MOXISOLV- moxidectin injection, solution
MoxiSolv by
Drug Labeling and Warnings
MoxiSolv by is a Animal medication manufactured, distributed, or labeled by Bimeda, Inc., Bimeda-MTC Animal Health. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
SPL UNCLASSIFIED SECTION
MoxiSolvTM Injection
(moxidectin)Injectable Solution for Beef and Nonlactating Dairy Cattle
Antiparasitic
Contains 10 mg moxidectin/mL
Not for use in female dairy cattle 20 months of age or older (including dry dairy cows), veal calves, and calves less than 8 weeks of age.
For Treatment of Infections and Infestations Due to Internal and External Parasites of Cattle
Consult your veterinarian for assistance in the diagnosis, treatment and control of parasitism. -
DESCRIPTION
PRODUCT DESCRIPTION
MoxiSolv Injection (moxidectin) is a ready-to-use, sterile solution containing 1% moxidectin. Moxidectin is an endectocide in the milbemycin chemical class which shares the distinctive mode of action characteristic of macrocyclic lactones. MoxiSolv Injection is specially formulated to allow moxidectin to be absorbed from the site of injection and distributed internally to the areas of the body affected by endo- and/or ectoparasitism. Moxidectin binds selectively and with high affinity to glutamate-gated chloride ion
channels which are critical to the function of invertebrate nerve and muscle cells. This interferes with neurotransmission resulting in paralysis and elimination of the parasite -
INDICATIONS & USAGE
INDICATIONS
MoxiSolv Injection, when administered at the recommended dose level of 0.2 mg/2.2 lb (0.2 mg/kg) body weight, is effective in the treatment and control of the following internal and external parasites of cattle:
Gastrointestinal Roundworms
Ostertagia ostertagi - Adults and L4 (including inhibited Larvae)
Haemonchus placei - Adults
Trichostrongylus axei - Adults and L4
Trichostrongylus colubriformis - Adults and L4
Cooperia oncophora - Adults
Cooperia pectinata - Adults
Cooperia punctata - Adults and L4
Cooperia spatulata - Adults
Cooperia surnabada - Adults and L4
Nematodirus helvetianus - Adults
Oesophagostomum radiatum - Adults and L4
Trichuris spp. - Adults
Lungworms
Dictyocaulus viviparus - Adults and L4
Cattle Grubs
Hypoderma bovis
Hypoderma lineatumMites
Psoroptes ovis
(Psoroptes communis var. bovis)
Lice
Linognathus vituli
Solenopotes capillatusPersistent Activity
Moxidectin injectable has been proven to effectively protect cattle from reinfection with Dictyocaulus viviparus and Oesophagostomum radiatum for 42 days after treatment, Haemonchus placei for 35 days after treatment, and Ostertagia ostertagi and Trichostrongylus axei for 14 days after treatment.Management Considerations for External Parasites
For most effective external parasite control, MoxiSolv Injection (moxidectin) should be administered to all cattle in the herd. Cattle entering the herd following this administration should be treated prior to introduction. Consult your veterinarian or a livestock entomologist for the most appropriate time to administer MoxiSolv Injection in your location to effectively control external parasites. -
DOSAGE & ADMINISTRATION
DOSAGE
The recommended rate of administration for MoxiSolv Injection is 1 mL for each 110 lb (50 kg) body weight to provide 0.2 mg moxidectin/2.2 lb (0.2 mg/kg) body weight. The table below will assist in the calculation of the appropriate volume of injectable which must be administered based on the weight of animal being treated. Be careful not to overdose animals; estimate animal's body weight as closely as possible or weigh animals individually.Weight (lb) 165 220 330 440 550 660 770 880 990 1100 Weight (kg) 75 100 150 200 250 300 350 400 450 500 Dose (mL) 1.5 2,0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0 Do not underdose. Ensure each animal receives a complete dose based on a current body weight. Underdosing may result in ineffective treatment, and encourage the development of parasite resistance.
ADMINISTRATION
MoxiSolv Injection should be administered by subcutaneous injection under the loose skin in front of or behind the shoulder (Figure 1). Needles 1/2 to 3/4 inch in length and 16 to 18 gauge are recommended for subcutaneous injections. Use sterile, dry equipment and aseptic procedures when withdrawing and administering MoxiSolv.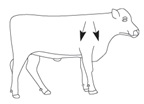
Figure 1. Sites for administration of MoxiSolv Injection
-
WARNINGS AND PRECAUTIONS
HUMAN WARNINGS
Not For Use in Humans. Keep this and all drugs out of the reach of childrenCONTACT INFORMATION:
To report suspected adverse drug events, for technical assistance or to obtain a copy of the Safety Data Sheet (SDS), contact Bimeda, Inc. at 1-888-524-6332. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or online at www.fda.gov/reportanimalae -
RESIDUE WARNING
RESIDUE WARNINGS
Cattle must not be slaughtered for human consumption within 21 days of treatment. This drug product is not approved for use in female dairy cattle 20 months of age or older, including dry dairy cows. Use in these cattle may cause drug residues in milk and/or in calves born to these cows. A withdrawal period has not been established for preruminating calves. Do not use in calves to be processed for veal. -
WARNINGS
ANIMAL SAFETY WARNINGS
Do not use in sick, debilitated, or underweight animals. In foreign countries there have been reports of adverse effects, including death. This product should not be used in calves less than 8 weeks of age because safety testing has not been done in the U.S. in calves less than 8 weeks of age.ENVIRONMENTAL WARNINGS
Studies indicate that when moxidectin comes in contact with the soil, it readily and tightly binds to the soil and becomes inactive. Free moxidectin may adversely affect fish and certain aquatic organisms. Do not contaminate water by direct application or by improper disposal of drug containers.OTHER WARNINGS
- Parasite resistance may develop to any dewormer, and has been reported for most classes of dewormers.
- Treatment with a dewormer used in conjunction with parasite management practices appropriate to the geographic area and the animal(s) to be treated may slow the development of parasite resistance.
- Fecal examinations or other diagnostic tests and parasite management history should be used to determine if the product is appropriate for the herd/flock, prior to the use of any dewormer. Following the use of any dewormer, effectiveness of treatment should be monitored (for example, with the use of a fecal egg count reduction test or another appropriate method).
- A decrease in a drug's effectiveness over time as calculated by fecal egg count reduction tests may indicate the development of resistance to the dewormer administered. Your parasite management plan should be adjusted accordingly based on regular monitoring.
-
PRECAUTIONS
PRECAUTIONS
MoxiSolv Injection (moxidectin) has been formulated specifically for subcutaneous injection in cattle and should not be given by other routes of administration. Subcutaneous injection can cause transient local tissue reaction that may result in trim loss of edible tissue at slaughter if animals are slaughtered within 35 days after treatment. This product should not be used in other animal species as severe adverse reactions, including fatalities in dogs, may result.
MoxiSolv Injection is effective against the migrating stage of cattle grubs (Hypoderma larvae). Treatment with MoxiSolv Injection (moxidectin) during the period when grubs are migrating through vital areas may cause undesirable host-parasite reactions. Killing H. lineatum when they are located in peri-esophageal tissues may cause bloat. Killing H. bovis when they are in the vertebral canal may cause staggering or hindlimb paralysis. Cattle should be treated as soon as possible after heel fly (warble fly) season to avoid this potential problem. Cattle treated with MoxiSolv Injection at the end of fly season can be retreated during the winter without danger of grub-related reactions. Consult your veterinarian for more information regarding these secondary grub reactions and the correct time to treat with MoxiSolv Injection. -
SPL UNCLASSIFIED SECTION
ANIMAL SAFETY
U.S. tolerance and toxicity studies have demonstrated that moxidectin injectable has an adequate margin of safety for use in cattle 8 weeks of age and older. No toxic signs were seen in growing cattle given up to 5 times the recommended dose. Calves as young as 8 weeks of age showed no toxic signs when treated with up to 3 times the recommended dose while nursing from cows concurrently treated with the recommended dose level of moxidectin injectable. Mild, transient ataxia was noted in growing cattle receiving 10 times the recommended dose and in bulls treated at 4.5 times the recommended dose. In breeding animals (bulls and cows in estrous and during early, mid and late pregnancy), treatment with at least 3 times the recommended dose had no effect on breeding performance.
Signs of toxicity include ataxia, excessive salivation, depression, and mydriasis. These signs usually occur within 12 to 48 hours post-treatment. -
STORAGE AND HANDLING
STORAGE
Store at 20°C - 25°C (68°F - 77°F); excursions permitted between 15°C - 30°C (59°F - 86°F). Use within 28 days of first puncture and puncture a maximum of 30 times with a needle or 5 times with a dosage delivery device. When using a draw-off spike or needle larger than 4-gauge, discard any remaining product immediately after use.DISPOSAL
Do not contaminate water by direct application or by improper disposal of drug containers. Dispose of containers in an approved landfill or by incineration. -
SPL UNCLASSIFIED SECTION
PACKAGE INFORMATION
MoxiSolv Injection is available in 200 mL and 500 mL plastic, multi-dose bottles.Restricted Drug (CA) - Use Only As Directed. Approved by FDA under ANADA # 200-788
Manufactured for
Bimeda, Inc.
Le Sueur, MN 56058
www.bimeda.com
Made in CanadaMoxiSolvTM is a trademark of Bimeda, Inc.
8MOX005 Rev. 07/24 - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MOXISOLV
moxidectin injection, solutionProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC: 61133-4013 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MOXIDECTIN (UNII: NGU5H31YO9) (MOXIDECTIN - UNII:NGU5H31YO9) MOXIDECTIN 10 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 61133-4013-2 500 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200788 10/02/2024 Labeler - Bimeda, Inc. (060492923) Registrant - Bimeda, Inc. (060492923) Establishment Name Address ID/FEI Business Operations Bimeda-MTC Animal Health 256232216 manufacture
Trademark Results [MoxiSolv]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 MOXISOLV 79310358 not registered Live/Pending |
BIMEDA ANIMAL HEALTH LIMITED 2021-03-09 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
