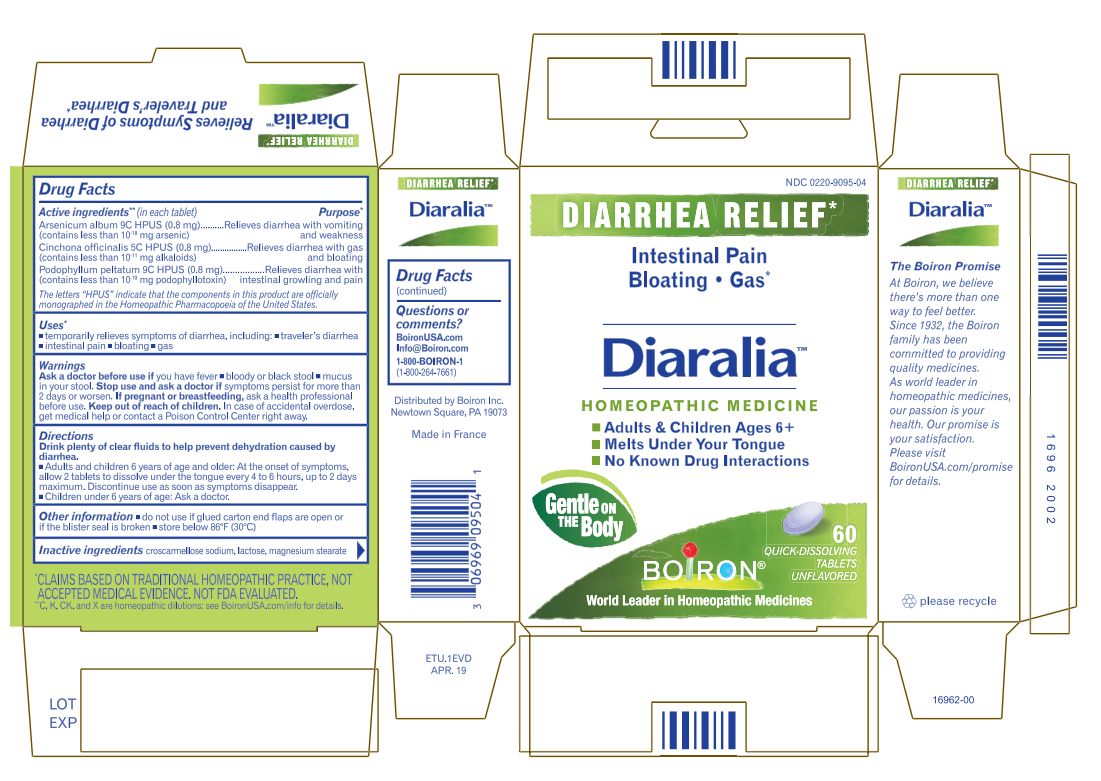
DIARALIA- arsenic trioxide,cinchona officinalis bark, podophyllum tablet
DIARALIA by
Drug Labeling and Warnings
DIARALIA by is a Homeopathic medication manufactured, distributed, or labeled by BOIRON, BOIRON, INC.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
-
PURPOSE
ARSENICUM ALBUM 9C (0.8MG) RELIEVES DIARRHEA WITH VOMITING AND WEAKNESS*
(CONTAINS LESS THAN 10 -18 MG ARSENIC)CINCHONA OFFICINALIS 5C (0.8MG) RELIEVES DIARRHEA WITH GAS AND BLOATING*
(CONTAINS LESS THAN 10 -11 MG ALKALOIDS)PODOPHYLLUM PELTATUM 9C (0.8MG) RELIEVES DIARRHEA WITH INTESTINAL RUMBLING AND PAIN*
(CONTAINS LESS THAN 10 -19 MG PODOPHYLLOTOXIN)
- WARNINGS
- STOP USE
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
DRINK PLENTY OF CLEAR FLUIDS TO HELP PREVENT DEHYDRATION CAUSED BY DIARRHEA.
ADULTS AND CHILDREN 6 YEARS OF AGE AND OLDER: AT THE ONSET OF SYMPTOMS, ALLOW 2 TABLETS TO DISSOLVE UNDER THE TONGUE EVERY 4 TO 6 HOURS, UP TO 2 DAYS MAXIMIUM. DISCONTINUE USE AS SOON AS SYMPTOMS DISAPPEAR.
CHILDERN UNDER 6 YEARS OF AGE: ASK A DOCTOR.
-
SPL UNCLASSIFIED SECTION
DO NOT USE IF GLUED CARTON END FLAPS ARE OPEN OR IF THE BLISTER SEAL IS BROKEN.
CONTAINS 60 MELTAWAY TABLETS
STORE BELOW 86° F (30° C)
THE LETTERS "HPUS" INDICATE THAT THE COMPONENTS IN THIS PRODUCT ARE OFFICIALLY MONOGRAPHED IN THE HOMEOPATHIC PHARMACOPOEIA OF THE UNITED STATES.
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
*C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details. - INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DIARALIA
arsenic trioxide,cinchona officinalis bark, podophyllum tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0220-9095 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PODOPHYLLUM (UNII: 2S713A4VP3) (PODOPHYLLUM - UNII:2S713A4VP3) PODOPHYLLUM 9 [hp_C] CINCHONA OFFICINALIS BARK (UNII: S003A158SB) (CINCHONA OFFICINALIS BARK - UNII:S003A158SB) CINCHONA OFFICINALIS BARK 5 [hp_C] ARSENIC TRIOXIDE (UNII: S7V92P67HO) (ARSENIC CATION (3+) - UNII:C96613F5AV) ARSENIC TRIOXIDE 9 [hp_C] Inactive Ingredients Ingredient Name Strength MAGNESIUM STEARATE (UNII: 70097M6I30) LACTOSE, UNSPECIFIED FORM (UNII: J2B2A4N98G) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) Product Characteristics Color white Score no score Shape ROUND Size 9mm Flavor Imprint Code Boiron Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0220-9095-04 60 in 1 CARTON 08/01/2019 1 60 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 08/01/2019 Labeler - BOIRON (282560473) Registrant - BOIRON, INC. (014892269) Establishment Name Address ID/FEI Business Operations BOIRON 282560473 manufacture(0220-9095)
Trademark Results [DIARALIA]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 DIARALIA 79243571 5847691 Live/Registered |
BOIRON 2018-09-12 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.