4390 FIRST AID KIT kit 4392 FIRST AID KIT kit
4392 First Aid Kit by
Drug Labeling and Warnings
4392 First Aid Kit by is a Otc medication manufactured, distributed, or labeled by Honeywell Safety Products USA, INC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Eyesaline Active ingredient
- Eyesaline Purpose
- Eyesaline Uses
-
Eyesaline
Warnings
For external use only-
Obtain immediate medical treatment for all open wounds in or near eyes.
To avoid contamination, do not touch tip of container to any surface.
Do not reuse. Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away.
- Eyesaline Directions
- Eyesaline Inactive ingredients
- Eyesaline Questions
- Alcohol Wipe Active ingredient
- Alcohol Wipe Purpose
- Alcohol Wipe Uses
- Alcohol Wipe Warnings
- Alcohol Wipe Directions
- Alcohol Wipe Other information
- Alcohol Wipe Inactive ingredient
- Alcohol Wipe Questions
- Aypanal Active ingredient
- Aypanal Purpose
- Aypanaly Uses
-
Aypanal
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 4,000 mg in 24 hours, which is the maximum daily amount
- child takes more than 5 doses in 24 hours, which is the maximum daily amount
- taken with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product:
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
- If a skin rash occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription).
- If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
Stop using and ask a doctor if- pain gets worse or lasts more than 10 days in adults
- pain gets worse or lasts more than 5 days in children under 12 years
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
-
Aypanal
Directions
do not take more than directed (see overdose warning)
adults and children 12 years of age or older
- take two tablets every 4-6 hours while symptoms last
- do not take more than 12 tablets in 24 hours
children 6 to under 12 years of age
- take 1 tablet every 4-6 hours while symptoms last
- do not take more than 5 tablets in 24 hours
children under 6 years consult a doctor
- Aypanal Other information
- Aypanal Inactive ingredients
- Aypanal Questions
- Neomycin Active ingredient
- Neomycin Purpose
- Neomycin Uses
-
Neomycin
Warnings
For external use only
Stop use and ask a doctor if- a rash or other allergic reaction develops
- you need to use longer than 1 week
Stop use and ask a doctor if
- the condition persists or gets worse
- a rash or other allergic reaction develops
- you need to use longer than 1 week
- Neomycin Direction
- Neomycin Other information
- Neomycin Inactive ingredient
- Neomycin Questions?
- Antiseptic Spray Active ingredient
- Antiseptic Spray Purpose
- Antiseptic Spray Uses
- Antiseptic Spray Warnings
- Antiseptic Spray Directions
- Antiseptic Spray Other information
- Antiseptic Spray Inactive ingredients
- Antiseptic Spray Questions
- Burn Spray Active ingredient
- Burn Spray Purpose
- Burn Spray Uses
-
Burn Spray
Warnings
For external use only
Flammable
- keep away from fire or flame
- contents under pressure
- do not puncture or incinerate container
- do not expose to temperatures above 120 0 F
Do not use
- in or near the eyes or other mucous membranes
- in case of serious burns
- in case of deep or puncture wounds
- for prolonged period of time
- on large portion of the body
- Burn Spray Directions
- Burn Spray Other information
- Burn Spray Inactive ingredients
- Hand Sanitizer Active ingredient
- Hand Santizer Purpose
- Hand Sanitizer Uses
- Hand Sanitizer Warnings
- Hand Sanitizer Directions
- Hand Sanitizer Other information
- Hand Sanitizer Inactive ingredients
- Hand Sanitizer Questions or Comments?
- Ammonia Active ingredient
- Ammonia Purpose
- Ammonia Uses
- Ammonia Warnings
- Ammonia Directions
- Ammonia Other information
- Ammonia Inactive ingredients
- Ammonia Questions or Comments?
- Cedaprin Active ingredient
- Cedaprin Purposse
- Cedaprin Uses
-
Cedaprin
Warnings
Allergy alert: Ibuprofen may cause a severe allergic reaction, especially in people allergic to aspirin. Symptoms may include:
- hives
- facial swelling
- asthma (wheezing)
- shock
- skin reddening
- rash
- blisters
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs [aspirin, ibuprofen, naproxen, or others]
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Heart attack and stroke warning:
- NSAID's, except aspirin, increase the risk of heart attack, heart failure, and stroke. These can be fatal. The risk is higher if you use more than directed or for longer than directed.
Do not use
- if you have ever had an allergic reaction to ibuprofen or any other pain reliever/fever reducer
- right before or after heart surgery
Ask a doctor before use if
- you have problems or serious side effectsfrom taking pain relievers or fever reducers
- stomach bleeding warning applies to you
- you have a history of stomach problems such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, kidney disease, asthma or had a stroke
- you are taking a diuretic
Ask a doctor or a pharmacist before use if you are
- taking aspirin for heart attack or stroke, because ibuprofen may decrease the benefit of aspirin
- under a doctors care for any serious condition
- taking any other drug
Stop use and ask a doctor if
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- you have symptoms of heart problems or stroke:
- chest pain
- trouble breathing
- weakness in oe part or side of body
- slurred speech
- leg swelling
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present in the painful area
- any new symptoms appea
-
Cedaprin
Directions
- do not take more than directed
- the smallest effective dose should be used
- adult and children 12 years of age and over:
- take 1 tablet every 4 to 6 hours while symptoms persist
- if pain or fever does not respond to 1 tablet, 2 tablets may be used
- do not exceed 6 tablets in 24 hours, unless directed by a doctor
- children under 12 years: ask a doctor
- Cedaprin Other information
- cedaprin Inactive ingredients
- Cedaprin Questions or Comments?
- Miralac Active ingredient
- Miralac Purpose
- Miralac Uses
-
Miralac
Warnings
Ask a doctor before use if you have
- kidney stones
- calcium-restricted diet
Ask a doctor before use if you are
presently taking a prescription drug. Antacids may interfere with certain prescription drugs
- Miralac Directions
- Miralac Other information
- Miralac Inactive ingredients
- Miralac Questions or Comments
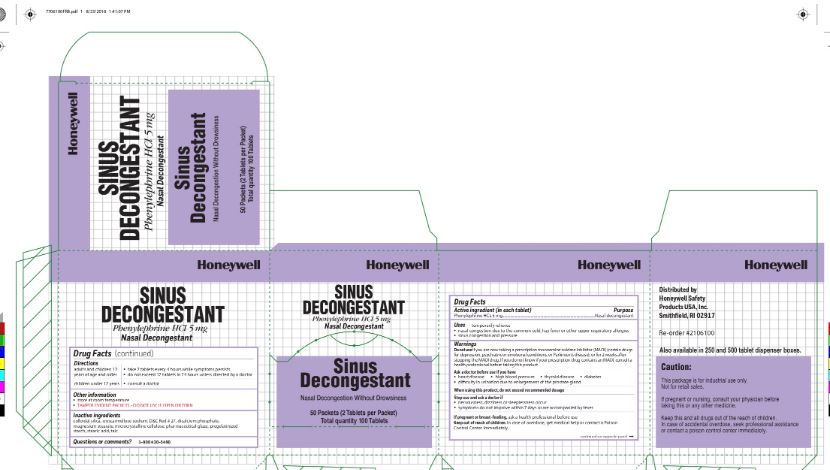
- Sinus decongestant Active ingredient
- Sinus Decongestant Purpose
- Sinus Decongestant Uses
-
Sinus Decongestant
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, consult a health professional before taking this product.
Ask a doctor before use if you have
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- difficulty in urination due to enlargement of the prostate gland
- Sinus Decongestant Directions
- Sinus Decongestant Other information
- Sinus Decongestant Inactive ingredients
- Sinus Decongestant Questions or Comments
-
4392
SF00004422 Kit Contents
1 1X3 PLASTIC 100/BOX
1 FINGERTIP "T" WOVEN 40/BOX
1 SWIFT KNUCKLE 40/BX
1 1 X 3 WOVEN 100/BOX
3 NEOMYCIN ANTIBIOTIC 10 PER
1 AMMONIA INHALANTS 10 PER
2 EYE DRESS PKT W/4 ADH STRIPS
1 TOURNIQUET, 1 PER
1 WIRE SPLINT 1 PER
1 ADH BAND, EXTRA LARGE, 6 PER
1 ALCOHOL PREP PADS 10P
1 O/H PUMP ANTISEPTIC 2 OZ ID F
1 O/H PUMP BURN RELIEF 2 OZ ID G
1 FIRST AID GUIDE ASHI
2 TAPE ADHESIVE 1"X 5 YD PLSTC
10 HAND SANITIZER 0.9G WJ BULK
4 GAUZE CLEAN-WRAP BDGE N/S 2"
4 GAUZE CLEAN-WRAP BDGE N/S 4"
4 BLOODSTOPPER
1 NON-ADHERENT PADS 2"X3" 10'S
1 GZE PADS STERILE 2"X 2" 10'S
1 GZE PADS STERILE 3"X 3" 25'S
1 ELASTIC BANDAGE 3" X 4.5YD
1 CPR FILTERSHIELD 77-100
1 COTTON TIPS 100 PER VIAL
1 AYPANAL NON-ASP IND 2/ENV 100
1 CEDAPRIN (IBUPROFEN) 2ENV 100
1 MIRALAC TABS IND PK 2/ENV 100
1 SINUS DECONGESTANT 2/ENV 100
1 4OZ BFS EYEWASH TRILINGUAL BOTTLE
1 SCISSOR BDGE 4" RED PLS HDL
1 KIT TWEEZER 3 1/2" SLANTED
1 400 EMPTY KIT BLANK
1 POCKET INSERT RED #400 KIT 5R
1 TONGUE BLADES SR WRAPPED 6'S
1 LBL STOCK 6-3/8"X4"
1 LBL STOCK 4"X2-7/8"
2 x2 PR LRG NITRILE GLVES ZIP BAG
2 WATER-JEL BURN DRESSING 4 X 4
8 CORNER STYROFOAM 3X3X3
2TRI BNDG NON WOVEN 40"X40"X56"
2 COLD PACK UNIT 4"X6" BULK
1 RED BIO BAGS 2/BX
- Eyesaline Principal Display Panel
- Alcohol Wipe Principal Display Panel
- Aypanal Principal Display Panel
- Neomycin Principal Display Panel
- Antiseptic Spray Principal Display Panel
- Burn Spray Principal Display Panel
- Hand Sanitizer Principal Display Panel
- Ammonia Principal Display Panel
- Cedaprin Principal Display Panel
- Miralac Principal Display Panel
- Sinus Decongestant Principal Display Panel
- 4390 Kit Label FAK4SHLF-CLSB
-
4390 Kit Contnets
FAK4SHLF-CLSB
1 1X3 PLASTIC 100/BOX
1 FINGERTIP "T" WOVEN 40/BOX
1 SWIFT KNUCKLE 40/BX
1 1 X 3 WOVEN 100/BOX
3 NEOMYCIN ANTIBIOTIC 10 PER
1 AMMONIA INHALANTS 10 PER
2 EYE DRESS PKT W/4 ADH STRIPS
1 TOURNIQUET, 1 PER
1 WIRE SPLINT 1 PER
1 ADH BAND, EXTRA LARGE, 6 PER
1 ALCOHOL PREP PADS 10P
1 O/H PUMP ANTISEPTIC 2 OZ ID F
1 O/H PUMP BURN RELIEF 2 OZ ID G
1 FIRST AID GUIDE ASHI
2 TAPE ADHESIVE 1"X 5 YD PLSTC
10 HAND SANITIZER 0.9G WJ BULK
4 GAUZE CLEAN-WRAP BDGE N/S 2"
4 GAUZE CLEAN-WRAP BDGE N/S 4"
4 BLOODSTOPPER
1 NON-ADHERENT PADS 2"X3" 10'S
1 GZE PADS STERILE 2"X 2" 10'S
1 GZE PADS STERILE 3"X 3" 25'S
1 ELASTIC BANDAGE 3" X 4.5YD
1 CPR FILTERSHIELD 77-100
1 COTTON TIPS 100 PER VIAL
1 AYPANAL NON-ASP IND 2/ENV 100
1 CEDAPRIN (IBUPROFEN) 2ENV 100
1 MIRALAC TABS IND PK 2/ENV 100
1 SINUS DECONGESTANT 2/ENV 100
1 4OZ BFS EYEWASH TRILINGUAL BOTTLE
1 SCISSOR BDGE 4" RED PLS HDL
1 KIT TWEEZER 3 1/2" SLANTED
1 POCKET INSERT RED #400 KIT 5R
1 TONGUE BLADES SR WRAPPED 6'S
1 LBL STOCK 6-3/8"X4"
1 LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
4 PR LRG NITRILE GLVES ZIP BAG
2 WATER-JEL BURN DRESSING 4 X 4
1 LBL CONTENTS ANSI 2015 CL B
1 LBL CAB CVR ANSI 2015 CL B
2 TRI BNDG NON WOVEN 40"X40"X56"
2 COLD PACK UNIT 4"X6" BULK
1 RED BIO BAGS 2/BX
- 4392 Kit Label SF00004422
-
INGREDIENTS AND APPEARANCE
4390 FIRST AID KIT
4390 first aid kit kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0498-4390 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-4390-01 1 in 1 KIT; Type 0: Not a Combination Product 10/18/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 118 mL Part 2 10 POUCH 4 mL Part 3 50 PACKET 100 Part 4 1 BOTTLE, SPRAY 59 mL Part 5 1 BOTTLE, SPRAY 59 mL Part 6 30 PACKET 27 g Part 7 10 PACKAGE 9 mL Part 8 10 AMPULE 3 mL Part 9 50 PACKET 100 Part 10 50 PACKET 100 Part 11 50 PACKET 100 Part 1 of 11 EYESALINE EMERGENCY EYEWASH
purified water liquidProduct Information Item Code (Source) NDC: 0498-0100 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 98.6 mL in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0100-02 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 12/18/2018 Part 2 of 11 ALCOHOL WIPE
isopropyl alcohol swabProduct Information Item Code (Source) NDC: 0498-0143 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0143-04 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 09/18/2018 Part 3 of 11 AYPANAL NON-ASPIRIN
acetaminophen tabletProduct Information Item Code (Source) NDC: 0498-2001 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) POVIDONE (UNII: FZ989GH94E) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) Product Characteristics Color white Score 2 pieces Shape ROUND Size 10mm Flavor Imprint Code circle;U Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 04/10/2012 Part 4 of 11 BURN RELIEF
lidocaine hydrochloride sprayProduct Information Item Code (Source) NDC: 0498-0221 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 24.64 mg in 1 mL Inactive Ingredients Ingredient Name Strength EDETATE DISODIUM (UNII: 7FLD91C86K) GLYCERIN (UNII: PDC6A3C0OX) TROLAMINE (UNII: 9O3K93S3TK) PROPYLPARABEN (UNII: Z8IX2SC1OH) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) OCTOXYNOL-9 (UNII: 7JPC6Y25QS) HYPROMELLOSES (UNII: 3NXW29V3WO) TEA TREE OIL (UNII: VIF565UC2G) METHYLPARABEN (UNII: A2I8C7HI9T) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0221-59 59 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 09/18/2018 Part 5 of 11 ANTISEPTIC
benzalkonium chloride sprayProduct Information Item Code (Source) NDC: 0498-0402 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.13 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) OCTOXYNOL 9 (UNII: 7JPC6Y25QS) GLYCERIN (UNII: PDC6A3C0OX) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) DIPROPYLENE GLYCOL (UNII: E107L85C40) EDETATE DISODIUM (UNII: 7FLD91C86K) TROLAMINE (UNII: 9O3K93S3TK) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0402-59 59 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 09/18/2018 Part 6 of 11 NEOMYCIN
antibiotic ointmentProduct Information Item Code (Source) NDC: 0498-0730 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN SULFATE 3.5 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0730-01 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333B 03/31/2010 Part 7 of 11 INSTANT HAND SANITIZER
alcohol liquidProduct Information Item Code (Source) NDC: 59898-420 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 mL in 100 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) CARBOMER COPOLYMER TYPE A (UNII: 71DD5V995L) WATER (UNII: 059QF0KO0R) TRIISOPROPANOLAMINE (UNII: W9EN9DLM98) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.9 mL in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 04/15/2010 Part 8 of 11 AMMONIA INHALENT
ammonia inhalent inhalantProduct Information Item Code (Source) NDC: 0498-3334 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AMMONIA (UNII: 5138Q19F1X) (AMMONIA - UNII:5138Q19F1X) AMMONIA 0.045 g in 0.3 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-3334-00 0.3 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/18/2018 Part 9 of 11 MIRALAC
calcium carbonate tabletProduct Information Item Code (Source) NDC: 0498-0303 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB, CARBONATE ION - UNII:7UJQ5OPE7D) CALCIUM CARBONATE 420 mg Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) MAGNESIUM STEARATE (UNII: 70097M6I30) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color white (white) Score 2 pieces Shape ROUND Size 11mm Flavor MINT (mint flavor) Imprint Code FR8 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part331 02/22/2012 Part 10 of 11 SINUS DECONGESTANT
phenylephrine tabletProduct Information Item Code (Source) NDC: 0498-3331 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) CALCIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: O7TSZ97GEP) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SHELLAC (UNII: 46N107B71O) STARCH, CORN (UNII: O8232NY3SJ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C RED NO. 27 (UNII: 2LRS185U6K) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) Product Characteristics Color red (red) Score no score Shape OVAL Size 11mm Flavor Imprint Code FR4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 09/18/2018 Part 11 of 11 CEDAPRIN
ibuprofen tabletProduct Information Item Code (Source) NDC: 0498-7502 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IBUPROFEN (UNII: WK2XYI10QM) (IBUPROFEN - UNII:WK2XYI10QM) IBUPROFEN 200 mg Inactive Ingredients Ingredient Name Strength SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) POVIDONE K30 (UNII: U725QWY32X) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) HYPROMELLOSES (UNII: 3NXW29V3WO) TRIACETIN (UNII: XHX3C3X673) STARCH, CORN (UNII: O8232NY3SJ) FERRIC OXIDE RED (UNII: 1K09F3G675) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) Product Characteristics Color red Score no score Shape ROUND Size 10mm Flavor Imprint Code G;2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-7502-01 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA079129 01/02/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/18/2018 4392 FIRST AID KIT
4392 first aid kit kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0498-4392 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-4392-01 1 in 1 KIT; Type 0: Not a Combination Product 10/18/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 50 PACKET 100 Part 2 1 BOTTLE 118 mL Part 3 10 POUCH 4 mL Part 4 50 PACKET 100 Part 5 1 BOTTLE, SPRAY 59 mL Part 6 1 BOTTLE, SPRAY 59 mL Part 7 30 PACKET 27 g Part 8 10 PACKAGE 9 mL Part 9 10 AMPULE 3 mL Part 10 50 PACKET 100 Part 11 50 PACKET 100 Part 1 of 11 CEDAPRIN
ibuprofen tabletProduct Information Item Code (Source) NDC: 0498-7502 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IBUPROFEN (UNII: WK2XYI10QM) (IBUPROFEN - UNII:WK2XYI10QM) IBUPROFEN 200 mg Inactive Ingredients Ingredient Name Strength SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) POVIDONE K30 (UNII: U725QWY32X) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) HYPROMELLOSES (UNII: 3NXW29V3WO) TRIACETIN (UNII: XHX3C3X673) STARCH, CORN (UNII: O8232NY3SJ) FERRIC OXIDE RED (UNII: 1K09F3G675) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) Product Characteristics Color red Score no score Shape ROUND Size 10mm Flavor Imprint Code G;2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-7502-01 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA079129 01/02/2017 Part 2 of 11 EYESALINE EMERGENCY EYEWASH
purified water liquidProduct Information Item Code (Source) NDC: 0498-0100 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 98.6 mL in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0100-02 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 12/18/2018 Part 3 of 11 ALCOHOL WIPE
isopropyl alcohol swabProduct Information Item Code (Source) NDC: 0498-0143 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0143-04 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 09/18/2018 Part 4 of 11 AYPANAL NON-ASPIRIN
acetaminophen tabletProduct Information Item Code (Source) NDC: 0498-2001 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) POVIDONE (UNII: FZ989GH94E) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) Product Characteristics Color white Score 2 pieces Shape ROUND Size 10mm Flavor Imprint Code circle;U Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 04/10/2012 Part 5 of 11 BURN RELIEF
lidocaine hydrochloride sprayProduct Information Item Code (Source) NDC: 0498-0221 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 24.64 mg in 1 mL Inactive Ingredients Ingredient Name Strength EDETATE DISODIUM (UNII: 7FLD91C86K) GLYCERIN (UNII: PDC6A3C0OX) TROLAMINE (UNII: 9O3K93S3TK) PROPYLPARABEN (UNII: Z8IX2SC1OH) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) OCTOXYNOL-9 (UNII: 7JPC6Y25QS) HYPROMELLOSES (UNII: 3NXW29V3WO) TEA TREE OIL (UNII: VIF565UC2G) METHYLPARABEN (UNII: A2I8C7HI9T) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0221-59 59 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 09/18/2018 Part 6 of 11 ANTISEPTIC
benzalkonium chloride sprayProduct Information Item Code (Source) NDC: 0498-0402 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.13 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) OCTOXYNOL 9 (UNII: 7JPC6Y25QS) GLYCERIN (UNII: PDC6A3C0OX) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) DIPROPYLENE GLYCOL (UNII: E107L85C40) EDETATE DISODIUM (UNII: 7FLD91C86K) TROLAMINE (UNII: 9O3K93S3TK) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0402-59 59 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 09/18/2018 Part 7 of 11 NEOMYCIN
antibiotic ointmentProduct Information Item Code (Source) NDC: 0498-0730 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN SULFATE 3.5 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-0730-01 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333B 03/31/2010 Part 8 of 11 INSTANT HAND SANITIZER
alcohol liquidProduct Information Item Code (Source) NDC: 59898-420 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 mL in 100 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) CARBOMER COPOLYMER TYPE A (UNII: 71DD5V995L) WATER (UNII: 059QF0KO0R) TRIISOPROPANOLAMINE (UNII: W9EN9DLM98) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.9 mL in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 04/15/2010 Part 9 of 11 AMMONIA INHALENT
ammonia inhalent inhalantProduct Information Item Code (Source) NDC: 0498-3334 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AMMONIA (UNII: 5138Q19F1X) (AMMONIA - UNII:5138Q19F1X) AMMONIA 0.045 g in 0.3 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0498-3334-00 0.3 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/18/2018 Part 10 of 11 MIRALAC
calcium carbonate tabletProduct Information Item Code (Source) NDC: 0498-0303 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB, CARBONATE ION - UNII:7UJQ5OPE7D) CALCIUM CARBONATE 420 mg Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) MAGNESIUM STEARATE (UNII: 70097M6I30) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color white (white) Score 2 pieces Shape ROUND Size 11mm Flavor MINT (mint flavor) Imprint Code FR8 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part331 02/22/2012 Part 11 of 11 SINUS DECONGESTANT
phenylephrine tabletProduct Information Item Code (Source) NDC: 0498-3331 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) CALCIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: O7TSZ97GEP) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SHELLAC (UNII: 46N107B71O) STARCH, CORN (UNII: O8232NY3SJ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C RED NO. 27 (UNII: 2LRS185U6K) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) Product Characteristics Color red (red) Score no score Shape OVAL Size 11mm Flavor Imprint Code FR4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 09/18/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/18/2018 Labeler - Honeywell Safety Products USA, INC (079287321) Establishment Name Address ID/FEI Business Operations James Alexander 040756421 manufacture(0498-3334) Establishment Name Address ID/FEI Business Operations Honeywell Safety Products USA, INC 079287321 pack(0498-4390, 0498-4392) Establishment Name Address ID/FEI Business Operations Ultra Seal Corporation 085752004 manufacture(0498-2001, 0498-0303, 0498-3331) Establishment Name Address ID/FEI Business Operations Water-Jel Technologies 155522589 manufacture(0498-0730, 0498-0402, 0498-0221, 59898-420) Establishment Name Address ID/FEI Business Operations Honeywell Safety Products USA, Inc. 167518617 manufacture(0498-0100) Establishment Name Address ID/FEI Business Operations Changzhou Maokang Medical 421317073 manufacture(0498-0143) Establishment Name Address ID/FEI Business Operations Granules of India Limited 918609236 manufacture(0498-7502)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.