Phenobarbital by West-Ward Pharmaceuticals Corp PHENOBARBITAL tablet
Phenobarbital by
Drug Labeling and Warnings
Phenobarbital by is a Prescription medication manufactured, distributed, or labeled by West-Ward Pharmaceuticals Corp. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
DESCRIPTION
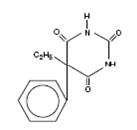
Phenobarbital is a barbituric acid derivative for oral administration and occurs as a white, odorless, slightly bitter powder that is soluble in chloroform, freely soluble in alcohol or ether, and slightly soluble in water. Its saturated solution has a pH of about 5.6. Chemically, it is 5-ethyl-5-phenylbarbituric acid with the molecular formula C12H12N2O3 (232.24). The structural formula is as follows:
Each Phenobarbital Tablet USP contains 15 mg, 30 mg, 60 mg or 100 mg of phenobarbital, USP.
Inactive Ingredients are as follows:
Anhydrous Lactose, Colloidal Silicon Dioxide, Corn Starch, Docusate Sodium with Sodium Benzoate, Lactose Monohydrate, Magnesium Stearate, Microcrystalline Cellulose, and Sodium Starch Glycolate.
- CLINICAL PHARMACOLOGY
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Phenobarbital is contraindicated in patients who are hypersensitive to barbiturates. In such patients, severe hepatic damage can occur from ordinary doses and is usually associated with dermatitis and involvement of parenchymatous organs. A personal or familial history of acute intermittent porphyria represents one of the few absolute contraindications to the use of barbiturates. Phenobarbital is also contraindicated in patients with marked impairment of liver function, or respiratory disease in which dyspnea or obstruction is evident. It should not be administered to persons with known previous addiction to the sedative/hypnotic group, since ordinary doses may be ineffectual and may contribute to further addiction.
- WARNINGS
-
PRECAUTIONS
General Precautions
Barbiturates induce liver microsomal enzyme activity. This accelerates the biotransformation of various drugs and is probably part of the mechanism of the tolerance encountered with barbiturates. Phenobarbital, therefore, should be used with caution in patients with decreased liver function. This drug should also be administered cautiously to patients with a history of drug dependence or abuse (see DRUG ABUSE AND DEPENDENCE).
Phenobarbital may decrease the potency of coumarin anticoagulants; therefore, patients receiving such concomitant therapy should have more frequent prothrombin determinations. As with other sedatives and hypnotics, elderly or debilitated patients may react to barbiturates with marked excitement, depression, or confusion.
The systemic effects of exogenous hydrocortisone and endogenous hydrocortisone (cortisol) may be diminished by phenobarbital. Thus, this product should be administered with caution to patients with borderline hypoadrenal function, regardless of whether it is of pituitary or of primary adrenal origin.
Information for Patients
Phenobarbital may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a car or operating machinery. The patient should be cautioned accordingly.
Drug Interactions
Phenobarbital in combination with alcohol, tranquilizers, and other central nervous system depressants has additive depressant effects, and the patient should be so advised. Patients taking this drug should be warned not to exceed the dosage recommended by their physician. Toxic effects and fatalities have occurred following overdoses of phenobarbital alone and in combination with other central nervous system depressants. Caution should be exercised in prescribing unnecessarily large amounts of phenobarbital for patients who have a history of emotional disturbances or suicidal ideation or who have misused alcohol and other CNS drugs (see OVERDOSAGE).
Usage in Pregnancy
Pregnancy Category B: Reproduction studies have been performed in animals and have revealed no evidence of impaired fertility or harm to the fetus due to phenobarbital. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
-
ADVERSE REACTIONS
The following adverse reactions have been reported:
CNS Depression
Residual sedation or “hangover”, drowsiness, lethargy, and vertigo. Emotional disturbances and phobias may be accentuated. In some persons, barbiturates such as phenobarbital repeatedly produce excitement rather than depression, and the patient may appear to be inebriated. Like other nonanalgesic hypnotic drugs, barbiturates, such as phenobarbital, when given in the presence of pain, may cause restlessness, excitement, and even delirium. Rarely, the use of barbiturates results in localized or diffused myalgic, neuralgic, or arthritic pain, especially in psychoneurotic patients with insomnia. The pain may appear in paroxysms, is most intense in the early morning hours, and is most frequently located in the region of the neck, shoulder girdle, and upper limbs. Symptoms may last for days after the drug is discontinued.
Allergic
Acquired hypersensitivity to barbiturates consists chiefly in allergic reactions that occur especially in persons who tend to have asthma, urticaria, angioedema, and similar conditions. Hypersensitivity reactions in this category include localized swelling, particularly of the eyelids, cheeks, or lips, and erythematous dermatitis. Rarely, exfoliative dermatitis (e.g., Stevens-Johnson syndrome and toxic epidermal necrolysis) may be caused by phenobarbital and can prove fatal. The skin eruption may be associated with fever, delirium, and marked degenerative changes in the liver and other parenchymatous organs. In a few cases, megaloblastic anemia has been associated with the chronic use of phenobarbital.
Other
Nausea and vomiting; headache.
To report SUSPECTED ADVERSE REACTIONS, contact Hikma Pharmaceuticals USA Inc. at 1-800-962-8364, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
DRUG ABUSE AND DEPENDENCE
Controlled Substance: Phenobarbital is a Schedule IV drug.
Dependence
Prolonged, uninterrupted use of barbiturates (particularly the short-acting drugs), even in therapeutic doses, may result in psychic and physical dependence. Withdrawal symptoms due to physical dependence following chronic use of large doses of barbiturates may include delirium, convulsions, and death.
-
OVERDOSAGE
The signs and symptoms of barbiturate poisoning are referable especially to the central nervous system and the cardiovascular system. Moderate intoxication resembles alcoholic inebriation. In severe intoxication, the patient is comatose, the level of reflex activity conforming in a general way to the intensity of the central depression. The deep reflexes may persist for some time despite coexistent coma. The Babinski sign is often positive. The EEG may be of the “burst-suppression” type, with brief periods of electrical silence. The pupils may be constricted and react to light, but late in the course of barbiturate poisoning they may show hypoxic paralytic dilatation. Respiration is affected early. Breathing may be either slow or rapid and shallow; Cheyne-Stokes rhythm may be present. Respiratory minute volume is diminished, and hypoxia and respiratory acidosis may develop. The blood pressure falls, owing partly to depression of medullary vasomotor centers; partly to a direct action of the drug on the myocardium, sympathetic ganglia, and vascular smooth muscle; partly to hypoxia.
The patient thus develops a typical shock syndrome, with a weak and rapid pulse, cold and clammy skin, and a rise in the hematocrit. Respiratory complications (atelectasis, pulmonary edema, and bronchopneumonia) and renal failure are much dreaded and not infrequent concomitant of severe barbiturate poisoning. There is usually hypothermia, sometimes with temperatures as low as 32°C.
Treatment
General management should consist of symptomatic and supportive therapy, including gastric lavage, administration of intravenous fluids, and maintenance of blood pressure, body temperature and adequate respiratory exchange. Dialysis will increase the rate of removal of barbiturates from the body fluids. Antibiotics may be required to control pulmonary complications.
- DOSAGE AND ADMINISTRATION
-
HOW SUPPLIED
Phenobarbital Tablets, USP 15 mg
White, Round Tablet; Debossed “WW 445” on one side and plain on the other side.
NDC: 0143-1445-01: Bottle of 100 tablets
NDC: 0143-1445-05: Bottle of 500 tabletsNDC: 0143-1495-01: Bottle of 100 tablets
NDC: 0143-1495-05: Bottle of 500 tabletsPhenobarbital Tablets, USP 30 mg
White, Round, Scored Tablet; Debossed “WW 450” on one side and Scored on the other side.
NDC: 0143-1450-01: Bottle of 100 tablets
NDC: 0143-1450-05: Bottle of 500 tabletsNDC: 0143-1500-01: Bottle of 100 tablets
NDC: 0143-1500-05: Bottle of 500 tabletsPhenobarbital Tablets, USP 60 mg
White, Round Tablet; Debossed “WW 455” on one side and plain on the other side.
NDC: 0143-1455-01: Bottle of 100 tablets
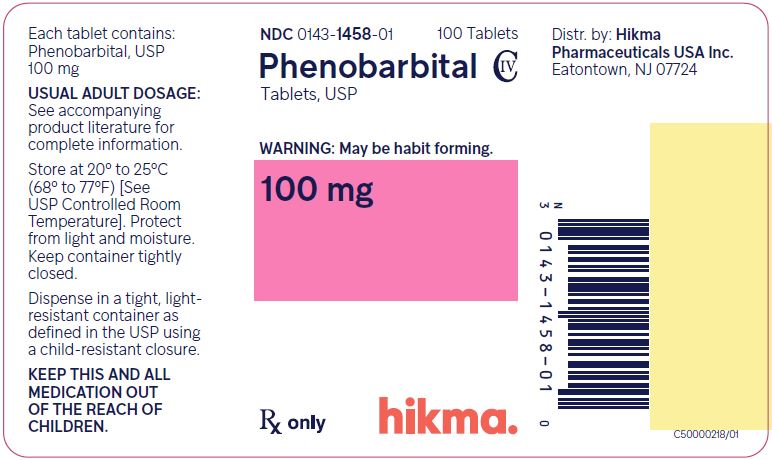
NDC: 0143-1455-05: Bottle of 500 tabletsPhenobarbital Tablets, USP 100 mg
White, Round, Scored Tablet; Debossed “WW 458” on one side and Scored on the other side.
NDC: 0143-1458-01: Bottle of 100 tablets
NDC: 0143-1458-05: Bottle of 500 tabletsStore at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Protect from light and moisture.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
Distr. by:
Hikma Pharmaceuticals USA Inc.
Eatontown, NJ 07724
C50000803/01 Revised July 2019
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PHENOBARBITAL
phenobarbital tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0143-1495 Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENOBARBITAL (UNII: YQE403BP4D) (PHENOBARBITAL - UNII:YQE403BP4D) PHENOBARBITAL 15 mg Inactive Ingredients Ingredient Name Strength CALCIUM STEARATE (UNII: 776XM7047L) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) Product Characteristics Color WHITE Score no score Shape ROUND Size 6mm Flavor Imprint Code WW;445 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0143-1495-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/05/2020 2 NDC: 0143-1495-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 03/05/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 03/05/2020 PHENOBARBITAL
phenobarbital tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0143-1500 Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENOBARBITAL (UNII: YQE403BP4D) (PHENOBARBITAL - UNII:YQE403BP4D) PHENOBARBITAL 30 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) DOCUSATE SODIUM (UNII: F05Q2T2JA0) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score 2 pieces Shape ROUND Size 6mm Flavor Imprint Code WW;450 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0143-1500-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/05/2020 2 NDC: 0143-1500-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 03/05/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 03/05/2020 PHENOBARBITAL
phenobarbital tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0143-1455 Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENOBARBITAL (UNII: YQE403BP4D) (PHENOBARBITAL - UNII:YQE403BP4D) PHENOBARBITAL 60 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) DOCUSATE SODIUM (UNII: F05Q2T2JA0) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score no score Shape ROUND Size 8mm Flavor Imprint Code WW;455 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0143-1455-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 01/11/1999 2 NDC: 0143-1455-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 01/11/1999 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 01/11/1999 PHENOBARBITAL
phenobarbital tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0143-1458 Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENOBARBITAL (UNII: YQE403BP4D) (PHENOBARBITAL - UNII:YQE403BP4D) PHENOBARBITAL 100 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) DOCUSATE SODIUM (UNII: F05Q2T2JA0) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score 2 pieces Shape ROUND Size 10mm Flavor Imprint Code WW;458 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0143-1458-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 01/07/2000 2 NDC: 0143-1458-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 01/07/2000 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 01/07/2000 PHENOBARBITAL
phenobarbital tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0143-1445 Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENOBARBITAL (UNII: YQE403BP4D) (PHENOBARBITAL - UNII:YQE403BP4D) PHENOBARBITAL 15 mg Inactive Ingredients Ingredient Name Strength CALCIUM STEARATE (UNII: 776XM7047L) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) Product Characteristics Color WHITE Score no score Shape ROUND Size 6mm Flavor Imprint Code West;ward;445 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0143-1445-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/05/2020 06/30/2021 2 NDC: 0143-1445-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 03/05/2020 06/30/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 03/05/2020 06/30/2021 PHENOBARBITAL
phenobarbital tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0143-1450 Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENOBARBITAL (UNII: YQE403BP4D) (PHENOBARBITAL - UNII:YQE403BP4D) PHENOBARBITAL 30 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) DOCUSATE SODIUM (UNII: F05Q2T2JA0) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score 2 pieces Shape ROUND Size 6mm Flavor Imprint Code West;ward;450 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0143-1450-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/05/2020 05/31/2021 2 NDC: 0143-1450-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 03/05/2020 05/31/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 03/05/2020 05/31/2021 Labeler - Hikma Pharmaceuticals USA Inc. (001230762)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.