4383 FIRST AID KIT- 4383 first aid kit 4384 FIRST AID KIT- 4384 first aid kit
4384 First Aid Kit by
Drug Labeling and Warnings
4384 First Aid Kit by is a Otc medication manufactured, distributed, or labeled by Honeywell Safety Products USA, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
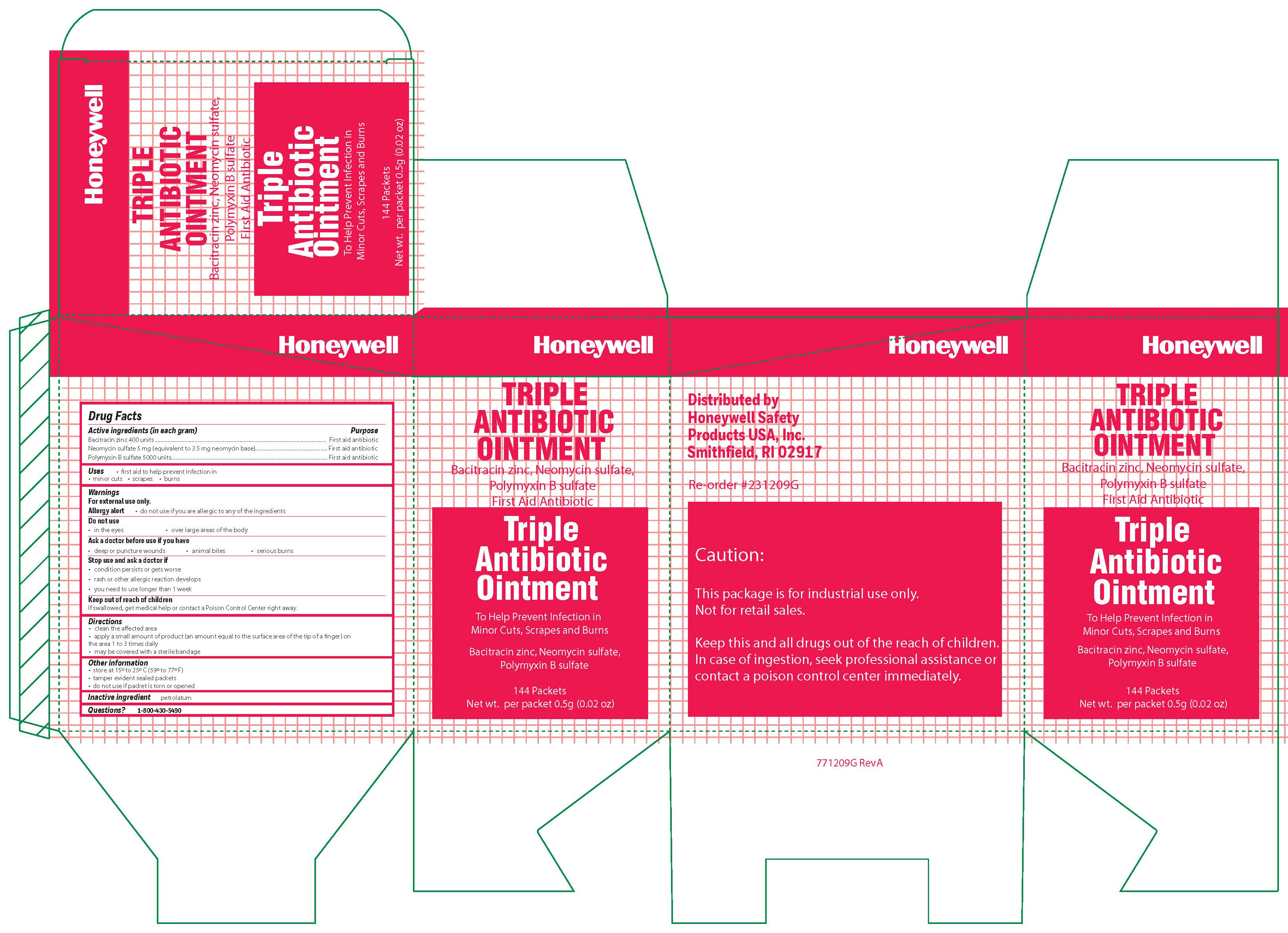
- Triple Active ingredient
- Triple Purpose
- Triple Uses
-
Triple
Warnings
For external use only
Allergy alert: do not use if you are allergic to any of the ingredients
Do not use
- in the eyes
- over large areas of the body
- Ask a doctor before use if you have
- a deep or puncture wounds
- animal bites
- serious burns
- Triple Directions
- Triple Other information
- Triple Inactive ingredient
- Triple Questions?
- BZK Wipe Active ingredient
- BZK Wipe Purpose
- BzK Wipe Uses
-
BZK Wipe
Warnings
For external use onlyDo not use
- in the eyes or over large areas of the body
- on mucous membranes
- on irritated skin
- in case of deep puncture wounds, animal bites or serious burns, consult a doctor
- longer than 1 week unless directed by a doctor
- BZK Wipe Directions
- BZK Wipe Other information
- BZK Wipe Inactive ingredient
- BZK Wipe Questions
- Eyewash Active ingredient
- Eyewassh Purpose
- Eyewash Uses
-
Eyewash
Warnings
For external use only
- Obtain immediate medical treatment for all open wounds in or near eyes.
- To avoid contamination, do not touch tip of container to any surface.
- Do not reuse.
- Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away.
- Eyeash Directions
- Eyewash Inactive ingredients
- Eyeash Questions
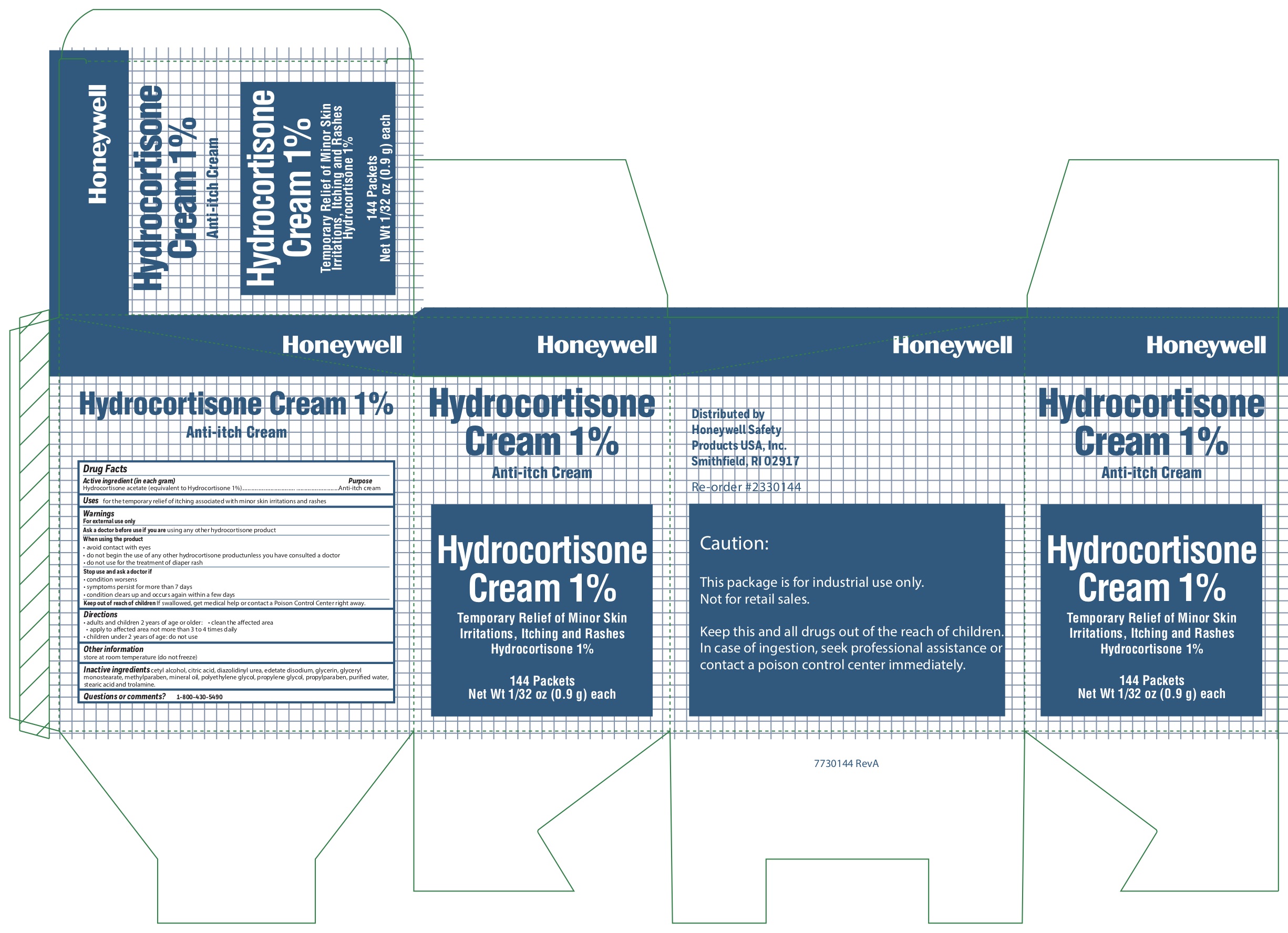
- Hydrocortisone Active ingredient (in each gram)
- Hydrcortisone Purpose
- Hydrocortisone Uses
-
Hydrocortisone
Warnings
For external use onlyWhen using the product
- avoid contact with eyes
- do not begin use of any other hydrocortisone product unless you have consulted a doctor
- do not use for the treatment of diaper rash
- Hydrocortisone Directions
- Hydrocortisone Other information
- Hydrocortisone Inactive ingredients
- Hydrocortisone Questions or Comments?
- Hand Sanitizer Active ingredient
- Hand Sanitizer Purpose
- Hand Sanitizer Uses
- Hand Sanitizer Warnings
- Hand Sanitizer Directions
- Hand Sanitizer Other information
- Hand Sanitizer Inactive ingredients
- Hand Sanitizer Questions or Comments?
- Isotonic Solution for Irrigation.
-
Description
NaCL Irrigation
Each 100 mL contains:
Sodium Chloride USP 0.9 g; Water for Injection USP qspH adjusted with Hydrochloric Acid NF
pH: 5.0 (4.5–7.0) Calculated Osmolarity: 310 mOsmol/literConcentration of Electrolytes (mEq/liter): Sodium 154; Chloride 154
0.9% Sodium Chloride Irrigation USP is sterile, nonpyrogenic, isotonic and contains no bacteriostatic or antimicrobial agents.The formula of the active ingredient is:
Ingredient Molecular Formula Molecular Weight
Sodium Chloride USP NaCl 58.44The plastic container is a copolymer of ethylene and propylene formulated and developed for parenteral drugs. The copolymer contains no plasticizers and exhibits virtually no leachability. The plastic container is also virtually impermeable to vapor transmission and, therefore, requires no overwrap to maintain the proper drug concentration. The safety of the plastic container has been confirmed by biological evaluation procedures. The material passes Class Vl testing as specified in the U.S. Pharmacopeia for Biological Tests — Plastic Containers. These tests have shown that the container is nontoxic and biologically inert.
The PIC™ Container is PVC-free and DEHP-free.
Ingredient Molecular Formula Molecular Weight
Sodium Chloride USP NaCl 58.44The plastic container is a copolymer of ethylene and propylene formulated and developed for parenteral drugs. The copolymer contains no plasticizers and exhibits virtually no leachability. The plastic container is also virtually impermeable to vapor transmission and, therefore, requires no overwrap to maintain the proper drug concentration. The safety of the plastic container has been confirmed by biological evaluation procedures. The material passes Class Vl testing as specified in the U.S. Pharmacopeia for Biological Tests — Plastic Containers. These tests have shown that the container is nontoxic and biologically inert.
The PIC™ Container is PVC-free and DEHP-free.
-
Clinical Pharmacology
NaCL Irrigant
0.9% Sodium Chloride Irrigation USP is utilized for a variety of clinical indications such as sterile irrigation of body cavities, tissues or wounds, indwelling urethral catheters, surgical drainage tubes, and for washing, rinsing or soaking surgical dressings, instruments and laboratory specimens. It also serves as a diluent or vehicle for drugs used for irrigation or other pharmaceutical preparations.
0.9% Sodium Chloride Irrigation USP provides an isotonic saline irrigation identical in composition with 0.9% Sodium Chloride Injection USP (normal saline).
Physiological irrigation solutions are considered generally compatible with living tissues and organs.
Sodium, the major cation of the extracellular fluid, functions primarily in the control of water distribution, fluid balance, and osmotic pressure of body fluids. Sodium is also associated with chloride and bicarbonate in the regulation of the acid-base equilibrium of body fluid.
Chloride, the major extracellular anion, closely follows the metabolism of sodium, and changes in the acid-base balance of the body are reflected by changes in the chloride concentration.
- Indication and Usage NaCl Irrigant
- Contraindications NaCl Irrigant
-
Warnings
NaCl Irrigant
FOR IRRIGATION ONLY. NOT FOR INJECTION.
Irrigating fluids have been demonstrated to enter the systemic circulation in relatively large volumes; thus, irrigation solutions must be regarded as systemic drugs. Absorption of large amounts can cause fluid and/or solute overload resulting in dilution of serum electrolyte concentrations, overhydration, congested states or pulmonary edema.
The risk of dilutional states is inversely proportional to the electrolyte concentrations of the administered parenteral solutions. The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentration.
Do not warm above 150°F (66°C).
After opening container, its contents should be used promptly to minimize the possibility of bacterial growth or pyrogen formation.
Discard unused portion of irrigating solution since it contains no preservatives.
-
Precautions
NaCl Irrigant
General
Use aseptic technique when preparing and administering sterile irrigation solutions.Use only if solution is clear and container and seal are intact.
Do not use for irrigation that may result in absorption of large amounts of fluid into the blood.
Caution should be observed when the solution is used for continuous irrigation or allowed to "dwell" inside body cavities because of possible absorption into the blood stream and the production of circulatory overload.
When used for irrigation via appropriate irrigation equipment, the administration set should be attached promptly. Unused portions should be discarded and a fresh container of appropriate size used for the start up of each cycle or repeat procedure. For repeated irrigations of urethral catheters, a separate container should be used for each patient.
Laboratory Tests
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid-base balance after prolonged irrigation, when fluid absorption is suspected, or whenever the condition of the patient warrants such evaluation.
Drug Interactions
Some additives may be incompatible. Consult with pharmacist.When introducing additives, use aseptic technique.Mix thoroughly.
Do not store.
Carcinogenesis, Mutagenesis, Impairment of FertilityStudies with 0.9% Sodium Chloride Irrigation USP have not been performed to evaluate carcinogenic potential, mutagenic potential, or effects on fertility.
Pregnancy
Teratogenic Effects
Animal reproduction studies have not been conducted with 0.9% Sodium Chloride Irrigation USP. It is also not known whether 0.9% Sodium Chloride Irrigation USP can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. 0.9% Sodium Chloride Irrigation USP should be given to a pregnant woman only if clearly needed.
Labor and Delivery
Safety and effectiveness of 0.9% Sodium Chloride Irrigation USP during labor and delivery have not been established. Caution should be exercised, and the fluid balance, glucose and electrolyte concentrations, and acid-base balance, of both mother and fetus should be evaluated periodically or whenever warranted by the condition of the patient or fetus.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when 0.9% Sodium Chloride Irrigation USP is administered to a nursing woman.
Pediatric Use
The safety and effectiveness of 0.9% Sodium Chloride Irrigation USP in pediatric patients have not been established. Its limited use in pediatric patients has been inadequate to fully define proper dosage and limitations for use.
Geriatric Use
Clinical studies of 0.9% Sodium Chloride Irrigation USP did not include a sufficient number of patients age 65 years and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function.Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.Frequent laboratory determinations and clinical evaluations are recommended to monitor changes in blood glucose, electrolyte concentrations, and renal function.
-
Adverse Reactions
Possible adverse effects arising from the irrigation of body cavities, tissues, or indwelling catheters and tubes can be minimized when proper procedures are followed. Displaced catheters or drainage tubes can lead to irrigation or infiltration of unintended structures or cavities. Excessive volume or pressure during irrigation of closed cavities may cause undue distension or disruption of tissues. Accidental contamination from careless technique may transmit infection.
If an adverse reaction does occur, discontinue administration of the irrigant, evaluate the patient, institute appropriate therapeutic countermeasures, and save the remainder of the fluid for examination if deemed necessary.
- Overdosage
-
Dosage and Administration
As required for irrigation.
When used as a diluent, or vehicle for other drugs, the drug manufacturer's recommendations should be followed.
Some additives may be incompatible. Consult with pharmacist. When introducing additives, use aseptic techniques. Mix thoroughly. Do not store.
Solutions should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permits.
-
How Supplied
0.9% Sodium Chloride Irrigation USP is supplied sterile and nonpyrogenic in PIC™ (Plastic Irrigation Container). The 1000 mL and 500 mL containers are packaged 16 per case, the 2000 mL containers are packaged 8 per case, and the 4000 mL containers are packaged 4 per case.
0.9% Sodium Chloride Irrigation USP
NDC Cat. No. REF SIZE
0264-2201-00 R5200-01 1000 mL
0264-2201-10 R5201-01 500 mL
0264-2201-50 R5205-01 2000 mL
0264-2201-70 R5207 ,,,,4000 mLExposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. Protect from freezing. It is recommended that the product be stored at room temperature (25°C); however, brief exposure up to 40°C does not adversely affect the product.
Do not warm above 150°F (66°C).
- SPL Unclassified Section
-
DIRECTIONS FOR USE OF PIC™ (PLASTIC IRRIGATION CONTAINER)
Not for injection.
Aseptic technique is required.
Caution – Before use, perform the following checks:
(a) Read the label. Ensure solution is the one ordered and is within the expiration date.
(b) Invert container and inspect the solution in good light for cloudiness, haze, or particulate matter; check the
container for leakage or damage. Any container which is suspect should not be used.Use only if solution is clear and container and seal are intact
Single unit container. Discard unused portion.
Outer Closure Removal – Grasp the container with one hand and turn the breakaway ring counterclockwise with the other hand until slight resistance is felt. Then, twisting the container in the opposite direction, turn the breakaway ring sharply until the entire outer cap is loose and can be lifted off.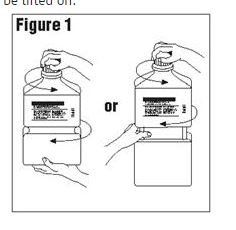
Figure 1
Connect the administration set through the sterile set port according to set instructions or remove screw cap and pour.
[Fig 2]
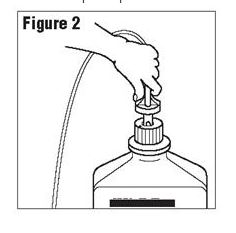
Figure 2
Do not warm above 150°F (66°C) to assure minimal bottle distortion. Keep bottles upright.
- SPL Unclassified Section
- Alcohol Wipe Active ingredient
- Alcohol Wipe Purpose
- Alcohol Wipe Uses
- Alcohol Wipe Warnings
- Alcohol Wipe Directions
- Alcohol Wipe Other information
- Alcohol Wipe Inactive ingredients
- Alcohol Wipe Questions
-
4383
148820 Kit Contents
1 EYE DRESS PKT W/4 ADH STRIPS
1 ADHESIVE TAPE W/P 1" X 10YDS
1 ADH BDG, CLOTH, 1"X3", 16 PER
1 FIRST AID GUIDE ASHI
1 EMERGENCY SURVIVAL BLANKET
2 ELASTIC ROLLED GZ 3" ST
2 ELASTIC ROLLED GZ 4" ST
2 BLOODSTOPPER
8 ABD COMBINE PAD 5" X 9"
1 GZE PADS STERILE 3"X 3" 10'S
1 GZE PADS STERILE 4"X 4" 10'S
6 ABD PADS 8"X10" STERILE
4 MULTI-TRAUMA DRESSING 12"X30"
2 MEDI-RIP BANDAGE 6"X5YDS EA
4 ELASTIC BANDAGE 3" X 4.5YD
2 ELASTIC BANDAGE 6" X 4.5 YD
1 CPR FILTERSHIELD 77-100
1 FLASHLIGHT STD
1 RADIO AM FREQ BATTERY POWERED
1 WATER JEL FACIAL DRS 12X16 EA
1 TRIPLE BIOTIC .5 GRAM PKT 20
1 SOD. CHLORIDE 0.9% 500ML EA
1 4OZ BFS EYEWASH TRILINGUAL BOTTLE
1 SPHYG ANEROID(NO PIN) ADULT
1 STETHESCOPE NURSES VARIOUS COLORS
1 PENLIGHT DISPOSABLE EACH
1 FORCEPS WITH MAGNIFIER
1 SCISSOR UTILITY SHEARS 7-1/4"
1 SCISSOR LISTER BDG S/S 5 1/2"
1 BG POLY 32" x 34"
4 SPLINT BOARD W/PAD SML 12"X6"
1 LBL STOCK 6-3/8"X4"
1 LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
1 10 PR NITRILE GLVES ZIP BAG
4 EMER.YELLOW BLKT 54"X80" POLYP
1 ANTISEPTIC HAND GEL 4OZ
1 ANTISEPTIC WIPES 20'S ZIP LOCK
1 HYDROCORTISONE 20'S ZIP LOCK
1 ALCOHOL WIPES 50'S ZIP LOCK
1 BAG FOR DELUXE TRAUMA KIT
4 COLD PACK 5"X 9" BULK
2 TRI BNDG NON WOVEN 40"X40"X56"
1 WOVEN KNUCKLE 8'S
1 FINGERTIP "T" 8/BX
-
4384
Z148820 Kit Contents
1 EYE DRESS PKT W/4 ADH STRIPS
1 ADHESIVE TAPE W/P 1" X 10YDS
1 ADH BDG, CLOTH, 1"X3", 16 PER
1 FIRST AID GUIDE ASHI
1 EMERGENCY SURVIVAL BLANKET
2 ELASTIC ROLLED GZ 3" ST
2 ELASTIC ROLLED GZ 4" ST
2 BLOODSTOPPER
8 ABD COMBINE PAD 5" X 9"
1 GZE PADS STERILE 3"X 3" 10'S
1 GZE PADS STERILE 4"X 4" 10'S
6 ABD PADS 8"X10" STERILE
4 MULTI-TRAUMA DRESSING 12"X30"
2 MEDI-RIP BANDAGE 6"X5YDS EA
4 ELASTIC BANDAGE 3" X 4.5YD
2 ELASTIC BANDAGE 6" X 4.5 YD
1 CPR FILTERSHIELD 77-100
1 FLASHLIGHT STD
1 RADIO AM FREQ BATTERY POWERED
1 WATER JEL FACIAL DRS 12X16 EA
1 TRIPLE BIOTIC .5 GRAM PKT 20
1 SOD. CHLORIDE 0.9% 500ML EA
1 4OZ BFS EYEWASH TRILINGUAL BOTTLE
1 SPHYG ANEROID(NO PIN) ADULT
1 STETHESCOPE NURSES VARIOUS COLORS
1 PENLIGHT DISPOSABLE EACH
1 FORCEPS WITH MAGNIFIER
1 SCISSOR UTILITY SHEARS 7-1/4"
1 SCISSOR LISTER BDG S/S 5 1/2"
1 BG POLY 32" x 34"
4 SPLINT BOARD W/PAD SML 12"X6"
1 LBL STOCK 6-3/8"X4"
1 LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
1 10 PR NITRILE GLVES ZIP BAG
4 EMER.YELLOW BLKT 54"X80" POLYP
1 ANTISEPTIC HAND GEL 4OZ
1 ANTISEPTIC WIPES 20'S ZIP LOCK
1 HYDROCORTISONE 20'S ZIP LOCK
1 ALCOHOL WIPES 50'S ZIP LOCK
1 BAG FOR DELUXE TRAUMA KIT
4 COLD PACK 5"X 9" BULK
2 TRI BNDG NON WOVEN 40"X40"X56"
1 WOVEN KNUCKLE 8'S
1 FINGERTIP "T" 8/BX
- Triple Principal Display Panel
- BZK Wipe Principal Display Panel
- Eyewash Principal Display Panel
- Hydrocortisone Principal Display Panel
- Hand Sanitizer Principal Display Panel
- Principal Display Panel 500 ml Container
- Alcohol Wipe Principal Display Panel
- 4383 Kit Label 148820
- 4384 Kit Label Z148820
-
INGREDIENTS AND APPEARANCE
4383 FIRST AID KIT
4383 first aid kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0498-4383 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-4383-01 1 in 1 KIT 09/13/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 118 mL Part 2 20 PACKET 10 g Part 3 20 PACKET 28 mL Part 4 20 PACKET 18 g Part 5 1 BOTTLE, PLASTIC 118 mL Part 6 50 POUCH 20 mL Part 7 1 CONTAINER 500 mL Part 8 20 PACKET 18 g Part 1 of 8 EYESALINE EMERGENCY EYEWASH
purified water liquidProduct Information Item Code (Source) NDC: 0498-0100 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 98.6 mL in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0100-02 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 12/18/2018 Part 2 of 8 TRIPLE ANTIBIOTIC
bacitracin zinc, polymyxin b sulfate, neomycin sulfate ointmentProduct Information Item Code (Source) NDC: 0498-0750 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [iU] in 1 g BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [iU] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0750-36 0.5 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333B 09/19/2018 Part 3 of 8 ANTISEPTIC TOWELETTE
benzalkonium chloride liquidProduct Information Item Code (Source) NDC: 0498-0501 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0501-00 1.4 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 12/22/2017 Part 4 of 8 HYDROCORTISONE
anti-itch cream ointmentProduct Information Item Code (Source) NDC: 0498-0800 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE ACETATE (UNII: 3X7931PO74) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE ACETATE 1 g in 100 g Inactive Ingredients Ingredient Name Strength LIGHT MINERAL OIL (UNII: N6K5787QVP) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) CETYL ALCOHOL (UNII: 936JST6JCN) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) EDETATE DISODIUM (UNII: 7FLD91C86K) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) TROLAMINE (UNII: 9O3K93S3TK) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 03/06/2013 10/15/2019 Part 5 of 8 INSTANT HAND SANITIZER
alcohol liquidProduct Information Item Code (Source) NDC: 59898-420 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 mL in 100 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) TRIISOPROPANOLAMINE (UNII: W9EN9DLM98) CARBOMER COPOLYMER TYPE A (UNII: 71DD5V995L) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59898-420-12 118 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 04/15/2010 Part 6 of 8 ALCOHOL WIPE
isopropyl alcohol swabProduct Information Item Code (Source) NDC: 0498-0143 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0143-04 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 09/18/2018 Part 7 of 8 SODIUM CHLORIDE
sodium chloride irrigantProduct Information Item Code (Source) NDC: 0264-2201 Route of Administration IRRIGATION Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 0.9 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 500 mL in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA016733 09/14/2009 Part 8 of 8 HYDROCORTISONE
anti-itch creamProduct Information Item Code (Source) NDC: 0498-0801 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE ACETATE (UNII: 3X7931PO74) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE ACETATE 1 g in 100 g Inactive Ingredients Ingredient Name Strength LIGHT MINERAL OIL (UNII: N6K5787QVP) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) CETYL ALCOHOL (UNII: 936JST6JCN) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) EDETATE DISODIUM (UNII: 7FLD91C86K) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) TROLAMINE (UNII: 9O3K93S3TK) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 10/15/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/13/2018 4384 FIRST AID KIT
4384 first aid kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0498-4384 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-4384-01 1 in 1 KIT 09/13/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 118 mL Part 2 20 PACKET 10 g Part 3 20 PACKET 28 mL Part 4 20 PACKET 18 g Part 5 1 BOTTLE, PLASTIC 118 mL Part 6 50 POUCH 20 mL Part 7 1 CONTAINER 500 mL Part 8 20 PACKET 18 g Part 1 of 8 EYESALINE EMERGENCY EYEWASH
purified water liquidProduct Information Item Code (Source) NDC: 0498-0100 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 98.6 mL in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0100-02 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 12/18/2018 Part 2 of 8 TRIPLE ANTIBIOTIC
bacitracin zinc, polymyxin b sulfate, neomycin sulfate ointmentProduct Information Item Code (Source) NDC: 0498-0750 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [iU] in 1 g BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [iU] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0750-36 0.5 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333B 09/19/2018 Part 3 of 8 ANTISEPTIC TOWELETTE
benzalkonium chloride liquidProduct Information Item Code (Source) NDC: 0498-0501 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0501-00 1.4 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 12/22/2017 Part 4 of 8 HYDROCORTISONE
anti-itch cream ointmentProduct Information Item Code (Source) NDC: 0498-0800 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE ACETATE (UNII: 3X7931PO74) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE ACETATE 1 g in 100 g Inactive Ingredients Ingredient Name Strength LIGHT MINERAL OIL (UNII: N6K5787QVP) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) CETYL ALCOHOL (UNII: 936JST6JCN) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) EDETATE DISODIUM (UNII: 7FLD91C86K) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) TROLAMINE (UNII: 9O3K93S3TK) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0800-34 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 03/06/2013 10/15/2019 Part 5 of 8 INSTANT HAND SANITIZER
alcohol liquidProduct Information Item Code (Source) NDC: 59898-420 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 mL in 100 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) TRIISOPROPANOLAMINE (UNII: W9EN9DLM98) CARBOMER COPOLYMER TYPE A (UNII: 71DD5V995L) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59898-420-12 118 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 04/15/2010 Part 6 of 8 ALCOHOL WIPE
isopropyl alcohol swabProduct Information Item Code (Source) NDC: 0498-0143 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0143-04 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 09/18/2018 Part 7 of 8 SODIUM CHLORIDE
sodium chloride irrigantProduct Information Item Code (Source) NDC: 0264-2201 Route of Administration IRRIGATION Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 0.9 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 500 mL in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA016733 09/14/2009 Part 8 of 8 HYDROCORTISONE
anti-itch creamProduct Information Item Code (Source) NDC: 0498-0801 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE ACETATE (UNII: 3X7931PO74) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE ACETATE 1 g in 100 g Inactive Ingredients Ingredient Name Strength LIGHT MINERAL OIL (UNII: N6K5787QVP) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) CETYL ALCOHOL (UNII: 936JST6JCN) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) EDETATE DISODIUM (UNII: 7FLD91C86K) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) TROLAMINE (UNII: 9O3K93S3TK) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 10/15/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/13/2018 Labeler - Honeywell Safety Products USA, Inc. (079287321) Registrant - Honeywell Safety Products USA, Inc. (079287321) Establishment Name Address ID/FEI Business Operations B. Braun Medical Inc. 037425308 label(0264-2201) Establishment Name Address ID/FEI Business Operations Honeywell Safety Products USA, Inc 079287321 pack(0498-4383, 0498-4384) Establishment Name Address ID/FEI Business Operations Water-Jel Technologies 155522589 manufacture(0498-0750, 0498-0800, 59898-420, 0498-0801) Establishment Name Address ID/FEI Business Operations Honeywell Safety Products USA, Inc. 167518617 manufacture(0498-0100) Establishment Name Address ID/FEI Business Operations Changzhou Maokang Medical 421317073 manufacture(0498-0501, 0498-0143)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.