DG HEALTH ESOMEPRAZOLE MAGNESIUM- esomeprazole capsule, delayed release
dg health esomeprazole magnesium by
Drug Labeling and Warnings
dg health esomeprazole magnesium by is a Otc medication manufactured, distributed, or labeled by Dolgencorp, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
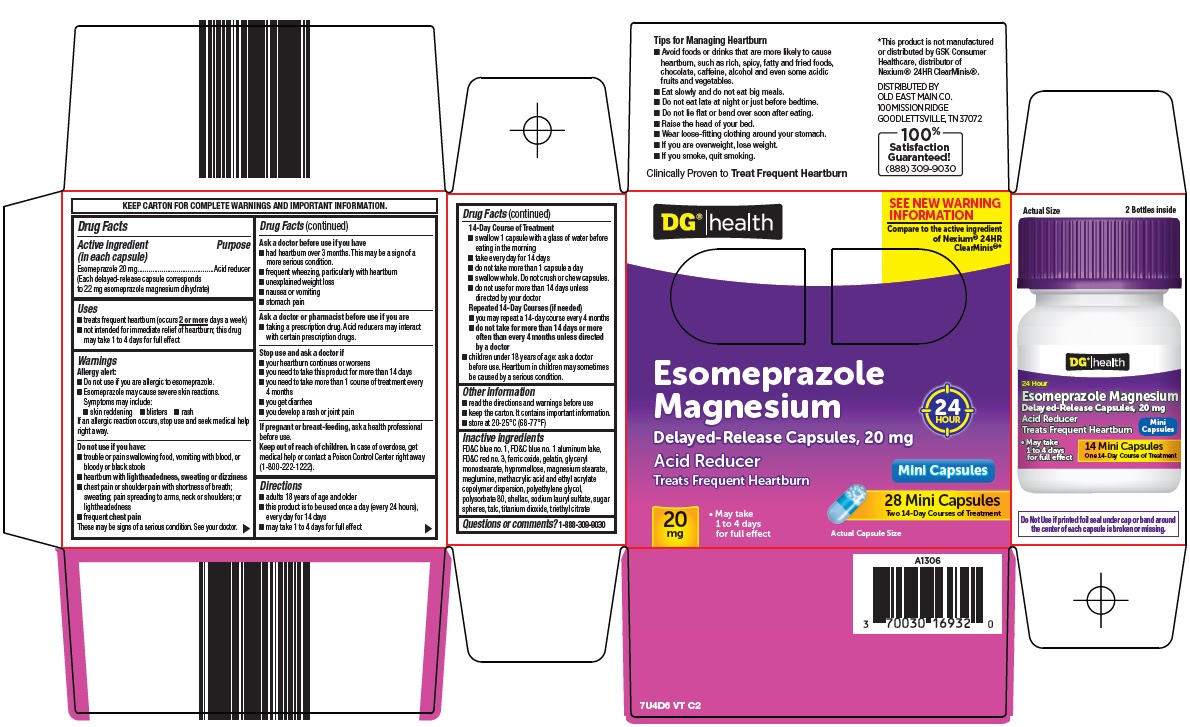
- Active ingredient (in each capsule)
- Purpose
- Uses
-
Warnings
Allergy alert:
- Do not use if you are allergic to esomeprazole.
-
Esomeprazole may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If an allergic reaction occurs, stop use and seek medical help right away.
Do not use if you have:
- trouble or pain swallowing food, vomiting with blood, or bloody or black stools
- heartburn with lightheadedness, sweating or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
These may be signs of a serious condition. See your doctor.
Ask a doctor before use if you have
- had heartburn over 3 months. This may be a sign of a more serious condition.
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Ask a doctor or pharmacist before use if you are
- taking a prescription drug. Acid reducers may interact with certain prescription drugs.
-
Directions
- adults 18 years of age and older
- this product is to be used once a day (every 24 hours), every day for 14 days
- may take 1 to 4 days for full effect
- 14-Day Course of Treatment
- swallow 1 capsule with a glass of water before eating in the morning
- take every day for 14 days
- do not take more than 1 capsule a day
- swallow whole. Do not crush or chew capsules.
- do not use for more than 14 days unless directed by your doctor
- Repeated 14-Day Courses (if needed)
- you may repeat a 14-day course every 4 months
- do not take for more than 14 days or more often than every 4 months unless directed by a doctor
- children under 18 years of age: ask a doctor before use. Heartburn in children may sometimes be caused by a serious condition.
- Other information
-
Inactive ingredients
FD&C blue no. 1, FD&C blue no. 1 aluminum lake, FD&C red no. 3, ferric oxide, gelatin, glyceryl monostearate, hypromellose, magnesium stearate, meglumine, methacrylic acid and ethyl acrylate copolymer dispersion, polyethylene glycol, polysorbate 80, shellac, sodium lauryl sulfate, sugar spheres, talc, titanium dioxide, triethyl citrate
- Questions or comments?
-
Package/Label Principal Display Panel
DG®|health
SEE NEW WARNING INFORMATION
Compare to the active ingredient of Nexium® 24HR ClearMinis®
Esomeprazole Magnesium
Delayed-Release Capsules, 20 mg
24 HOUR
Acid Reducer
Mini Capsules
Treats Frequent Heartburn
28 Mini Capsules
Two 14-Day Courses of Treatment
20 mg
May take 1 to 4 for full effect
Actual Capsule Size
-
INGREDIENTS AND APPEARANCE
DG HEALTH ESOMEPRAZOLE MAGNESIUM
esomeprazole capsule, delayed releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 55910-250 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ESOMEPRAZOLE (UNII: N3PA6559FT) (ESOMEPRAZOLE - UNII:N3PA6559FT) ESOMEPRAZOLE 20 mg Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C BLUE NO. 1 ALUMINUM LAKE (UNII: J9EQA3S2JM) FD&C RED NO. 3 (UNII: PN2ZH5LOQY) FERRIC OXIDE RED (UNII: 1K09F3G675) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MEGLUMINE (UNII: 6HG8UB2MUY) METHACRYLIC ACID AND ETHYL ACRYLATE COPOLYMER (UNII: NX76LV5T8J) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) SHELLAC (UNII: 46N107B71O) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color BLUE Score no score Shape CAPSULE Size 11mm Flavor Imprint Code 7U4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 55910-250-01 1 in 1 CARTON 07/14/2020 1 14 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC: 55910-250-02 2 in 1 CARTON 07/14/2020 2 14 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC: 55910-250-03 3 in 1 CARTON 03/18/2021 3 14 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA207193 07/14/2020 Labeler - Dolgencorp, LLC (068331990)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.