GLYCATE- glycopyrrolate tablet
GLYCATE by
Drug Labeling and Warnings
GLYCATE by is a Prescription medication manufactured, distributed, or labeled by Carwin Pharmaceutical Associates, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
DESCRIPTION
Glycopyrrolate Tablets, USP 1.5 mg contains the synthetic anticholinergic, glycopyrrolate. Glycopyrrolate is a quaternary ammonium compound with the following chemical name: 3-[(cyclopentylhydroxyacetyl) oxy]-1,1-dimethylpyrrolidinium bromide.
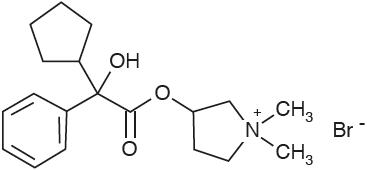
C19H28BrNO3 MW: 398.33
Each tablet contains:
Glycopyrrolate, USP 1.5 mg Inactive Ingredients: Dibasic Calcium Phosphate, Anhydrous Lactose, Magnesium Stearate, Povidone, Sodium Starch Glycolate
-
CLINICAL PHARMACOLOGY
Glycopyrrolate, like other anticholinergic (antimuscarinic) agents, inhibits the action of acetylcholine on structures innervated by postganglionic cholinergic nerves and on smooth muscles that respond to acetylcholine by lack cholinergic innervation. These peripheral cholinergic receptors are present in the autonomic effector cells of smooth muscle, cardiac muscle, the sino-atrial node, the atrioventricular node, exocrine glands, and, to a limited degree, in the autonomic ganglia. Thus, it diminishes the volume and free acidity of gastric secretions and controls excessive pharyngeal, tracheal, and bronchial secretions.
Glycopyrrolate antagonizes muscarinic symptoms (e.g., bronchorrhea, bronchospasm, bradycardia, and intestinal hypermotility) induced by cholinergic drugs such as the anticholinesterases.
The highly polar quaternary ammonium group of glycopyrrolate limits its passage across lipid membranes, such as the blood-brain barrier, in contrast to atropine sulfate and scopolamine hydrobromide, which are non-polar tertiary amines which penetrate lipid barriers easily.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Glaucoma; obstructive uropathy (for example, bladder neck obstruction due to prostatic hypertrophy); obstructive disease of the gastrointestinal tract (as in achalasia, pyloroduodenal stenosis, etc.); paralytic ileus; intestinal atony of the elderly or debilitated patient; unstable cardiovascular status in acute hemorrhage; severe ulcerative colitis; toxic megacolon complicating ulcerative colitis; myasthenia gravis. Glycopyrrolate tablets are contraindicated in those patients with a hypersensitivity to glycopyrrolate.
-
WARNINGS
In the presence of a high environmental temperature, heat prostration (fever and heat stroke due to decreased sweating) can occur with the use of Glycopyrrolate Tablets, USP.
Diarrhea may be an early symptom of incomplete intestinal obstruction, especially in patients with ileostomy or colostomy. In this instance treatment with this drug would be inappropriate and possibly harmful.
Glycopyrrolate Tablets, USP may produce drowsiness or blurred vision. In this event, the patient should be warned not to engage in activities requiring mental alertness such as operating a motor vehicle or other machinery, or performing hazardous work while taking this drug.
Theoretically, with overdosage, a curare-like action may occur, i.e., neuromuscular blockade leading to muscular weakness and possible paralysis.
-
PRECAUTIONS
Use Glycopyrrolate Tablets, USP with caution in the elderly and in all patients with:
- Autonomic neuropathy.
- Hepatic or renal disease.
- Ulcerative colitis - large doses may suppress intestinal motility to the point of producing a paralytic ileus and for this reason may precipitate or aggravate the "toxic megacolon," a serious complication of the disease.
- Hyperthyroidism, coronary heart disease, congestive heart failure, cardiac tachyarrhythmias, tachycardia, hypertension and prostatic hypertrophy.
- Hiatal hernia associated with reflux esophagitis, since anticholinergic drugs may aggravate this condition.
Pregnancy
The safety of this drug during pregnancy has not been established. The use of any drug during pregnancy requires that the potential benefits of the drug be weighed against possible hazards to mother and child. Reproduction studies in rats revealed no teratogenic effects from glycopyrrolate; however, the potent anticholinergic action of this agent resulted in diminished rates of conception and of survival at weaning, in a dose-related manner. Other studies in dogs suggest that this may be due to diminished seminal secretion which is evident at high doses of glycopyrrolate. Information on possible adverse effects in the pregnant female is limited to uncontrolled data derived from marketing experience. Such experience has revealed no reports of teratogenic or other fetus-damaging potential. No controlled studies to establish the safety of the drug in pregnancy have been performed.
-
ADVERSE REACTIONS
Anticholinergics produce certain effects, most of which are extensions of their fundamental pharmacological actions. Adverse reactions to anticholinergics in general may include xerostomia; decreased sweating; urinary hesitancy and retention; blurred vision; tachycardia; palpitations; dilation of the pupil; cycloplegia; increased ocular tension; loss of taste; headaches; nervousness; mental confusion; drowsiness; weakness; dizziness; insomnia; nausea; vomiting; constipation; bloated feeling; impotence; suppression of lactation; severe allergic reaction or drug idiosyncrasies including anaphylaxis, urticaria and other dermal manifestations.
Glycopyrrolate Tablets, USP is chemically a quaternary ammonium compound; hence, its passage across lipid membranes, such as the blood-brain barrier, is limited in contrast to atropine sulfate and scopolamine hydrobromide. For this reason the occurrence of CNS related side effects is lower, in comparison to their incidence following administration of anticholinergics which are chemically tertiary amines that can cross this barrier readily.
Call your doctor for medical advice about side effects. You may voluntarily report side effects to FDA AT 1-800-FDA-1088. Questions or comments? Call Carwin Pharmaceutical Associates, LLC at 1-844-700-5011.
-
OVERDOSAGE
The symptoms of overdosage of glycopyrrolate are peripheral in nature rather than central.
- To guard against further absorption of the drug - use gastric lavage, cathartics, and/or enemas.
- To combat peripheral anticholinergic effects (residual mydriasis, dry mouth, etc.) - utilize a quaternary ammonium anticholinesterase, such as neostigmine methylsulfate.
- To combat hypotension - use pressor amines (norepinephrine, metaraminol) i.v.; and supportive care.
- To combat respiratory depression - administer oxygen; utilize a respiratory stimulant such as Dopram® i.v.; artificial respiration.
-
DOSAGE AND ADMINISTRATION
The dosage of Glycopyrrolate Tablets, USP should be adjusted to the needs of the individual patient to assure symptomatic controlwith a minimum of adverse reactions. The presently recommended maximum daily dosage of glycopyrrolate is 8 mg.
Glycopyrrolate Tablets, USP 1 mg
The recommended initial dosage of Glycopyrrolate 1 mg tablets for adults is one tablet three times daily (in the morning, early afternoon, and at bedtime). Some patients may require two tablets at bedtime to assure overnight control of symptoms.
For maintenance, a dosage of one tablet twice a day is frequently adequate.
-
HOW SUPPLIED
Glycate® (Glycopyrrolate Tablets, USP) 1.5 mg are compressed white tablets debossed GP on one side and 1.5 on the other and are supplied in bottles of 100 (15370-160-10) and a physician sample (15370-160-03).
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 1.5 mg Tablet Bottle Label
NDC: 15370-160-10
Glycate®
(Glycopyrrolate
Tablets, USP 1.5 mg)WHITE DYE-FREE
100 Tablets
Rx Onlycarwin
PHARMACEUTICAL ASSOCIATES
-
INGREDIENTS AND APPEARANCE
GLYCATE
glycopyrrolate tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 15370-160 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCOPYRROLATE (UNII: V92SO9WP2I) (GLYCOPYRRONIUM - UNII:A14FB57V1D) GLYCOPYRROLATE 1.5 mg Inactive Ingredients Ingredient Name Strength CALCIUM PYROPHOSPHATE (UNII: X69NU20D19) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) MAGNESIUM STEARATE (UNII: 70097M6I30) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE (WHITE TO OFF-WHITE) Score no score Shape ROUND (DEBOSSED ON BOTH SIDES) Size 9mm Flavor Imprint Code GP;15 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 15370-160-10 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/10/2018 2 NDC: 15370-160-03 3 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/10/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091522 06/10/2018 Labeler - Carwin Pharmaceutical Associates, LLC (079217215)
Trademark Results [GLYCATE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 GLYCATE 85773181 4506691 Live/Registered |
Argent Development Group, LLC 2012-11-06 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.