interrupt or permanently discontinue IMFINZI based on the severity [see Dosage and Administration (2.3)].
In clinical studies enrolling 1889 patients with various cancers who received IMFINZI [see Adverse Reactions (6.1)], nephritis (reported as any of the following increased creatinine or urea, acute kidney injury, renal failure, decreased glomerular filtration rate, tubulointerstitial nephritis, decreased creatinine clearance, glomerulonephritis, and nephritis) occurred in 6.3% of patients including Grade 3 (1.1%), Grade 4 (0.2%), and Grade 5 (0.1%) immune-mediated nephritis. The median time to onset was 2 months (range: 1 day to 14.2 months). IMFINZI was discontinued in 0.3% of the 1889 patients. Nephritis resolved in 50% of patients. Systemic corticosteroids were required in 0.6% of patients, with 0.4% receiving high-dose corticosteroids.
5.6 Immune-Mediated Dermatologic Reactions
IMFINZI can cause immune-mediated rash. Other dermatologic reactions have occurred with other products in this class including Stevens Johnson Syndrome (SJS)/toxic epidermal necrolysis (TEN) [see Warnings and Precautions (5.7)].
Monitor for signs and symptoms of rash. Initiate prednisone 1 to 2 mg per kg per day or equivalent, for moderate (Grade 2) rash or dermatitis lasting for more than 1 week or severe (Grade 3-4) rash or dermatitis, followed by taper. Interrupt or permanently discontinue IMFINZI based on the severity [see Dosage and Administration (2.3)].
In clinical studies enrolling 1889 patients with various cancers who received IMFINZI [see Adverse Reactions (6.1)], 26% of patients developed rash or dermatitis. Rash or dermatitis led to discontinuation of IMFINZI in 0.1% of the 1889 patients. Rash resolved in 62% of patients. Systemic corticosteroids were required in 2% of patients, including high-dose corticosteroids in 1% of patients.
5.7 Other Immune-Mediated Adverse Reactions
IMFINZI can cause severe and fatal immune-mediated adverse reactions. These immune-mediated reactions may involve any organ system. While immune-mediated reactions usually manifest during treatment with IMFINZI, immune-mediated adverse reactions can also manifest after discontinuation of IMFINZI.
For suspected Grade 2 immune-mediated adverse reactions, exclude other causes and initiate corticosteroids as clinically indicated. For severe (Grade 3 or 4) adverse reactions, administer corticosteroids, prednisone 1 to 4 mg per kg per day or equivalent, followed by taper. Interrupt or permanently discontinue IMFINZI, based on the severity of the reaction [see Dosage and Administration (2.3)]. If uveitis occurs in combination with other immune-mediated adverse reactions, evaluate for Vogt-Koyanagi-Harada syndrome, which has been observed with other products in this class and may require treatment with systemic steroids to reduce the risk of permanent vision loss.
The following clinically significant, immune-mediated adverse reactions occurred at an incidence of less than 1% each in 1889 patients who received IMFINZI: aseptic meningitis, hemolytic anemia, immune thrombocytopenic purpura, myocarditis, myositis, and ocular inflammatory toxicity, including uveitis and keratitis [see Adverse Reactions (6.1)]. The following clinically significant, immune-mediated adverse reactions have been reported with other products in this class: bullous dermatitis, Stevens Johnson Syndrome (SJS)/toxic epidermal necrolysis (TEN), pancreatitis, systemic inflammatory response syndrome, rhabdomyolysis, myasthenia gravis, histiocytic necrotizing lymphadenitis, vasculitis, hemolytic anemia, iritis, encephalitis, facial and abducens nerve paresis, demyelination, polymyalgia rheumatica, autoimmune neuropathy, Guillain-Barré syndrome, and Vogt-Koyanagi-Harada syndrome.
5.8 Infection
IMFINZI can cause serious infections, including fatal cases.
Monitor patients for signs and symptoms of infection. For Grade 3 or higher infections, withhold IMFINZI and resume once clinically stable [see Dosage and Administration (2.3)].
In clinical studies enrolling 1889 patients with various cancers who received IMFINZI [see Adverse Reactions (6.1)], infections occurred in 43% of patients, including Grade 3 (8%), Grade 4 (1.9%), and Grade 5 (1%). In the urothelial carcinoma cohort in Study 1108 the most common Grade 3 or higher infection was urinary tract infections, which occurred in 4% of patients. In the PACIFIC study, the most common Grade 3 or higher infection was pneumonia, which occurred in 5% of patients. The overall incidence of infections in IMFINZI-treated patients (56%) in the PACIFIC study was higher compared to patients in other studies (38%) in which radiation therapy was generally not administered immediately prior to initiation of IMFINZI.
5.9 Infusion-Related Reactions
IMFINZI can cause severe or life-threatening infusion-related reactions.
Monitor for signs and symptoms of infusion-related reactions. Interrupt, slow the rate of, or permanently discontinue IMFINZI based on the severity [see Dosage and Administration (2.3)]. For Grade 1 or 2 infusion-related reactions, consider using pre-medications with subsequent doses.
In clinical studies enrolling 1889 patients with various cancers [see Adverse Reactions (6.1)], infusion-related reactions occurred in 2.2% of patients, including Grade 3 (0.3%).
5.10 Embryo-Fetal Toxicity
Based on its mechanism of action and data from animal studies, IMFINZI can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of durvalumab to cynomolgus monkeys from the onset of organogenesis through delivery resulted in increased premature delivery, fetal loss and premature neonatal death. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with IMFINZI and for at least 3 months after the last dose of IMFINZI [see Use in Specific Populations (8.1, 8.3)].
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the labeling.
- Immune-Mediated Pneumonitis [see Warnings and Precautions (5.1)].
- Immune-Mediated Hepatitis [see Warnings and Precautions (5.2)].
- Immune-Mediated Colitis [see Warnings and Precautions (5.3)].
- Immune-Mediated Endocrinopathies [see Warnings and Precautions (5.4)].
- Immune-Mediated Nephritis [see Warnings and Precautions (5.5)].
- Immune-Mediated Dermatologic Reactions [see Warnings and Precautions (5.6)].
- Other Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.7)].
- Infection [see Warnings and Precautions (5.8)].
- Infusion-Related Reactions [see Warnings and Precautions (5.9)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described in the Warnings and Precautions section reflect exposure to IMFINZI in 1889 patients from the PACIFIC study (a randomized, placebo-controlled study that enrolled 475 patients with Stage III NSCLC), Study 1108 (an open-label, single-arm, multicohort study that enrolled 191 patients with urothelial carcinoma and 779 patients with other solid tumors), and additional open-label, single-arm trial that enrolled 444 patients with metastatic lung cancer, an indication for which durvalumab is not approved. In these trials, IMFINZI was administered at a dose of 10 mg/kg every 2 weeks. Among the 1889 patients, 38% were exposed for 6 months or more and 18% were exposed for 12 months or more. The data also reflects exposure to IMFINZI in combination with chemotherapy in 265 patients from the CASPIAN study (a randomized, open-label study in patients with ES-SCLC). In the CASPIAN study, IMFINZI was administered at a dose of 1500 mg every 3 or 4 weeks.
The data described in this section reflect exposure to IMFINZI in patients with locally advanced or metastatic urothelial carcinoma enrolled in Study 1108, in patients with Stage III NSCLC enrolled in the PACIFIC study and in patients with ES-SCLC enrolled in the CASPIAN study.
Urothelial Carcinoma
The safety of IMFINZI was evaluated in 182 patients with locally advanced or metastatic urothelial carcinoma in the urothelial carcinoma cohort of Study 1108 whose disease has progressed during or after one standard platinum-based regimen. Patients received IMFINZI 10 mg/kg intravenously every 2 weeks [see Clinical Studies (14.1)]. The median duration of exposure was 2.3 months (range: 1 day to 12.1 months).
Thirty-one percent (31%) of patients had a drug delay or interruption for an adverse reaction. The most common (> 2%) were liver injury (4.9%), urinary tract infection (3.3%), acute kidney injury (3.3%), and musculoskeletal pain (2.7%).
The most common adverse reactions (≥ 15%) were fatigue (39%), musculoskeletal pain (24%), constipation (21%), decreased appetite (19%), nausea (16%), peripheral edema (15%), and urinary tract infection (15%). The most common Grade 3 or 4 adverse reactions (≥ 3%) were fatigue, urinary tract infection, musculoskeletal pain, abdominal pain, dehydration, and general physical health deterioration.
Eight patients (4.4%) who were treated with IMFINZI experienced Grade 5 adverse reactions of cardiorespiratory arrest, general physical health deterioration, sepsis, ileus, pneumonitis, or immune-mediated hepatitis. Three additional patients were experiencing infection and disease progression at the time of death. IMFINZI was discontinued for adverse reactions in 3.3% of patients. Serious adverse reactions occurred in 46% of patients. The most frequent serious adverse reactions (> 2%) were acute kidney injury (4.9%), urinary tract infection (4.4%), musculoskeletal pain (4.4%), liver injury (3.3%), general physical health deterioration (3.3%), sepsis, abdominal pain, and pyrexia/tumor associated fever (2.7% each).
Table 3 summarizes the adverse reactions that occurred in ≥ 10% of patients, while Table 4 summarizes the Grade 3 - 4 laboratory abnormalities that occurred in ≥ 1% of patients treated with IMFINZI in the urothelial carcinoma cohort of Study 1108.
Table 3. Adverse Reactions in ≥ 10% of Patients in Study 1108 Urothelial Carcinoma Cohort IMFINZI
N = 182Adverse Reaction All Grades
(%)Grades 3 – 4
(%)- * Includes abdominal pain upper, abdominal pain lower, and flank pain
- † Includes asthenia, lethargy, and malaise
- ‡ Includes edema, localized edema, edema peripheral, lymphedema, peripheral swelling, scrotal edema, and scrotal swelling
- § Includes cystitis, candiduria, and urosepsis
- ¶ Includes back pain, musculoskeletal chest pain, musculoskeletal pain and discomfort, myalgia, and neck pain
- # Includes dermatitis, dermatitis acneiform, dermatitis psoriasiform, psoriasis, rash maculo-papular, rash pruritic, rash papular, rash pustular, skin toxicity, eczema, erythema, erythema multiforme, rash erythematous, acne, and lichen planus
Gastrointestinal Disorders
Constipation
21
1.1
Nausea
16
1.6
Abdominal pain*
14
2.7
Diarrhea/Colitis
13
1.1
General Disorders and Administration
Fatigue†
39
6
Peripheral edema‡
15
1.6
Pyrexia/Tumor associated fever
14
0.5
Infections
Urinary tract infection§
15
4.4
Metabolism and Nutrition Disorders
Decreased appetite/Hypophagia
19
0.5
Musculoskeletal and Connective Tissue Disorders
Musculoskeletal pain¶
24
3.8
Respiratory, Thoracic, and Mediastinal Disorders
Dyspnea/Exertional Dyspnea
13
2.2
Cough/Productive Cough
10
0
Skin and Subcutaneous Tissue Disorders
Rash#
11
0.5
Table 4. Grade 3-4 Laboratory Abnormalities Worsened from Baseline Occurring in ≥ 1% Patients in Study 1108 Urothelial Carcinoma Cohort Laboratory Abnormality
Grades 3 - 4
%
Hyponatremia
12
Lymphopenia
11
Anemia
8
Increased alkaline phosphatase
4.1
Hypermagnesemia
4.2
Hypercalcemia
3
Hyperglycemia
3
Increased AST
2.4
Increased ALT
0.6
Hyperbilirubinemia
1.2
Increased creatinine
1.2
Neutropenia
1.2
Hyperkalemia
1.2
Hypokalemia
1.2
Hypoalbuminemia
1.2
Non-Small Cell Lung Cancer
The safety of IMFINZI in patients with Stage III NSCLC who completed concurrent platinum-based chemoradiotherapy within 42 days prior to initiation of study drug was evaluated in the PACIFIC study, a multicenter, randomized, double-blind, placebo-controlled study. A total of 475 patients received IMFINZI 10 mg/kg intravenously every 2 weeks. The study excluded patients who had disease progression following chemoradiation, with active or prior autoimmune disease within 2 years of initiation of the study or with medical conditions that required systemic immunosuppression [see Clinical Studies (14.2)].
The study population characteristics were: median age of 64 years (range: 23 to 90), 45% age 65 years or older, 70% male, 69% White, 27% Asian, 75% former smoker, 16% current smoker, and 51% had WHO performance status of 1. All patients received definitive radiotherapy as per protocol, of which 92% received a total radiation dose of 54 Gy to 66 Gy. The median duration of exposure to IMFINZI was 10 months (range: 0.2 to 12.6).
IMFINZI was discontinued due to adverse reactions in 15% of patients. The most common adverse reactions leading to IMFINZI discontinuation were pneumonitis or radiation pneumonitis in 6% of patients. Serious adverse reactions occurred in 29% of patients receiving IMFINZI. The most frequent serious adverse reactions reported in at least 2% of patients were pneumonitis or radiation pneumonitis (7%) and pneumonia (6%). Fatal pneumonitis or radiation pneumonitis and fatal pneumonia occurred in < 2% of patients and were similar across arms. The most common adverse reactions (occurring in ≥ 20% of patients) were cough, fatigue, pneumonitis or radiation pneumonitis, upper respiratory tract infections, dyspnea, and rash.
Table 5 summarizes the adverse reactions that occurred in at least 10% of patients treated with IMFINZI.
Table 5. Adverse Reactions Occurring in ≥ 10% Patients in the PACIFIC Study - * The PACIFIC study was not designed to demonstrate statistically significant difference in adverse reaction rates for IMFINZI, as compared to placebo, for any specific adverse reaction listed in Table 4
- † Includes acute interstitial pneumonitis, interstitial lung disease, pneumonitis, pulmonary fibrosis
- ‡ Includes dyspnea, and exertional dyspnea
- § Includes abdominal pain, abdominal pain lower, abdominal pain upper, and flank pain
- ¶ Includes autoimmune hypothyroidism and hypothyroidism
- # Includes rash erythematous, rash generalized, rash macular, rash maculopapular, rash papular, rash pruritic, rash pustular, erythema, eczema, rash, and dermatitis
- Þ Includes pruritus generalized and pruritus
- ß Includes asthenia and fatigue
- à Includes laryngitis, nasopharyngitis, peritonsillar abscess, pharyngitis, rhinitis, sinusitis, tonsillitis, tracheobronchitis, and upper respiratory tract infection
- è Includes lung infection, pneumocystis jirovecii pneumonia, pneumonia, pneumonia adenoviral, pneumonia bacterial, pneumonia cytomegaloviral, pneumonia haemophilus, pneumonia klebsiella, pneumonia necrotising, pneumonia pneumococcal, and pneumonia streptococcal
IMFINZI
N = 475
Placebo*
N = 234
Adverse Reaction
All Grades
(%)
Grades 3-4
(%)
All Grades
(%)
Grades 3-4
(%)
Respiratory, Thoracic, and Mediastinal Disorders
Cough/Productive Cough
40
0.6
30
0.4
Pneumonitis†/Radiation Pneumonitis
34
3.4
25
3
Dyspnea‡
25
1.5
25
2.6
Gastrointestinal Disorders
Diarrhea
18
0.6
19
1.3
Abdominal pain§
10
0.4
6
0.4
Endocrine Disorders
Hypothyroidism¶
12
0.2
1.7
0
Skin and Subcutaneous Tissue Disorders
Rash#
23
0.6
12
0
PruritusÞ
12
0
6
0
General Disorders
Fatigueß
34
0.8
32
1.3
Pyrexia
15
0.2
9
0
Infections
Upper respiratory tract infectionsà
26
0.4
19
0
Pneumoniaè
17
7
12
6
Other adverse reactions occurring in less than 10% of patients treated with IMFINZI were dysphonia, dysuria, night sweats, peripheral edema, and increased susceptibility to infections.
Table 6 summarizes the laboratory abnormalities that occurred in at least 20% of patients treated with IMFINZI.
Table 6. Laboratory Abnormalities Worsening from Baseline Occurring in ≥ 20% of Patients in the PACIFIC Study - * Graded according to NCI CTCAE version 4.0
- † Each test incidence is based on the number of patients who had both baseline and at least one on-study laboratory measurement available: IMFINZI (range: 464 to 470) and placebo (range: 224 to 228)
IMFINZI
Placebo
Laboratory Abnormality
All Grades*
(%)†
Grade 3 or 4
(%)
All Grades*
(%)†
Grade 3 or 4
(%)
Chemistry
Hyperglycemia
52
8
51
8
Hypocalcemia
46
0.2
41
0
Increased ALT
39
2.3
22
0.4
Increased AST
36
2.8
21
0.4
Hyponatremia
33
3.6
30
3.1
Hyperkalemia
32
1.1
29
1.8
Increased GGT
24
3.4
22
1.7
Hematology
Lymphopenia
43
17
39
18
Small Cell Lung Cancer
The safety of IMFINZI in combination with etoposide and either carboplatin or cisplatin in previously untreated ES-SCLC was evaluated in CASPIAN, a randomized, open-label, multicenter, active-controlled trial. A total of 265 patients received IMFINZI 1500 mg in combination with chemotherapy every 3 weeks for 4 cycles followed by IMFINZI 1500 mg every 4 weeks until disease progression or unacceptable toxicity. The trial excluded patients with active or prior autoimmune disease or with medical conditions that required systemic corticosteroids or immunosuppressants [see Clinical Studies (14.3)]. Among 265 patients receiving IMFINZI, 49% were exposed for 6 months or longer and 19% were exposed for 12 months or longer.
Among 266 patients receiving chemotherapy alone, 57% of the patients received 6 cycles of chemotherapy and 8% of the patients received PCI after chemotherapy.
IMFINZI was discontinued due to adverse reactions in 7% of the patients receiving IMFINZI plus chemotherapy. These include pneumonitis, hepatotoxicity, neurotoxicity, sepsis, diabetic ketoacidosis and pancytopenia (1 patient each). Serious adverse reactions occurred in 31% of patients receiving IMFINZI plus chemotherapy. The most frequent serious adverse reactions reported in at least 1% of patients were febrile neutropenia (4.5%), pneumonia (2.3%), anemia (1.9%), pancytopenia (1.5%), pneumonitis (1.1%) and COPD (1.1%). Fatal adverse reactions occurred in 4.9% of patients receiving IMFINZI plus chemotherapy. These include pancytopenia, sepsis, septic shock, pulmonary artery thrombosis, pulmonary embolism, and hepatitis (1 patient each) and sudden death (2 patients). The most common adverse reactions (occurring in ≥ 20% of patients) were nausea, fatigue/asthenia and alopecia.
Table 7 summarizes the adverse reactions that occurred in patients treated with IMFINZI plus chemotherapy.
Table 7. Adverse Reactions Occurring in ≥ 10% Patients in the CASPIAN study IMFINZI with etoposide and either carboplatin or cisplatin
N = 265Etoposide and either carboplatin or cisplatin
N = 266Adverse Reaction All Grades (%) Grade 3-4 (%) All Grades (%) Grade 3-4 (%) - * Includes hyperthyroidism and Basedow's disease
- † Includes rash erythematous, rash generalized, rash macular, rash maculopapular, rash papular, rash pruritic, rash pustular, erythema, eczema, rash and dermatitis
Respiratory, thoracic and mediastinal disorders
Cough/Productive Cough
15
0.8
9
0
Gastrointestinal disorders
Nausea
34
0.4
34
1.9
Constipation
17
0.8
19
0
Vomiting
15
0
17
1.1
Diarrhea
10
1.1
11
1.1
Endocrine disorders
Hyperthyroidism*
10
0
0.4
0
Skin and subcutaneous tissue disorders
Alopecia
31
1.1
34
0.8
Rash†
11
0
6
0
General disorders and administration site conditions
Fatigue/Asthenia
32
3.4
32
2.3
Metabolism and nutrition disorders
Decreased appetite
18
0.8
17
0.8
Table 8 summarizes the laboratory abnormalities that occurred in at least 20% of patients treated with IMFINZI plus chemotherapy.
Table 8. Laboratory Abnormalities Worsening from Baseline Occurring in ≥ 20%* of Patients in the CASPIAN study - * The frequency cut off is based on any grade change from baseline
- † Graded according to NCI CTCAE version 4.03
- ‡ Each test incidence is based on the number of patients who had both baseline and at least one on-study laboratory measurement available: IMFINZI (range: 258 to 263) and chemotherapy (range: 253 to 262) except magnesium IMFINZI + chemotherapy (18) and chemotherapy (16)
- § LLN = lower limit of normal
IMFINZI with Etoposide and either Carboplatin or Cisplatin
Etoposide and either Carboplatin or Cisplatin
Laboratory Abnormality
Chemistry
Hyponatremia
11
13
Hypomagnesemia
11
6
Hyperglycemia
5
5
Increased Alkaline Phosphatase
4.9
3.5
Increased ALT
4.9
2.7
Increased AST
4.6
1.2
Hypocalcemia
3.5
2.4
Blood creatinine increased
3.4
1.1
Hyperkalemia
1.5
3.1
TSH decreased < LLN§ and ≥ LLN at baseline
NA
NA
Hematology
Neutropenia
41
48
Lymphopenia
14
13
Anemia
13
22
Thrombocytopenia
12
15
6.2 Immunogenicity
As with all therapeutic proteins, there is a potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to durvalumab to the incidence of antibodies to other products may be misleading.
Of 2280 patients who received IMFINZI 10 mg/kg every 2 weeks or 20 mg/kg every 4 weeks as a single-agent, 69 patients (3%) tested positive for treatment-emergent anti-drug antibodies (ADA) and 12 (0.5%) tested positive for neutralizing antibodies. The development of ADA against durvalumab appears to have no clinically relevant effect on its pharmacokinetics or safety.
Of 201 patients in the CASPIAN study who received IMFINZI 1500 mg every 3 weeks in combination with chemotherapy for four doses followed by IMFINZI 1500 mg every 4 weeks no patients tested positive for treatment-emergent ADA.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk summary
Based on its mechanism of action and data from animal studies, IMFINZI can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1)]. There are no data on the use of IMFINZI in pregnant women.
In animal reproduction studies, administration of durvalumab to pregnant cynomolgus monkeys from the confirmation of pregnancy through delivery resulted in an increase in premature delivery, fetal loss, and premature neonatal death (see Data). Human immunoglobulin G1 (IgG1) is known to cross the placental barrier; therefore, durvalumab has the potential to be transmitted from the mother to the developing fetus. Apprise pregnant women of the potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
As reported in the literature, the PD-1/PD-L1 pathway plays a central role in preserving pregnancy by maintaining maternal immune tolerance to the fetus. In mouse allogeneic pregnancy models, disruption of PD-L1 signaling was shown to result in an increase in fetal loss. The effects of durvalumab on prenatal and postnatal development were evaluated in reproduction studies in cynomolgus monkeys. Durvalumab was administered from the confirmation of pregnancy through delivery at exposure levels approximately 6 to 20 times higher than those observed at the recommended clinical dose of 10 mg/kg (based on AUC). Administration of durvalumab resulted in premature delivery, fetal loss (abortion and stillbirth), and increase in neonatal deaths. Durvalumab was detected in infant serum on postpartum Day 1, indicating the presence of placental transfer of durvalumab. Based on its mechanism of action, fetal exposure to durvalumab may increase the risk of developing immune-mediated disorders or altering the normal immune response and immune-mediated disorders have been reported in PD-1 knockout mice.
8.2 Lactation
Risk Summary
There is no information regarding the presence of durvalumab in human milk, the effects on the breastfed infant, or the effects on milk production. Human IgG1 is excreted in human milk. Durvalumab was present in the milk of lactating cynomolgus monkeys and was associated with premature neonatal death (see Data).
Because of the potential for adverse reactions in breastfed infants, advise women not to breastfeed during treatment with IMFINZI and for at least 3 months after the last dose.
Data
In lactating cynomolgus monkeys, durvalumab was present in breast milk at about 0.15% of maternal serum concentrations after administration of durvalumab from the confirmation of pregnancy through delivery at exposure levels approximately 6 to 20 times higher than those observed at the recommended clinical dose of 10 mg/kg (based on AUC). Administration of durvalumab resulted in premature neonatal death.
8.3 Females and Males of Reproductive Potential
Contraception
Females
Based on its mechanism of action and data from animal studies, IMFINZI can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)]. Advise females of reproductive potential to use effective contraception during treatment with IMFINZI and for at least 3 months following the last dose of IMFINZI.
8.4 Pediatric Use
The safety and effectiveness of IMFINZI have not been established in pediatric patients.
8.5 Geriatric Use
Of the 182 patients treated with IMFINZI in patients with urothelial carcinoma, 112 patients were 65 years or older and 34 patients were 75 years or older. The overall response rate in patients 65 years or older was 15% (17/112) and was 12% (4/34) in patients 75 years or older. Grade 3 or 4 adverse reactions occurred in 38% (42/112) of patients 65 years or older and 35% (12/34) of patients 75 years or older.
Of the 476 patients treated with IMFINZI in the PACIFIC study, 45% were 65 years or older, while 7.6% were 75 years or older. No overall differences in safety or effectiveness were observed between patients 65 years or older and younger patients. The PACIFIC study did not include sufficient numbers of patients aged 75 years and over to determine whether they respond differently from younger patients.
Of the 265 patients with ES-SCLC treated with IMFINZI in combination with chemotherapy, 101 (38%) patients were 65 years or older and 19 (7.2%) patients were 75 years or older. There were no clinically meaningful differences in safety or efficacy between patients 65 years or older and younger patients.
-
11 DESCRIPTION
Durvalumab is a programmed cell death ligand 1 (PD-L1) blocking antibody. Durvalumab is a human immunoglobulin G1 kappa (IgG1κ) monoclonal antibody that is produced by recombinant DNA technology in Chinese Hamster Ovary (CHO) cell suspension culture.
IMFINZI (durvalumab) Injection for intravenous use is a sterile, preservative-free, clear to opalescent, colorless to slightly yellow solution, free from visible particles.
Each 500 mg vial of IMFINZI contains 500 mg of durvalumab in 10 mL solution. Each mL contains durvalumab, 50 mg, L-histidine (2 mg), L-histidine hydrochloride monohydrate (2.7 mg), α,α-trehalose dihydrate (104 mg), Polysorbate 80 (0.2 mg), and Water for Injection, USP.
Each 120 mg vial of IMFINZI contains 120 mg of durvalumab in 2.4 mL solution. Each mL contains durvalumab, 50 mg, L-histidine (2 mg), L-histidine hydrochloride monohydrate (2.7 mg), α,α-trehalose dihydrate (104 mg), Polysorbate 80 (0.2 mg), and Water for Injection, USP.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Expression of programmed cell death ligand-1 (PD-L1) can be induced by inflammatory signals (e.g., IFN-gamma) and can be expressed on both tumor cells and tumor-associated immune cells in the tumor microenvironment. PD-L1 blocks T-cell function and activation through interaction with PD-1 and CD80 (B7.1). By binding to its receptors, PD-L1 reduces cytotoxic T-cell activity, proliferation, and cytokine production.
Durvalumab is a human immunoglobulin G1 kappa (IgG1κ) monoclonal antibody that binds to PD-L1 and blocks the interaction of PD-L1 with PD-1 and CD80 (B7.1). Blockade of PD-L1/PD-1 and PD-L1/CD80 interactions releases the inhibition of immune responses, without inducing antibody dependent cell-mediated cytotoxicity (ADCC).
PD-L1 blockade with durvalumab led to increased T-cell activation in vitro and decreased tumor size in co-engrafted human tumor and immune cell xenograft mouse models.
12.3 Pharmacokinetics
The pharmacokinetics of durvalumab as a single agent was studied in patients with doses ranging from 0.1 mg/kg (0.01 times the approved recommended dosage) to 20 mg/kg (2 times the approved recommended dosage) administered once every two, three, or four weeks.
PK exposure increased more than dose-proportionally at doses < 3 mg/kg (0.3 times the approved recommended dosage) and dose proportionally at doses ≥ 3 mg/kg every 2 weeks. Steady state was achieved at approximately 16 weeks.
The pharmacokinetics of durvalumab is similar when assessed as a single agent and when in combination with chemotherapy.
Distribution
The geometric mean (% coefficient of variation [CV%]) steady state volume of distribution (Vss) was 5.6 (18%) L.
Elimination
Durvalumab clearance decreases over time, with a mean maximal reduction (CV%) from baseline values of approximately 23% (57%) resulting in a geometric mean (CV%) steady state clearance (CLss) of 8.2 mL/h (39%) at day 365; the decrease in CLss is not considered clinically relevant. The geometric mean (CV%) terminal half-life, based on baseline CL was approximately 18 (24%) days.
Specific Populations
Age (19–96 years), body weight (31-149 kg), sex, albumin levels, lactate dehydrogenase (LDH) levels, creatinine levels, soluble PD-L1, tumor type, race, mild renal impairment (creatinine clearance (CLcr) 60 to 89 mL/min), moderate renal impairment (CLcr 30 to 59 mL/min), mild hepatic impairment (bilirubin ≤ ULN and AST > ULN or bilirubin > 1 to 1.5x ULN and any AST), or ECOG/WHO performance status had no clinically significant effect on the pharmacokinetics of durvalumab.
The effect of severe renal impairment (CLcr 15 to 29 mL/min) or moderate hepatic impairment (bilirubin > 1.5 to 3x ULN and any AST) or severe hepatic impairment (bilirubin > 3x ULN and any AST) on the pharmacokinetics of durvalumab is unknown.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic and genotoxic potential of durvalumab have not been evaluated.
Animal fertility studies have not been conducted with durvalumab. In repeat-dose toxicology studies with durvalumab in sexually mature cynomolgus monkeys of up to 3 months duration, there were no notable effects on the male and female reproductive organs.
13.2 Animal Toxicology and/or Pharmacology
In animal models, inhibition of PD-L1/PD-1 signaling increased the severity of some infections and enhanced inflammatory responses. M. tuberculosis-infected PD-1 knockout mice exhibit markedly decreased survival compared with wild-type controls, which correlated with increased bacterial proliferation and inflammatory responses in these animals. PD-L1 and PD-1 knockout mice have also shown decreased survival following infection with lymphocytic choriomeningitis virus.
-
14 CLINICAL STUDIES
14.1 Urothelial Carcinoma
The efficacy of IMFINZI was evaluated in the urothelial carcinoma cohort of Study 1108 (NCT01693562), a multicenter, multi-cohort, open-label clinical trial. In Study 1108, 182 patients with locally advanced or metastatic urothelial carcinoma were enrolled. Patients had progressed while on or after a platinum-based therapy, including those who progressed within 12 months of receiving therapy in a neo-adjuvant or adjuvant setting. These patients had initiated IMFINZI at least 13 weeks prior to the data cut-off date. The trial excluded patients with a history of immunodeficiency; medical conditions that required systemic immunosuppression (not to exceed 10 mg per day of prednisone or equivalent); history of severe autoimmune disease; untreated CNS metastases; HIV; active tuberculosis, or hepatitis B or C infection. All patients received IMFINZI 10 mg/kg intravenously every 2 weeks for up to 12 months or until unacceptable toxicity or disease progression. Tumor assessments were performed at Weeks 6, 12, and 16, then every 8 weeks for the first year and every 12 weeks thereafter. The major efficacy outcome measures were confirmed Overall Response Rate (ORR) according to RECIST v1.1 as assessed by Blinded Independent Central Review (BICR), and duration of response (DoR).
The median age was 67 years (range: 34 to 88), 72% were male, 64% were White. Sixty-six percent (66%) of patients had visceral metastasis (bone, liver, or lung), including 34% with liver metastasis. Lymph node only metastasis were present in 13% of patients. Sixty-six percent (66%) of patients had ECOG score of 1 and 41% of patients had a baseline creatinine clearance < 60 mL/min. The Bellmunt risk score (which includes ECOG score, baseline hemoglobin, and liver metastases) was 0 in 23%, 1 in 38%, 2 in 29%, and 3 in 9% of patients. Twenty percent (20%) of patients had disease progression following platinum-containing neoadjuvant or adjuvant chemotherapy as their only prior line of therapy. Seventy percent (70%) of patients received prior cisplatin, 30% prior carboplatin and 35% received ≥ 2 prior lines of systemic therapy.
Tumor specimens were evaluated prospectively for PD-L1 expression on tumor cells (TC) and immune cells (IC) at a central laboratory using the VENTANA PD-L1 (SP263) Assay. Of the 182 patients, 52% were classified as PD-L1 high (if ICs involve > 1% of the tumor area, TC ≥ 25% or IC ≥ 25%; if ICs involve ≤ 1% of the tumor area, TC ≥ 25% or IC = 100%), 40% as PD-L1 low/negative (did not meet criterion for PD-L1 high), and samples for 8% were not evaluable.
Table 9 summarizes the results in the urothelial carcinoma cohort of Study 1108. The median follow-up time was 5.6 months. In 37 patients who had received only neoadjuvant or adjuvant therapy prior to study entry, 24% responded.
Among the total 31 responding patients, 45% had ongoing responses of 6 months or longer and 16% had ongoing responses of 12 months or longer.
Table 9. Efficacy Results for Study 1108 Urothelial Carcinoma Cohort All Patients
N = 182PD-L1
High
N = 95PD-L1 Low/Negative
N = 73PD-L1
NE
N = 14Overall Response Rate by BICR n (%) (95% CI)
31 (17%)
(11.9, 23.3)25 (26%)
(17.8, 36.4)3 (4%)
(0.9, 11.5)3 (21%)
(4.7, 50.8)Complete Response, n
5
3
1
1
Partial Response, n
26
22
2
2
Median Duration of Response months
(range)
NR
(0.9+, 19.9+)NR
(0.9+, 19.9+)12.3
(1.9+, 12.3)NR
(2.3+, 2.6+)BICR = Blinded Independent Central Review; NR = Not Reached; + denotes a censored value
14.2 Non-Small Cell Lung Cancer (NSCLC)
The efficacy of IMFINZI was evaluated in the PACIFIC study (NCT02125461), a multicenter, randomized, double-blind, placebo-controlled study in patients with unresectable Stage III NSCLC who completed at least 2 cycles of concurrent platinum-based chemotherapy and definitive radiation within 42 days prior to initiation of the study drug and had a WHO performance status of 0 or 1. The study excluded patients who had progressed following concurrent chemoradiation, patients with active or prior documented autoimmune disease within 2 years of initiation of the study or patients with medical conditions that required systemic immunosuppression. Randomization was stratified by sex, age (< 65 years vs. ≥ 65 years), and smoking history (smoker vs. non-smoker). Patients were randomized 2:1 to receive IMFINZI 10 mg/kg or placebo intravenously every 2 weeks for up to 12 months or until unacceptable toxicity or confirmed RECIST v1.1-defined progression. Assessment of tumor status was performed every 8 weeks. The major efficacy outcome measures were progression-free survival (PFS) as assessed by a BICR RECIST v1.1, and overall survival (OS). Additional efficacy outcome measures included ORR and DoR assessed by BICR.
A total of 713 patients were randomized: 476 patients to the IMFINZI arm and 237 to the placebo arm. The study population characteristics were: median age of 64 years (range: 23 to 90); 70% male; 69% White and 27% Asian; 16% current smokers, 75% former smokers, and 9% never smokers; 51% WHO performance status of 1; 53% with Stage IIIA and 45% were Stage IIIB; 46% with squamous and 54% with non-squamous histology. All patients received definitive radiotherapy as per protocol, of which 92% received a total radiation dose of 54 Gy to 66 Gy; 99% of patients received concomitant platinum-based chemotherapy (55% cisplatin-based, 42% carboplatin-based chemotherapy, and 2% switched between cisplatin and carboplatin).
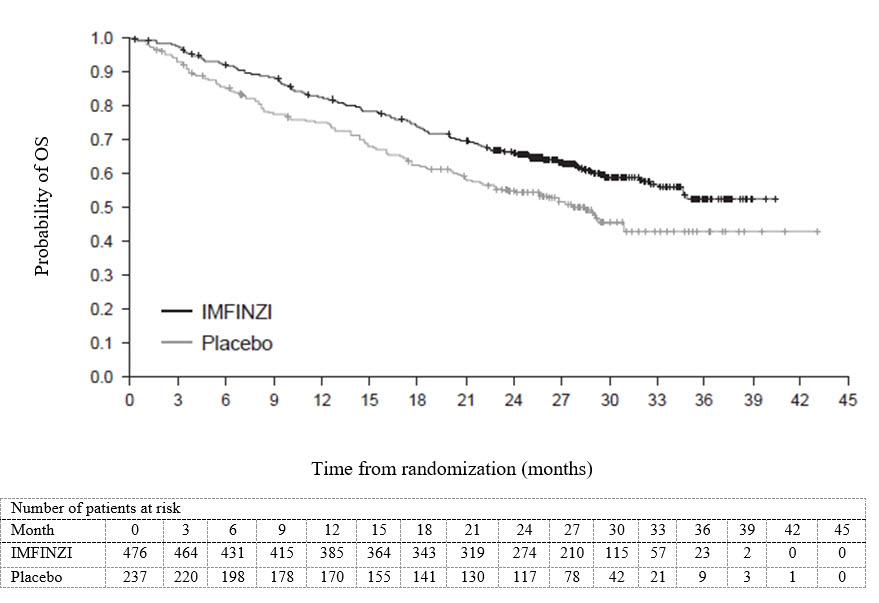
At a pre-specified interim analysis for OS based on 299 events (61% of total planned events), the study demonstrated a statistically significant improvement in OS in patients randomized to IMFINZI compared to placebo. The pre-specified interim analysis of PFS based on 371 events (81% of total planned events) demonstrated a statistically significant improvement in PFS in patients randomized to IMFINZI compared to placebo. Table 10 and Figure 1 summarizes the efficacy results for PACIFIC.
Table 10. Efficacy Results for the PACIFIC Study Endpoint IMFINZI (N = 476)* Placebo (N = 237)* - * Among the ITT population, 7% in the IMFINZI arm and 10% in the placebo arm had non-measurable disease as assessed by BICR according to RECIST v1.1
- † OS results are based on the interim OS analysis conducted at 299 OS events which occurred 46 months after study initiation
- ‡ Two-sided p-value based on a log-rank test stratified by sex, age, and smoking history
- § Compared with allocated α of 0.00274 (Lan DeMets spending function approximating O’Brien Fleming boundary) for interim analysis
- ¶ As assessed by BICR RECIST v1.1
- # PFS results are based on the interim PFS analysis conducted at 371 PFS events which occurred 33 months after study initiation
- Þ Pike estimator
- ß Compared with allocated α of 0.011035 (Lan DeMets spending function approximating O’Brien Fleming boundary) for interim analysis
Overall Survival (OS)†
Number of deaths
183 (38%0
116 (49%0
Median in months (95% CI)
NR
(34.7, NR)
28.7
(22.9, NR)
Hazard Ratio (95% CI)‡
0.68 (0.53, 0.87)
0.0025
Number (%) of patients with event
214 (45%)
157 (66%)
Median in months (95% CI)
16.8 (13.0, 18.1)
5.6 (4.6, 7.8)
0.52 (0.42, 0.65)
< 0.0001
Figure 1 Kaplan-Meier Curves of Overall Survival in the PACIFIC Study
14.3 Small Cell Lung Cancer (SCLC)
The efficacy of IMFINZI in combination with etoposide and either carboplatin or cisplatin in previously untreated ES-SCLC was investigated in CASPIAN, a randomized, multicenter, active-controlled, open-label trial (NCT03043872). Eligible patients had WHO Performance Status of 0 or 1 and were suitable to receive a platinum-based chemotherapy regimen as first-line treatment for SCLC. Patients with asymptomatic or treated brain metastases were eligible. Choice of platinum agent was at the investigator’s discretion, taking into consideration the calculated creatinine clearance. Patients with history of chest radiation therapy; a history of active primary immunodeficiency; autoimmune disorders including paraneoplastic syndrome; active or prior documented autoimmune or inflammatory disorders; use of systemic immunosuppressants within 14 days before the first dose of the treatment except physiological dose of systemic corticosteroids were ineligible.
Randomization was stratified by the planned platinum-based therapy in cycle 1 (carboplatin or cisplatin).
The evaluation of efficacy for ES-SCLC relied on comparison between:
- IMFINZI 1500 mg, and investigator’s choice of carboplatin (AUC 5 or 6 mg/mL/min) or cisplatin (75-80 mg/m2) on Day 1 and etoposide (80-100 mg/m2) intravenously on Days 1, 2, and 3 of each 21-day cycle for 4 cycles, followed by IMFINZI 1500 mg every 4 weeks until disease progression or unacceptable toxicity, or
- Investigator’s choice of carboplatin (AUC 5 or 6 mg/mL/min) or cisplatin (75-80 mg/m2) on Day 1 and etoposide (80-100 mg/m2) intravenously on Days 1, 2, and 3 of each 21-day cycle, up to 6 cycles. After completion of chemotherapy, prophylactic cranial irradiation (PCI) as administered per investigator discretion.
Administration of IMFINZI as a single agent was permitted beyond disease progression if the patient was clinically stable and deriving clinical benefit as determined by the investigator.
The major efficacy outcome measure was overall survival (OS) of IMFINZI plus chemotherapy vs. chemotherapy alone. Additional efficacy outcome measures were investigator-assessed progression-free survival (PFS) and objective response rate (ORR), per RECIST v1.1.
The study population characteristics were: median age of 63 years (range: 28 to 82); 40% age 65 or older; 70% male; 84% White, 15% Asian, and 0.9% Black; 65% WHO/ECOG PS of 1; and 93% were former/current smokers. Ninety percent of patients had Stage IV disease and 10% had brain metastasis at baseline. A total of 25% of the patients received cisplatin and 74% of the patients received carboplatin. In the chemotherapy alone arm, 57% of the patients received 6 cycles of chemotherapy, and 8% of the patients received PCI.
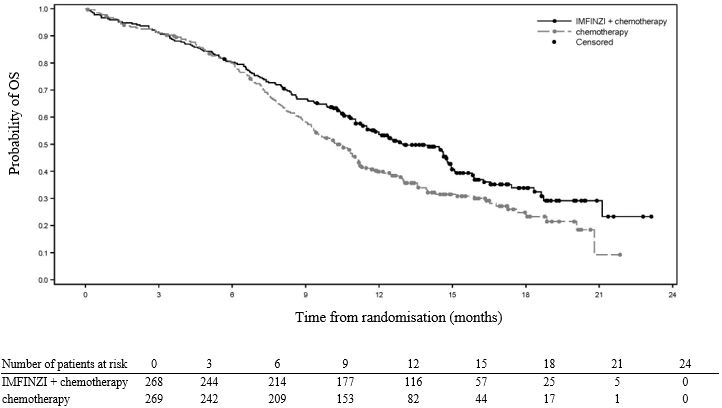
The OS results are summarized in Table 11 and Figure 2.
Table 11. OS Result for the CASPIAN Study - * At a pre-specified interim analysis, 336 OS events (79% of total planned events) were observed, and the boundary for declaring efficacy (0.0178) was determined by a Lan-Demets alpha spending function with O’Brien Fleming type boundary
- † The analysis was performed using the stratified log-rank test, adjusting for planned platinum therapy in Cycle 1 (carboplatin or cisplatin) and using the rank tests of association approach
Endpoint
IMFINZI with Etoposide and either Carboplatin or Cisplatin
(n=268)
Etoposide and either Carboplatin or Cisplatin
(n=269)
Overall Survival (OS)
Number of deaths (%)*
155 (58)
181 (67)
Median OS (months) (95% CI)
13.0
(11.5, 14.8)
10.3
(9.3, 11.2)
Hazard Ratio (95% CI)†
0.73 (0.59, 0.91)
p-value*
0.0047
Figure 2. Kaplan-Meier Curves of Overall Survival in the CASPIAN Study
Investigator-assessed PFS (96% of total planned events) showed a HR of 0.78 (95% CI: 0.65, 0.94), with median PFS of 5.1 months (95% CI: 4.7, 6.2) in the IMFINZI plus chemotherapy arm and 5.4 months (95% CI: 4.8, 6.2) in the chemotherapy alone arm. The investigator-assessed confirmed ORR was 68% (95% CI: 62%, 73%) in the IMFINZI plus chemotherapy arm and 58% (95% CI: 52%, 63%) in the chemotherapy alone arm.
In the exploratory subgroup analyses of OS based on the planned platinum chemotherapy received at cycle 1, the HR was 0.70 (95% CI 0.55, 0.89) in patients who received carboplatin, and the HR was 0.88 (95% CI 0.55, 1.41) in patients who received cisplatin.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
IMFINZI (durvalumab) Injection is a clear to opalescent, colorless to slightly yellow solution supplied in a carton containing one single-dose vial either as:
- 500 mg/10 mL (NDC: 0310-4611-50)
- 120 mg/2.4 mL (NDC: 0310-4500-12)
Store in a refrigerator at 2°C to 8°C (36°F to 46°F) in original carton to protect from light.
Do not freeze. Do not shake.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Inform patients of the risk of immune-mediated adverse reactions that may require corticosteroid treatment and interruption or discontinuation of IMFINZI, including:
- Pneumonitis: Advise patients to contact their healthcare provider immediately for any new or worsening cough, chest pain, or shortness of breath [see Warnings and Precautions (5.1)].
- Hepatitis: Advise patients to contact their healthcare provider immediately for jaundice, severe nausea or vomiting, pain on the right side of abdomen, lethargy, or easy bruising or bleeding [see Warnings and Precautions (5.2)].
- Colitis: Advise patients to contact their healthcare provider immediately for diarrhea, blood or mucus in stools, or severe abdominal pain [see Warnings and Precautions (5.3)].
- Endocrinopathies: Advise patients to contact their healthcare provider immediately for signs or symptoms of hypothyroidism, hyperthyroidism, adrenal insufficiency, type 1 diabetes mellitus, or hypophysitis [see Warnings and Precautions (5.4)].
- Nephritis: Advise patients to contact their healthcare provider immediately for signs or symptoms of nephritis [see Warnings and Precautions (5.5)].
- Dermatological Reactions: Advise patients to contact their healthcare provider immediately for signs or symptoms of severe dermatological reactions [see Warnings and Precautions (5.6)].
- Other Immune-Mediated Adverse Reactions: Advise patients to contact their healthcare provider immediately for signs or symptoms of aseptic meningitis, thrombocytopenic purpura, myocarditis, hemolytic anemia, myositis, uveitis, and keratitis [see Warnings and Precautions (5.7)].
- Infection: Advise patients to contact their healthcare provider immediately for infection [see Warnings and Precautions (5.8)].
- Infusion-Related Reactions: Advise patients to contact their healthcare provider immediately for signs or symptoms of infusion-related reactions [see Warnings and Precautions (5.9)].
-
Embryo-Fetal Toxicity: Advise females of reproductive potential that IMFINZI can cause harm to a fetus and to inform their healthcare provider of a known or suspected pregnancy [see Warnings and Precautions (5.10) and Use in Specific Populations (8.1, 8.3)].
Advise females of reproductive potential to use effective contraception during treatment and for at least 3 months after the last dose of IMFINZI [see Use in Specific Populations (8.3)]. - Lactation: Advise female patients not to breastfeed while taking IMFINZI and for at least 3 months after the last dose [see Warnings and Precautions (5.10) and Use in Specific Populations (8.2)].
Manufactured for:
AstraZeneca Pharmaceuticals LP
Wilmington, DE 19850By: AstraZeneca UK Limited
1 Francis Crick Ave.
Cambridge, England CB2 0AA
US License No. 2043IMFINZI is a registered trademark of AstraZeneca group of companies.
© AstraZeneca 2020 -
MEDICATION GUIDE
MEDICATION GUIDE
IMFINZI® (im-FIN-zee)
(durvalumab)
InjectionWhat is the most important information I should know about IMFINZI?
IMFINZI is a medicine that may treat a type of cancer in the bladder and urinary tract or certain types of lung cancer by working with your immune system.
IMFINZI can cause your immune system to attack normal organs and tissues and can affect the way they work. These problems can sometimes become serious or life-threatening and can lead to death.
Call or see your healthcare provider right away if you develop any symptoms of the following problems or these symptoms get worse:
Lung problems (pneumonitis). Signs and symptoms of pneumonitis may include:
- new or worsening cough
- shortness of breath
- chest pain
Liver problems (hepatitis). Signs and symptoms of hepatitis may include:
- yellowing of your skin or the whites of your eyes
- severe nausea or vomiting
- pain on the right side of your stomach area (abdomen)
- drowsiness
- dark urine (tea colored)
- bleeding or bruising more easily than normal
- feeling less hungry than usual
Intestinal problems (colitis). Signs and symptoms of colitis may include:
- diarrhea or more bowel movements than usual
- stools that are black, tarry, sticky, or have blood or mucus
- severe stomach area (abdomen) pain or tenderness
Hormone gland problems (especially the thyroid, adrenals, pituitary, and pancreas). Signs and symptoms that your hormone glands are not working properly may include:
- headaches that will not go away or unusual headaches
- extreme tiredness
- weight gain or weight loss
- dizziness or fainting
- feeling more hungry or thirsty than usual
- hair loss
- changes in mood or behavior, such as decreased sex drive, irritability, or forgetfulness
- feeling cold
- constipation
- your voice gets deeper
- urinating more often than usual
- nausea or vomiting
- stomach area (abdomen) pain
Kidney problems, including nephritis and kidney failure. Signs of kidney problems may include:
- decrease in the amount of urine
- blood in your urine
- swelling of your ankles
- loss of appetite
Skin problems. Signs of these problems may include:
- rash
- itching
- skin blistering
Problems in other organs. Signs and symptoms may include:
- neck stiffness
- headache
- confusion
- fever
- chest pain, shortness of breath, or irregular heartbeat (myocarditis)
- changes in mood or behavior
- low red blood cells (anemia)
- excessive bleeding or bruising
- muscle weakness or muscle pain
- blurry vision, double vision, or other vision problem
- eye pain or redness
Severe Infections. Signs and symptoms may include:
- fever
- cough
- frequent urination
- pain when urinating
- flu-like symptoms
Severe infusion reactions. Signs and symptoms of severe infusion reactions may include:
- chills or shaking
- itching or rash
- flushing
- shortness of breath or wheezing
- dizziness
- fever
- feel like passing out
- back or neck pain
- facial swelling
Getting medical treatment right away may help keep these problems from becoming more serious.
Your healthcare provider will check you for these problems during your treatment with IMFINZI. Your healthcare provider may treat you with corticosteroid or hormone replacement medicines. Your healthcare provider may delay or completely stop treatment with IMFINZI, if you have severe side effects.
What is IMFINZI?
IMFINZI is a prescription medicine used to treat adults with:
-
a type of cancer in the bladder and urinary tract called urothelial carcinoma. IMFINZI may be used when your urothelial carcinoma:
- o has spread or cannot be removed by surgery, and
- o you have tried chemotherapy that contains platinum, and it did not work or is no longer working.
-
a type of lung cancer called non-small cell lung cancer (NSCLC). IMFINZI may be used when your NSCLC:
- o has not spread outside your chest
- o cannot be removed by surgery, and
- o has responded or stabilized with initial treatment with chemotherapy that contains platinum, given at the same time as radiation therapy.
-
a type of lung cancer called small cell lung cancer (SCLC). IMFINZI may be used with the chemotherapy medicines etoposide and carboplatin or cisplatin as your first treatment when your SCLC:
- o has spread within your lungs or to other parts of the body, (extensive-stage small cell lung cancer, or ES-SCLC).
It is not known if IMFINZI is safe and effective in children.
Before you receive IMFINZI, tell your healthcare provider about all of your medical conditions, including if you:
- have immune system problems such as Crohn’s disease, ulcerative colitis, or lupus.
- have had an organ transplant.
- have lung or breathing problems.
- have liver problems.
- are being treated for an infection.
- are pregnant or plan to become pregnant. IMFINZI can harm your unborn baby. If you are able to become pregnant, you should use an effective method of birth control during your treatment and for at least 3 months after the last dose of IMFINZI. Talk to your healthcare provider about birth control methods that you can use during this time. Tell your healthcare provider right away if you become pregnant during treatment with IMFINZI.
- are breastfeeding or plan to breastfeed. It is not known if IMFINZI passes into your breast milk. Do not breastfeed during treatment and for at least 3 months after the last dose of IMFINZI.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How will I receive IMFINZI?
- Your healthcare provider will give you IMFINZI into your vein through an intravenous (IV) line over 60 minutes.
- IMFINZI is usually given every 2, 3 or 4 weeks.
- Your healthcare provider will decide how many treatments you need.
- Your healthcare provider will test your blood to check you for certain side effects.
- If you miss any appointments, call your healthcare provider as soon as possible to reschedule your appointment.
What are the possible side effects of IMFINZI?
IMFINZI can cause serious side effects, including:
See "What is the most important information I should know about IMFINZI?"
The most common side effects of IMFINZI in people with urothelial carcinoma include:
- feeling tired
- muscle or bone pain
- constipation
- decreased appetite
- nausea
- swelling of your arms and legs
- urinary tract infection
The most common side effects of IMFINZI in people with NSCLC include:
- cough
- feeling tired
- inflammation in the lungs (pneumonitis)
- upper respiratory tract infections
- shortness of breath
- rash
The most common side effects of IMFINZI when used with other anticancer medicines in people with ES-SCLS include:
- nausea
- hair loss
- feeling tired or weak
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of IMFINZI. Ask your healthcare provider or pharmacist for more information.
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of IMFINZI.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. If you would like more information about IMFINZI, talk with your healthcare provider. You can ask your healthcare provider for information about IMFINZI that is written for health professionals.
What are the ingredients in IMFINZI?
Active ingredient: durvalumab
Inactive ingredients: L-histidine, L-histidine hydrochloride monohydrate, α,α-trehalose dihydrate, polysorbate 80, water for injection, USP.
Manufactured for: AstraZeneca Pharmaceuticals LP, Wilmington, DE 19850
By: AstraZeneca UK Limited
1 Francis Crick Ave.
Cambridge, England CB2 0AA
US License No. 2043
IMFINZI is a registered trademark of AstraZeneca group of companies.
For more information, call 1-800-236-9933 or go to www.IMFINZI.com
© AstraZeneca 2020
This Medication Guide has been approved by the U.S. Food and Drug Administration. Issued: 3/2020
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
NDC: 0310-4500-12
Rx only
IMFINZI™
(durvalumab)
Injection120 mg/2.4 mL
(50 mg/mL)For Intravenous Infusion After Dilution
Single-dose vial. Discard unused portion.
Store at 2° to 8°C (36° to 46°F).
Do not freeze or shake.
Keep vial in original carton to protect from light.
Attention Pharmacist: Dispense the accompanying Medication Guide to each patient.
Do not use if vial seal is broken or missing.Must dilute before use.
See prescribing information.AstraZeneca
-
PACKAGE/LABEL DISPLAY PANEL
NDC: 0310-4611-50
Rx only
IMFINZI™
(durvalumab)
Injection500 mg/10 mL
(50 mg/mL)For Intravenous Infusion After Dilution
Single-dose vial. Discard unused portion.
Store at 2° to 8°C (36° to 46°F).
Do not freeze or shake.
Keep vial in original carton to protect from light.
Attention Pharmacist: Dispense the accompanying Medication Guide to each patient.
Do not use if vial seal is broken or missing.Must dilute before use.
See prescribing information.AstraZeneca
-
INGREDIENTS AND APPEARANCE
IMFINZI
durvalumab injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0310-4500 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DURVALUMAB (UNII: 28X28X9OKV) (DURVALUMAB - UNII:28X28X9OKV) DURVALUMAB 120 mg in 2.4 mL Inactive Ingredients Ingredient Name Strength HISTIDINE (UNII: 4QD397987E) 4.8 mg in 2.4 mL HISTIDINE MONOHYDROCHLORIDE MONOHYDRATE (UNII: X573657P6P) 6.5 mg in 2.4 mL TREHALOSE DIHYDRATE (UNII: 7YIN7J07X4) 250 mg in 2.4 mL POLYSORBATE 80 (UNII: 6OZP39ZG8H) 0.5 mg in 2.4 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0310-4500-12 1 in 1 CARTON 05/01/2017 1 2.4 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA761069 05/01/2017 IMFINZI
durvalumab injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0310-4611 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DURVALUMAB (UNII: 28X28X9OKV) (DURVALUMAB - UNII:28X28X9OKV) DURVALUMAB 500 mg in 10 mL Inactive Ingredients Ingredient Name Strength HISTIDINE (UNII: 4QD397987E) 20 mg in 10 mL HISTIDINE MONOHYDROCHLORIDE MONOHYDRATE (UNII: X573657P6P) 27 mg in 10 mL TREHALOSE DIHYDRATE (UNII: 7YIN7J07X4) 1040 mg in 10 mL POLYSORBATE 80 (UNII: 6OZP39ZG8H) 2 mg in 10 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0310-4611-50 1 in 1 CARTON 05/01/2017 1 10 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA761069 05/01/2017 Labeler - AstraZeneca Pharmaceuticals LP (054743190) Registrant - AstraZeneca PLC (230790719)
Trademark Results [IMFINZI]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 IMFINZI 86483410 4887954 Live/Registered |
AstraZeneca AB 2014-12-18 |
 IMFINZI 79218364 5488935 Live/Registered |
AstraZeneca AB 2017-08-24 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.