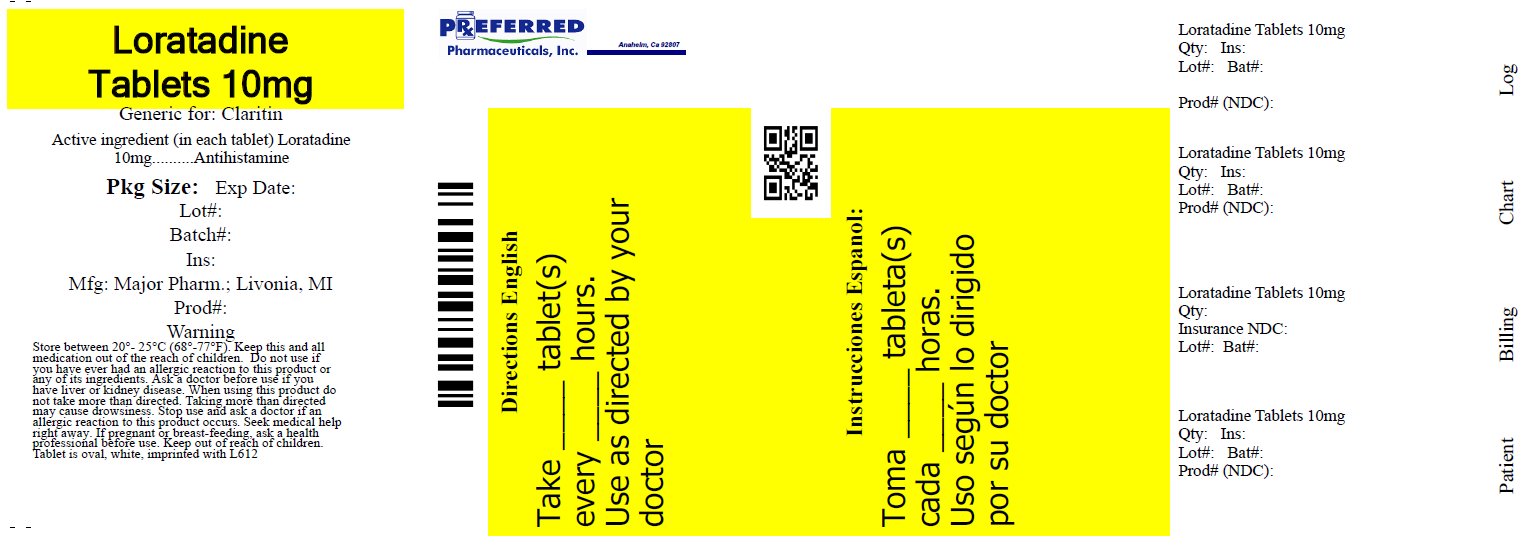
Loratadine by Preferred Pharmaceuticals, Inc. LORATADINE tablet
Loratadine by
Drug Labeling and Warnings
Loratadine by is a Otc medication manufactured, distributed, or labeled by Preferred Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product
do not take more than directed. Taking more than directed may cause drowsiness.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
COMPARE TO the active ingredient of CLARITIN®
Non-Drowsy* - Allergy
Original Prescription Strength
*When taken as directed. See Drug Facts Panel.
Loratadine Tablets, 10 mg/Antihistamine
Indoor & Outdoor Allergies
24 HOUR
Actual Size
24 Hour Relief of:
Sneezing; Runny Nose; Itchy, Watery Eyes; Itchy Throat or Nose
NDC: 68788-7653
Repackaged by Preferred Pharmaceuticals, Inc.
-
INGREDIENTS AND APPEARANCE
LORATADINE
loratadine tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68788-7653(NDC:0904-6852) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 10 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) Product Characteristics Color WHITE Score no score Shape OVAL Size 8mm Flavor Imprint Code L612 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68788-7653-0 10 in 1 BOTTLE; Type 0: Not a Combination Product 02/28/2020 2 NDC: 68788-7653-1 14 in 1 BOTTLE; Type 0: Not a Combination Product 02/28/2020 3 NDC: 68788-7653-5 15 in 1 BOTTLE; Type 0: Not a Combination Product 02/28/2020 4 NDC: 68788-7653-3 30 in 1 BOTTLE; Type 0: Not a Combination Product 02/28/2020 5 NDC: 68788-7653-9 90 in 1 BOTTLE; Type 0: Not a Combination Product 02/28/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076301 02/28/2020 Labeler - Preferred Pharmaceuticals, Inc. (791119022) Registrant - Preferred Pharmaceuticals, Inc. (791119022) Establishment Name Address ID/FEI Business Operations Preferred Pharmaceuticals, Inc. 791119022 REPACK(68788-7653)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.