VYBRIQUE- sildenafil citrate film
Vybrique by
Drug Labeling and Warnings
Vybrique by is a Prescription medication manufactured, distributed, or labeled by IBSA Pharma Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to useVYBRIQUEsafely and effectively. See full prescribing information forVYBRIQUE.
VYBRIQUETM(sildenafil)oral film
Initial U.S. Approval:1998INDICATIONS AND USAGE
VYBRIQUE is a phosphodiesterase-5 (PDE5) inhibitor indicated for the treatment of erectile dysfunction (ED). ( 1)
DOSAGE AND ADMINISTRATION
Dosage
- For most patients, the recommended dosage is 50 mg orally, taken as needed, approximately 1 hour before sexual activity. However, VYBRIQUE may be taken anywhere from 30 minutes to 4 hours before sexual activity. (
2.1)
- Based on effectiveness and toleration, may increase to a maximum of 100 mg or decrease to 25 mg. (
2.1)
- Maximum recommended dosing frequency is once per day. (
2.1)
- Dosage Modifications for Drug Interactions: Refer to the full prescribing information for recommended dosage. (
2.2)
- Recommended Dosage in Specific Populations: Refer to the full prescribing information for recommended dosage. ( 2.3)
Administration
- Administer with or without food.
- Place oral film directly onto the tongue where it will disintegrate and can then be swallowed with saliva without the need for water or other liquids.
- Do not cut or chew VYBRIQUE.
DOSAGE FORMS AND STRENGTHS
Oral film: 25 mg, 50 mg, 75 mg, 100 mg of sildenafil ( 3)
CONTRAINDICATIONS
- Administration of VYBRIQUE to patients using nitric oxide donors, such as organic nitrates or organic nitrites in any form. VYBRIQUE was shown to potentiate the hypotensive effect of nitrates. (
4.1,
7.1,
12.2)
- Known hypersensitivity to sildenafil or any component of oral film. (
4.2)
- Administration with guanylate cyclase (GC) stimulators, such as riociguat. ( 4.3)
WARNINGS AND PRECAUTIONS
- Patients should not use VYBRIQUE if sexual activity is inadvisable due to cardiovascular status. (
5.1)
- Patients should seek emergency treatment if an erection lasts >4 hours.
- Use VYBRIQUE with caution in patients predisposed to priapism. (
5.2)
- Patients should stop VYBRIQUE and seek medical care if a sudden loss of vision occurs in one or both eyes, which could be a sign of non-arteritic anterior ischemic optic neuropathy (NAION). VYBRIQUE should be used with caution, and only when the anticipated benefits outweigh the risks, in patients with a history of NAION. Patients with a “crowded” optic disc may also be at an increased risk of NAION. (
5.3)
- Patients should stop VYBRIQUE and seek prompt medical attention in the event of sudden decrease or loss of hearing. (
5.4)
- Caution is advised when VYBRIQUE is co-administered with alpha-blockers or antihypertensives. Concomitant use may lead to hypotension. ( 5.5)
ADVERSE REACTIONS
Most common adverse reactions (≥ 2%) include headache, flushing, dyspepsia, abnormal vision, nasal congestion, back pain, myalgia, nausea, dizziness and rash. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact IBSA Pharma Inc. at 1-800-587-3513 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatchDRUG INTERACTIONS
- VYBRIQUE can potentiate the hypotensive effects of nitrates, alpha blockers, and antihypertensives. (
4.1,
5.5,
7.1,
7.2,
7.3,
12.2)
- With concomitant use of alpha blockers, initiate VYBRIQUE at 25 mg dose. (
2.2)
- CYP3A4 inhibitors (e.g., ritonavir, ketoconazole, itraconazole, erythromycin) increase VYBRIQUE exposure. (
2.2,
7.4,
12.3)
- Ritonavir: Do not exceed a maximum single dose of 25 mg in a 48- hour period. (
2.2,
5.6)
- Erythromycin or strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, saquinavir): Consider a starting dose of 25 mg. ( 2.2, 7.4)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 12/2025
- For most patients, the recommended dosage is 50 mg orally, taken as needed, approximately 1 hour before sexual activity. However, VYBRIQUE may be taken anywhere from 30 minutes to 4 hours before sexual activity. (
2.1)
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1. INDICATIONSAND USAGE
2. DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 DosageModificationsforDrug Interactions
2.3 RecommendedDosage inSpecificPopulations
2.4 Important Administration Instructions
3. DOSAGE FORMS AND STRENGTHS
4. CONTRAINDICATIONS
4.1 Nitrates
4.2 Hypersensitivity Reactions
4.3 Concomitant Guanylate Cyclase (GC) Stimulators
5. WARNINGS AND PRECAUTIONS
5.1 CardiovascularRisk
5.2 Prolonged Erection and Priapism
5.3 Effects on the Eye
5.4 SuddenHearing Loss
5.5 Hypotension when Co-administered with Alpha-blockers or Antihypertensives
5.6 Adverse Reactions with the Concomitant Use of Ritonavir
5.7 Combination with other PDE5 Inhibitors or Other Erectile Dysfunction Therapies
5.8 Effects on Bleeding
5.9 Counseling Patients About Sexually Transmitted Diseases
6. ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7. DRUG INTERACTIONS
7.1 Nitrates
7.2 Alpha-blockers
7.3 Amlodipine
7.4 Ritonavir and other CYP3A4 inhibitors
7.5 Alcohol
8. USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1. INDICATIONSAND USAGE
-
2.
DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
For most patients, the recommended dosage is 50 mg orally administered on the tongue, taken as needed, approximately 1 hour before sexual activity. However, VYBRIQUE may be taken anywhere from 30 minutes to 4 hours before sexual activity. The maximum recommended dosing frequency is once per day.
Based on effectiveness and tolerability the dosage may be increased to a maximum recommended dose of 100 mg or decreased to 25 mg, not to exceed once per day. For administration instructions, see Dosage and Administration (2.4).
2.2 DosageModificationsforDrug Interactions
Nitrates
Concomitant use of nitrates in any form is contraindicated [see Contraindications (4.1), Drug Interactions (7.1) and Clinical Pharmacology (12.2)].
Alpha Blockers
Initiate VYBRIQUE at 25 mg orally in patients on concomitant therapy with an alpha-blocker. Patients should be stable on alpha-blocker therapy prior to initiating VYBRIQUE [seeWarningsand Precautions (5.5), Drug Interactions(7.2), and Clinical Pharmacology(12.2)]. For administration instructions, seeDosageand Administration (2.4).
Ritonavir
The maximum recommended dose and dosing frequency is 25 mg orally taken once within a 48-hour period in ritonavir-treated patients. Concomitant administration of ritonavir increased the blood levels of sildenafil by 11-fold [seeWarnings andPrecautions (5.6), Drug Interactions (7.4), and Clinical Pharmacology (12.3)]. For administration instructions, see Dosage and Administration (2.4).
OtherCYP3A4 Inhibitors
The recommended starting dosage is 25 mg orally, in patients taking strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, or saquinavir) or erythromycin. Clinical data have shown that co-administration with saquinavir or erythromycin increased blood levels of sildenafil by about 3-fold [seeDrug Interactions (7.4) and Clinical Pharmacology (12.3)]. For administration instructions, see Dosage and Administration (2.4).
2.3 RecommendedDosage inSpecificPopulations
Age Greater than 65years
The recommended starting dosage is 25 mg orally in patients greater than 65 years of age [see Use in Specific Populations(8.5)]. For administration instructions, see Dosage and Administration (2.4).
Renal Impairment
The recommended starting dosage is 25 mg orally, in patients with severe renal impairment (creatinine clearance less than 30 mL/minute). The recommended starting dosage is 50 mg, in patients with mild (creatinine clearance 50 to 80 mL/min) and moderate (creatinine clearance 30 to 49 mL/min) renal impairment [see Use in Specific Populations(8.6) and Clinical Pharmacology (12.3)]. For administration instructions, see Dosage and Administration (2.4).
Hepatic Impairment
The recommended starting dosage is 25 mg orally in patients with any degree of hepatic impairment. In volunteers with mild and moderate degrees of hepatic impairment (Child-Pugh Class A and B), sildenafil clearance was reduced, resulting in higher plasma exposure of sildenafil (47% for Cmax and 85% for AUC), and the pharmacokinetics of sildenafil in patients with severe hepatic impairment (Child-Pugh Class C) have not been studied [see Use in Specific Populations (8.7)and Clinical Pharmacology (12.3)]. For administration instructions, see Dosage and Administration (2.4).
-
3.
DOSAGE FORMS AND STRENGTHS
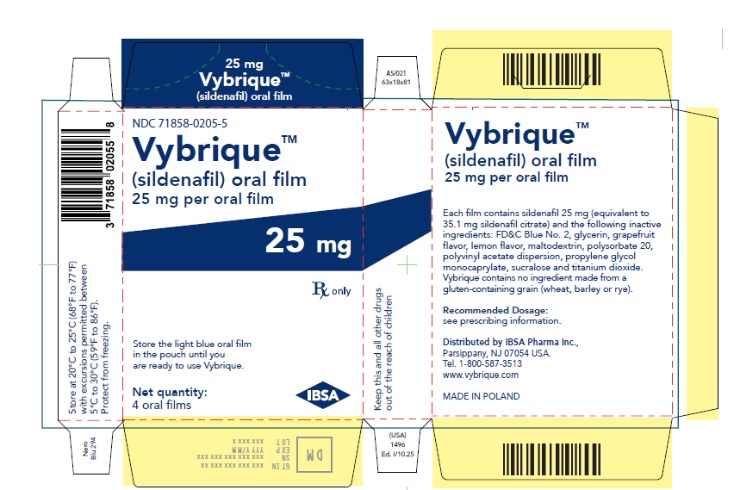
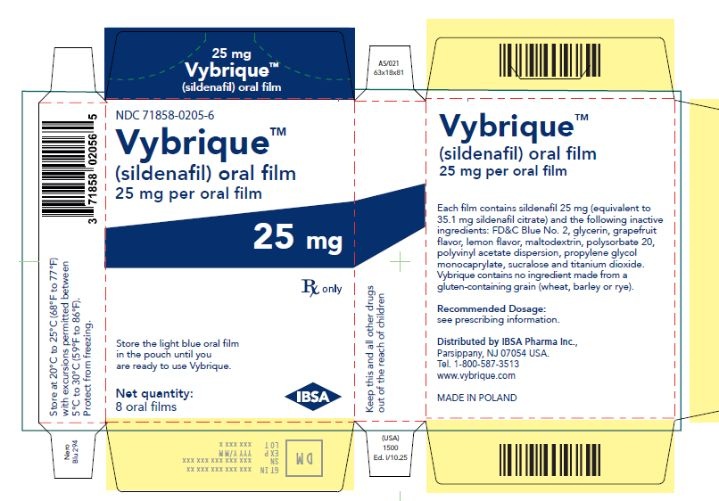
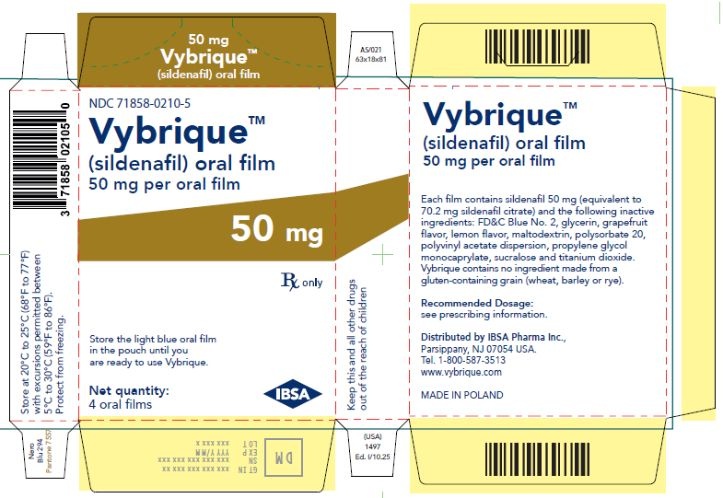
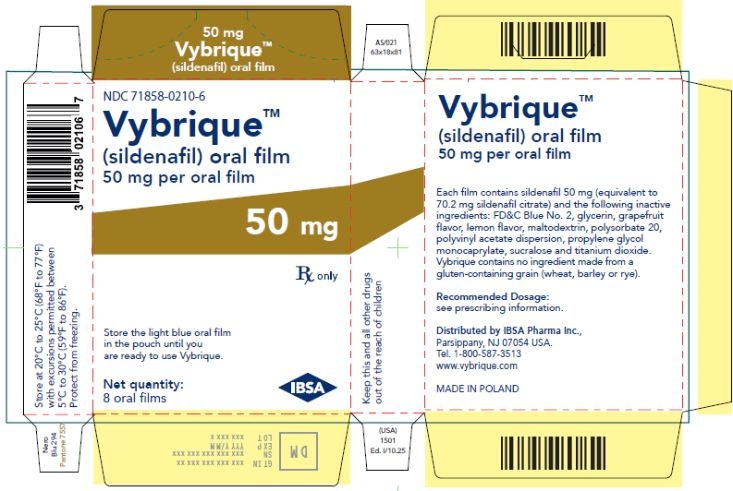
Oral Film:
- 25 mg sildenafil, opaque light blue, thin, flexible oral film imprinted with identification code S 25
- 50 mg sildenafil, opaque light blue, thin, flexible oral film imprinted with identification code S 50
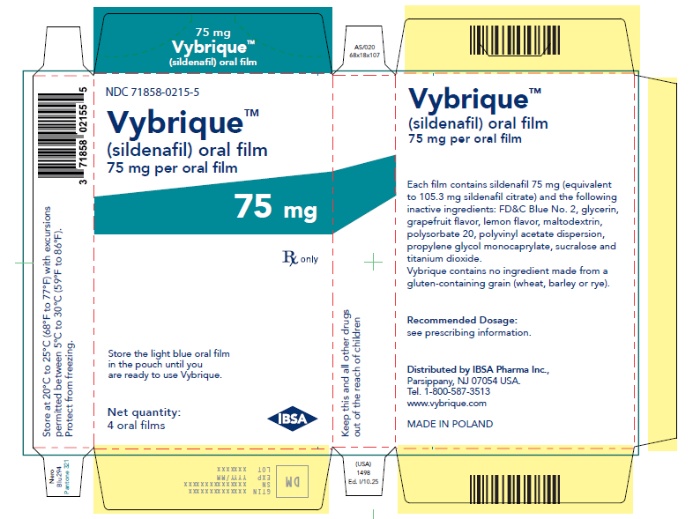
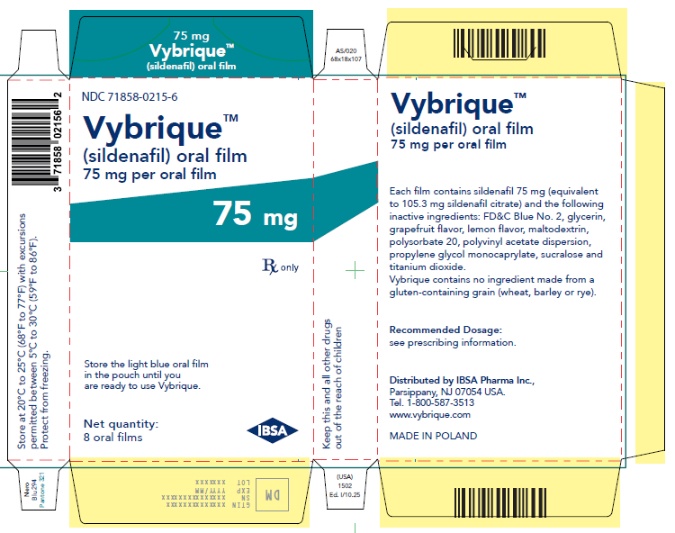
- 75 mg sildenafil, opaque light blue, thin, flexible oral film imprinted with identification code S 75
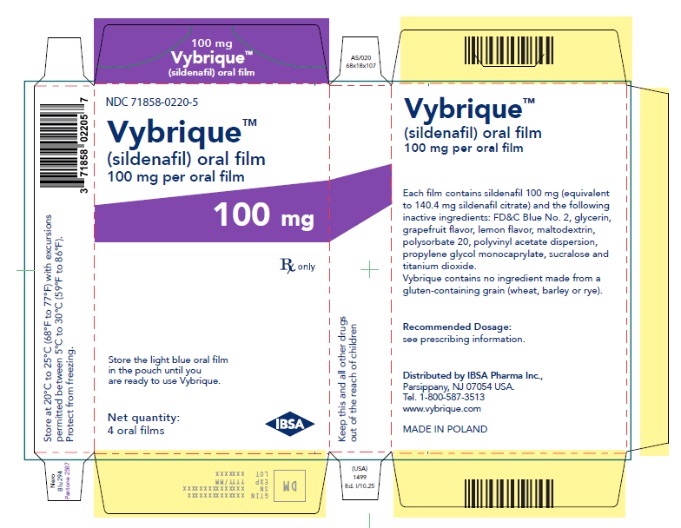
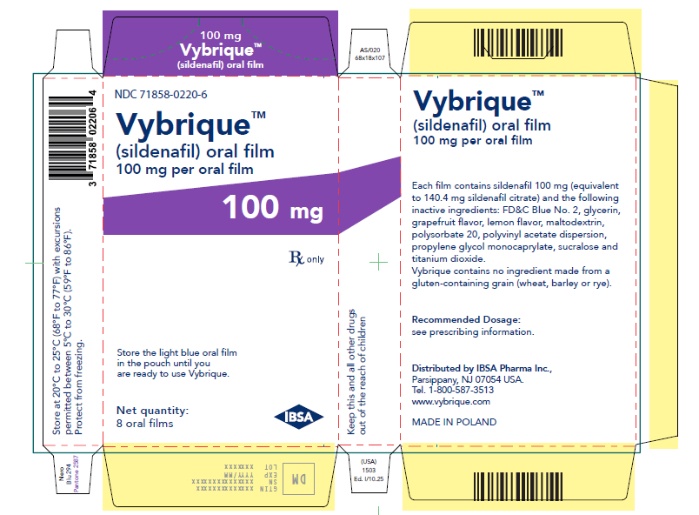
- 100 mg sildenafil, opaque light blue, thin, flexible oral film imprinted with identification code S 100
- 25 mg sildenafil, opaque light blue, thin, flexible oral film imprinted with identification code S 25
-
4.
CONTRAINDICATIONS
4.1 Nitrates
Consistent with its known effects on the nitric oxide/cGMP pathway [seeClinical Pharmacology (12.1,12.2)], VYBRIQUE potentiates the hypotensive effects of nitrates, and its administration to patients who are using nitric oxide donors such as organic nitrates or organic nitrites in any form either regularly and/or intermittently is therefore contraindicated.
After patients have taken VYBRIQUE it is unknown when nitrates, if necessary, can be safely administered. Although plasma levels of sildenafil at 24 hours post dose are much lower than at peak concentration, it is unknown whether nitrates can be safely co-administered at this time point [seeDosage and Administration (2.2), Drug Interactions (7.1), and Clinical Pharmacology (12.2)].
4.2 Hypersensitivity Reactions
VYBRIQUE is contraindicated in patients with a known hypersensitivity to sildenafil or any VYBRIQUE component. Hypersensitivity reactions, including rash and urticaria, have been reported [see Adverse Reactions (6.1)].
-
5.
WARNINGS AND PRECAUTIONS
5.1 CardiovascularRisk
General
There is a potential for cardiac risk of sexual activity in patients with preexisting cardiovascular disease. Therefore, treatments for erectile dysfunction, including VYBIRQUE should not be used in men for whom sexual activity is inadvisable because of their underlying cardiovascular status.
The following groups of patients were not included in clinical safety and efficacy trials for sildenafil, and therefore, until further information is available, VYBRIQUE is not recommended for use in the following groups:
- Patients who have suffered a myocardial infarction, stroke, or life-threatening arrhythmia within the last 6 months
- Patients with resting hypotension (BP <90/50 mmHg) or hypertension (BP >170/110 mmHg)
- Patients with cardiac failure or coronary artery disease causing unstable angina.
Blood PressureDecreases
Patients with left ventricular outflow obstruction (e.g., aortic stenosis, idiopathic hypertrophic subaortic stenosis) and those with severely impaired autonomic control of blood pressure can be sensitive to the actions of vasodilators, including VYBRIQUE.
As with other PDE5 inhibitors, VYBRIQUE has systemic vasodilatory properties that resulted in transient decreases in supine blood pressure in healthy volunteers. In healthy subjects aged 18 to 45 years, VYBRIQUE 100 mg resulted in mean maximal decreases relative to placebo of 6 mmHg systolic and 3 mmHg diastolic. In healthy subjects aged 65 years and older, VYBRIQUE 100 mg resulted in mean maximal decreases relative to placebo of 14 mmHg systolic and 8 mmHg diastolic [see Clinical Pharmacology (12.2)].
Prior to prescribing VYBRIQUE, carefully consider whether patients with underlying cardiovascular disease could be affected adversely by such vasodilatory effects, especially in combination with sexual activity.
5.2 Prolonged Erection and Priapism
Prolonged erection greater than 4 hours and priapism (painful erections greater than 6 hours in duration) have been reported infrequently since market approval of sildenafil. In the event of an erection that persists longer than 4 hours, instruct the patient to seek immediate medical assistance. If priapism is not treated immediately, penile tissue damage and permanent loss of potency could result.
Use VYBRIQUE with caution in patients with anatomical deformation of the penis (such as angulation, cavernosal fibrosis or Peyronie’s disease), or in patients who have conditions which may predispose them to priapism (such as sickle cell anemia, multiple myeloma, or leukemia).
5.3 Effects on the Eye
Advise patients to stop use of all phosphodiesterase type 5 (PDE5) inhibitors, including VYBRIQUE and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a rare condition and a cause of decreased vision including permanent loss of vision, that has been reported rarely post-marketing in temporal association with the use of all PDE5 inhibitors. Based on published literature, the annual incidence of NAION is 2.5-11.8 cases per 100,000 in males aged 50 years and older.
An observational case-crossover study evaluated the risk of NAION when PDE5 inhibitor use, as a class, occurred immediately before NAION onset (within 5 half-lives), compared to PDE5 inhibitor use in a prior time period. The results suggest an approximate 2-fold increase in the risk of NAION, with a risk estimate of 2.15 (95% CI 1.06, 4.34). A similar study reported a consistent result, with a risk estimate of 2.27 (95% CI 0.99, 5.20). Other risk factors for NAION, such as the presence of “crowded” optic disc, may have contributed to the occurrence of NAION in these studies.
Neither the rare post-marketing reports, nor the association of PDE5 inhibitor use and NAION in the observational studies, substantiate a causal relationship between PDE5 inhibitor use and NAION [see Adverse Reactions(6.2)].
Consider whether patients with underlying NAION risk factors could be adversely affected by use of PDE5 inhibitors. Individuals who have already experienced NAION are at increased risk of NAION recurrence. Therefore, use PDE5 inhibitors, including VYBRIQUE with caution in these patients and only when the anticipated benefits outweigh the risks. Individuals with “crowded” optic disc are also considered at greater risk for NAION compared to the general population, however, evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including VYBRIQUE, for this uncommon condition.
Patients with known hereditary degenerative retinal disorders, including retinitis pigmentosa, were not included in the sildenafil citrate clinical trials, and use in these patients is not recommended.
5.4 SuddenHearing Loss
Advise patients to stop taking PDE5 inhibitors, including VYBRIQUE and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including sildenafil. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions (6.1,6.2)].
5.5 Hypotension when Co-administered with Alpha-blockers or Antihypertensives
Alpha-blockers
Caution is advised when PDE5 inhibitors are co-administered with alpha-blockers. PDE5 inhibitors, including sildenafil, and alpha-adrenergic blocking agents are both vasodilators with blood pressure lowering effects. When vasodilators are used in combination, an additive effect on blood pressure may occur. In some patients, concomitant use of these two drug classes can lower blood pressure significantly [see Drug Interactions (7.2) and Clinical Pharmacology (12.2)]leadingto symptomatic hypotension (e.g., dizziness, lightheadedness, fainting).
Consider the following:
- Patients should be stable on alpha-blocker therapy prior to initiating a PDE5 inhibitor. Patients who demonstrate hemodynamic instability on alpha-blocker therapy alone are at increased risk of symptomatic hypotension with concomitant use of PDE5 inhibitors.
- In patients who are stable on alpha-blocker therapy, PDE5 inhibitors should be initiated at the lowest dose
[see
Dosage andAdministration(2.2)].
- In patients already taking an optimized dose of a PDE5 inhibitor, alpha-blocker therapy should be initiated at the lowest dose. Stepwise increase in alpha-blocker dose may be associated with further lowering of blood pressure when taking a PDE5 inhibitor.
- Safety of combined use of PDE5 inhibitors and alpha-blockers may be affected by other variables, including intravascular volume depletion and other antihypertensive drugs.
Antihypertensives
VYBRIQUE has systemic vasodilatory properties and may further lower blood pressure in patients taking antihypertensive medications.
When amlodipine, 5 mg or 10 mg, and sildenafil 100 mg were orally administered concomitantly to hypertensive patients, mean additional blood pressure reductions of 8 mmHg systolic and 7 mmHg diastolic were noted [see DrugInteractions (7.3) and Clinical Pharmacology (12.2) ].
5.6 Adverse Reactions with the Concomitant Use of Ritonavir
The concomitant administration of the protease inhibitor ritonavir substantially increases serum concentrations of sildenafil (11-fold increase in AUC). Data from subjects exposed to high systemic levels of sildenafil are limited. Decreased blood pressure, syncope, and prolonged erection were reported in some healthy volunteers exposed to high doses of sildenafil (200-800 mg). To decrease the chance of adverse reactions in patients taking ritonavir, the maximum recommended dosage and dosing frequency of VYBRIQUE is 25 mg once in a 48-hour period [seeDosage and Administration(2.2), Drug Interactions (7.4), and Clinical Pharmacology (12.3)].
5.7 Combination with other PDE5 Inhibitors or Other Erectile Dysfunction Therapies
The safety and efficacy of combinations of VYBRIQUE with other PDE5 inhibitors, including pulmonary arterial hypertension (PAH) treatments containing sildenafil or other treatments for erectile dysfunction, have not been studied. Such combinations may further lower blood pressure. Therefore, the use of such combinations is not recommended.
5.8 Effects on Bleeding
There have been postmarketing reports of bleeding events in patients who have taken sildenafil. A causal relationship between sildenafil and these events has not been established. In humans, sildenafil has no effect on bleeding time when taken alone or with aspirin. However, in vitro studies with human platelets indicate that sildenafil potentiates the antiaggregatory effect of sodium nitroprusside (a nitric oxide donor). In addition, the combination of heparin and sildenafil had an additive effect on bleeding time in the anesthetized rabbit, but this interaction has not been studied in humans.
The safety of sildenafil is unknown in patients with bleeding disorders and patients with active peptic ulceration.
5.9 Counseling Patients About Sexually Transmitted Diseases
The use of VYBRIQUE offers no protection against sexually transmitted diseases. Counseling of patients about the protective measures necessary to guard against sexually transmitted diseases, including the Human Immunodeficiency Virus (HIV), should be considered.
- Patients who have suffered a myocardial infarction, stroke, or life-threatening arrhythmia within the last 6 months
-
6.
ADVERSE REACTIONS
The following are discussed in more detail in other sections of the labeling:
- Cardiovascular
[seeWarnings and Precautions (5.1)]
- Prolonged Erection and Priapism
[seeWarnings and Precautions (5.2)]
- Effects on the Eye
[seeWarnings and Precautions (5.3)]
- Hearing Loss
[seeWarnings and Precautions (5.4)]
- Hypotension when Co-administered with Alpha-blockers or Anti-hypertensives
[seeWarnings and Precautions (5.5)]
- Adverse Reactions with the Concomitant Use of Ritonavir
[seeWarnings and Precautions5.6)]
- Combination with other PDE5 Inhibitors or Other Erectile Dysfunction Therapies [see
Warnings and Precautions (5.7)]
- Effects on Bleeding [seeWarnings and Precautions (5.8)]
The most common adverse reactions reported in clinical trials (≥ 2%) of sildenafil are headache, flushing, dyspepsia, abnormal vision, nasal congestion, back pain, myalgia, nausea, dizziness, and rash.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Sildenafil was administered to over 3700 patients (aged 19-87 years) during pre-marketing clinical trials worldwide. Over 550 patients were treated for longer than one year.
In placebo-controlled clinical studies, the discontinuation rate due to adverse reactions for sildenafil (2.5%) was not significantly different from placebo (2.3%).
In fixed-dose studies, the incidence of some adverse reactions increased with dose. The type of adverse events in flexible-dose studies was similar to that for fixed dose studies. At doses above the recommended dose range, adverse reactions were similar to those detailed in Table 1 but generally were reported more frequently.
Table 1: Adverse Reactions Reported by ≥2% of Patients Treated with Sildenafil and More Frequent than Placebo in Fixed-Dose Clinical Studies Adverse Reaction 25 mg
(n=312)50 mg
(n=511)100 mg
(n=506)Placebo
(n=607)Headache 16% 21% % 28 % 7 Flushing 10% 19% 18% 2% Dyspepsia 3% 9% 17% 2% Abnormal vision† 1% 2% 11% 1% Nasal congestion 4% 4% 9% 2% Back pain 3% 4% 4% 2% Myalgia 2% 2% 4% 1% Nausea 2% 3% 3% 1% Dizziness 3% 4% 3% 2% Rash 1% 2% 3% 1% †Abnormal Vision: Mild to moderate in severity and transient, predominantly color tinge to vision, but also increased sensitivity to light, or blurred vision. When sildenafil was taken as recommended (on an as-needed basis) in flexible-dose, placebo-controlled clinical trials of two to twenty-six weeks duration, patients took sildenafil at least once weekly, and the following adverse reactions were reported:
Table 2 : Adverse Reactions Reported by ≥2% of Patients Treated with Sildenafil and More Frequent than Placebo in Flexible-Dose Clinical Studies Adverse Reaction Sildenafil
N=734Placebo
N=725Headache 16% 4% Flushing 10% 1% Dyspepsia 7% 2% Nasal Congestion 4% 2% Abnormal Vision† 3% 0% Back pain 2% 2% Dizziness 2% 1% Rash 2% 1% †Abnormal Vision: Mild to moderate in severity and transient, predominantly color tinge to vision, but also increased sensitivity to light or blurred vision. In these studies, only one patient discontinued due to abnormal vision. When VYBRIQUE was taken as recommended (on an as-needed basis) in a flexible-dose, placebo-controlled clinical trial of twelve weeks duration, patients took VYBRIQUE at least once weekly, not more than once per day, and the following adverse reactions were reported:
Table 3: Adverse Reactions Reported by ≥2% of Patients Treated with VYBRIQUE and More Frequent than Placebo in a Flexible-Dose Clinical Study AdverseReactions VYBRIQUE
N=354PLACEBO
N=121Headache 11% 4% Flushing 4% 0% Dyspepsia 4% 1% Nasal Congestion 3% 0% Dizziness 2% 0% In this study, none of the patients discontinued due to adverse reactions to VYBRIQUE.
The following events occurred in <2% of patients in controlled clinical trials of sildenafil; a causal relationship to sildenafil is uncertain. Reported events include those with a plausible relation to drug use; omitted are minor events and reports too imprecise to be meaningful:
Body as a Whole:face edema, photosensitivity reaction, shock, asthenia, pain, chills, accidental fall, abdominal pain, allergic reaction, chest pain, accidental injury.
Cardiovascular:angina pectoris, AV block, migraine, syncope, tachycardia, palpitation, hypotension, postural hypotension, myocardial ischemia, cerebral thrombosis, cardiac arrest, heart failure, abnormal electrocardiogram, cardiomyopathy.
Digestive:vomiting, glossitis, colitis, dysphagia, gastritis, gastroenteritis, esophagitis, stomatitis, dry mouth, liver function tests abnormal, rectal hemorrhage, gingivitis.
Hemic and Lymphatic:anemia and leukopenia.
Metabolic and Nutritional:thirst, edema, gout, unstable diabetes, hyperglycemia, peripheral edema, hyperuricemia, hypoglycemic reaction, hypernatremia.
Musculoskeletal:arthritis, arthrosis, myalgia, tendon rupture, tenosynovitis, bone pain, myasthenia, synovitis.
Nervous:ataxia, hypertonia, neuralgia, neuropathy, paresthesia, tremor, vertigo, depression, insomnia, somnolence, abnormal dreams, reflexes decreased, hypesthesia.
Respiratory:asthma, dyspnea, laryngitis, pharyngitis, sinusitis, bronchitis, sputum increased, cough increased.
Skin and Appendages:urticaria, herpes simplex, pruritus, sweating, skin ulcer, contact dermatitis, exfoliative dermatitis.
Special Senses:sudden decrease or loss of hearing, mydriasis, conjunctivitis, photophobia, tinnitus, eye pain, ear pain, eye hemorrhage, cataract, dry eyes.
Urogenital:cystitis, nocturia, urinary frequency, breast enlargement, urinary incontinence, abnormal ejaculation, genital edema and anorgasmia.
Analysis of the safety database from sildenafil controlled clinical trials showed no apparent difference in adverse reactions in patients taking sildenafil with and without antihypertensive medication. This analysis was performed retrospectively and was not powered to detect any pre-specified difference in adverse reactions.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of sildenafil. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events have been chosen for inclusion either due to their seriousness, reporting frequency, lack of clear alternative causation, or a combination of these factors.
Cardiovascular andCerebrovascular:serious cardiovascular, cerebrovascular, and vascular events, including myocardial infarction, sudden cardiac death, ventricular arrhythmia, cerebrovascular hemorrhage, transient ischemic attack, hypertension, subarachnoid and intracerebral hemorrhages, and pulmonary hemorrhage have been reported post-marketing in temporal association with the use of sildenafil. Most, but not all, of these patients had preexisting cardiovascular risk factors. Many of these events were reported to occur during or shortly after sexual activity, and a few were reported to occur shortly after the use of sildenafil without sexual activity. Others were reported to have occurred hours to days after the use of sildenafil and sexual activity. It is not possible to determine whether these events are related directly to sildenafil, to sexual activity, to the patient’s underlying cardiovascular disease, to a combination of these factors, or to other factors [see Warnings and Precautions (5.1) and Patient Counseling Information (17)].
Hemic and Lymphatic: vaso-occlusive crisis: In a small, prematurely terminated study of sildenafil in patients with pulmonary arterial hypertension (PAH) secondary to sickle cell disease, vaso-occlusive crises requiring hospitalization were more commonly reported in patients who received sildenafil than in those randomized to placebo. The clinical relevance of this finding to men treated with sildenafil for ED is not known.
Nervous:seizure, seizure recurrence, anxiety, and transient global amnesia.
Respiratory:epistaxis
Special senses:
Hearing:Cases of sudden decrease or loss of hearing have been reported postmarketing in temporal association with the use of PDE5 inhibitors, including sildenafil. In some of the cases, medical conditions and other factors were reported that may have also played a role in the otologic adverse events. In many cases, medical follow-up information was limited. It is not possible to determine whether these reported events are related directly to the use of sildenafil, to the patient’s underlying risk factors for hearing loss, a combination of these factors, or to other factors [see Warnings and Precautions (5.4),Patient Counseling Information (17)].
Ocular:diplopia, temporary vision loss/decreased vision, ocular redness or bloodshot appearance, ocular burning, ocular swelling/pressure, increased intraocular pressure, retinal edema, retinal vascular disease or bleeding, and vitreous traction/detachment.
Non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision including permanent loss of vision, has been reported rarely post-marketing in temporal association with the use of phosphodiesterase type 5 (PDE5) inhibitors, including sildenafil. Most, but not all, of these patients had underlying anatomic or vascular risk factors for developing NAION, including but not necessarily limited to: low cup to disc ratio (“crowded disc”), age over 50, diabetes, hypertension, coronary artery disease, hyperlipidemia and smoking [seeWarnings and Precautions (5.3), Patient Counseling Information (17)].
Urogenital: prolonged erection, priapism [see Warnings and Precautions (5.2) and Patient Counseling Information (17)],and hematuria.
- Cardiovascular
[seeWarnings and Precautions (5.1)]
-
7.
DRUG INTERACTIONS
7.1 Nitrates
Administration of sildenafil with nitric oxide donors such as organic nitrates or organic nitrites in any form is contraindicated. Consistent with its known effects on the nitric oxide/cGMP pathway, sildenafil was shown to potentiate the hypotensive effects of nitrates [seeDosage and Administration (2.2), Contraindications (4.1), Clinical Pharmacology (12.2)] .
7.2 Alpha-blockers
Use caution when co-administering alpha-blockers with sildenafil because of potential additive blood pressure lowering effects. When sildenafil is co-administered with an alpha-blocker, patients should be stable on alpha blocker therapy prior to initiating sildenafil treatment and sildenafil should be initiated at the lowest dose [seeDosage andAdministration(2.2),Warnings and Precautions (5.5), Clinical Pharmacology (12.2)].
7.3 Amlodipine
When sildenafil 100 mg was co-administered with amlodipine (5 mg or 10 mg) to hypertensive patients, the mean additional reduction on supine blood pressure was 8 mmHg systolic and 7 mmHg diastolic [seeWarnings and Precautions (5.5), Clinical Pharmacology (12.2)].
7.4 Ritonavir and other CYP3A4 inhibitors
Co-administration of ritonavir, a strong CYP3A4 inhibitor, greatly increased the systemic exposure of sildenafil (11-fold increase in AUC). It is therefore recommended not to exceed a maximum single dose of 25 mg of sildenafil in a 48 hour period [seeDosage and Administration(2.2), Warnings and Precautions (5.6), Clinical Pharmacology (12.3)].
Co-administration of erythromycin, a moderate CYP3A4 inhibitor, resulted in 160% and 182% increases in sildenafil Cmax and AUC, respectively. Co-administration of saquinavir, a strong CYP3A4 inhibitor, resulted in 140% and 210% increases in sildenafil Cmax and AUC, respectively. Stronger CYP3A4 inhibitors such as ketoconazole or itraconazole could be expected to have greater effects than seen with saquinavir. A starting dose of 25 mg of sildenafil should be considered in patients taking erythromycin or strong CYP3A4 inhibitors (such as saquinavir, ketoconazole, itraconazole) [see Dosage and Administration(2.2), Clinical Pharmacology (12.3)].
7.5 Alcohol
In a drug-drug interaction study of sildenafil 50 mg given with alcohol 0.5 g/kg, in which mean maximum blood alcohol levels of 0.08% was achieved, sildenafil did not potentiate the hypotensive effect of alcohol in healthy volunteers [seeClinical Pharmacology (12.2)].
-
8.
USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
VYBRIQUE is not indicated for use in females.
There are no data with the use of VYBRIQUE in pregnant women to inform any drug-associated risks for adverse developmental outcomes. Animal reproduction studies conducted with sildenafil did not show adverse developmental outcomes when administered during organogenesis in rats and rabbits at oral doses up to 16 and 32 times, respectively, the maximum recommended human dose (MRHD) of 100 mg/day on a mg/m2 basis ( see Data).
Data
Animal Data
No evidence of teratogenicity, embryotoxicity or fetotoxicity was observed in rats and rabbits which received oral doses up to 200 mg/kg/day during organogenesis. These doses represent, respectively, about 16 and 32 times the MRHD on a mg/m2 basis in a 50 kg subject. In the rat pre- and postnatal development study, the no observed adverse effect dose was 30 mg/kg/day given for 36 days, about 2 times the MRHD on a mg/m2 basis in a 50 kg subject.
8.2 Lactation
Risk Summary
VYBRIQUE is not indicated for use in females.
Limited data indicate that sildenafil and its active metabolite are present in human milk. There is no information on the effects on the breastfed child, or the effects on milk production.
8.4 Pediatric Use
VYBRIQUE is not indicated for use in pediatric patients. Safety and effectiveness have not been established in pediatric patients.
8.5 Geriatric Use
Healthy elderly volunteers (65 years or over) had a reduced clearance of sildenafil resulting in approximately 84% and 107% higher plasma AUC values of sildenafil and its active N-desmethyl metabolite, respectively, compared to those seen in healthy young volunteers (18-45 years) [seeClinical Pharmacology (12.3)].Due to age-differences in plasma protein binding, the corresponding increase in the AUC of free (unbound) sildenafil and its active N-desmethyl metabolite were 45% and 57%, respectively [seeClinical Pharmacology (12.3)].
Of the total number of subjects in clinical studies of sildenafil, 18% were 65 years and older, while 2% were 75 years and older. No overall differences in safety or efficacy were observed between older (≥ 65 years of age) and younger adult (< 65 years of age) subjects.
Of the total number of subjects in the clinical study of VYBRIQUE, 31% were 65 years and older. No overall differences in safety or efficacy were observed between older (≥ 65 years of age) and younger (< 65 years of age) subjects.
A starting dose of VYBRIQUE 25 mg is recommended in patients 65 years of age and older due to the higher systemic exposure in older subjects, and larger decreases in blood pressure observed in older subjects in a clinical pharmacology study [seeDosage and Administration(2.3),Warnings and Precautions (5.1), Clinical Pharmacology(12.2].
8.6 Renal Impairment
No dose adjustment is required for mild (CLcr=50-80 mL/min) and moderate (CLcr=30-49 mL/min) renal impairment. In volunteers with severe renal impairment (Clcr<30 mL/min), sildenafil clearance was reduced, resulting in higher plasma exposure of sildenafil (~2 fold), approximately doubling of Cmax and AUC. A starting dose of 25 mg should be considered in patients with severe renal impairment [seeDosage and Administration (2.3),Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
In volunteers with hepatic impairment (Child-Pugh Class A and B), sildenafil clearance was reduced, resulting in higher plasma exposure of sildenafil (47% for Cmax and 85% for AUC). The pharmacokinetics of sildenafil in patients with severely impaired hepatic function (Child-Pugh Class C) have not been studied. A starting dose of 25 mg should be considered in patients with any degree of hepatic impairment [seeDosage and Administration (2.3),Clinical Pharmacology (12.3)].
-
10
OVERDOSAGE
In studies in healthy volunteers administered single sildenafil doses up to 800 mg, adverse reactions were similar to those seen at lower doses, but incidence rates and severities were increased.
In cases of overdose, standard supportive measures should be adopted as required. Renal dialysis is not expected to accelerate clearance as sildenafil is highly bound to plasma proteins and it is not eliminated in the urine.
-
11
DESCRIPTION
VYBRIQUE (sildenafil) oral film is for treatment of erectile dysfunction and contains sildenafil citrate, a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5).
Sildenafil citrate is designated chemically as 1-[[3-(6,7-dihydro-1-methyl-7-oxo-3-propyl-1H-pyrazolo[4,3-d]pyrimidin-5-yl)-4-ethoxyphenyl]sulfonyl]-4-methylpiperazine citrate, has the molecular formula C 22H 39N 6O 4S ‧ C 6H 8O 7, and the following structural formula:
![Sildenafil citrate is designated chemically as 1-[[3-(6,7-dihydro-1-methyl-7-oxo-3-propyl-1H-pyrazolo[4,3-d]pyrimidin-5-yl)-4-ethoxyphenyl]sulfonyl]-4-methylpiperazine citrate, has the molecular formu](https://fda.report/DailyMed/9213fec1-e0c0-4ef7-9718-d97922a3f4d8/vybrique-sildenafil-citrate-oral-film-01.jpg)
Sildenafil citrate is a white to off-white crystalline powder with a solubility of 3.5 mg/mL in water and a molecular weight of 666.7.
VYBRIQUE is formulated as an opaque light blue, thin, flexible oral film with the characteristic lemon and grapefruit scent. The product is available in four different strengths 25, 50, 75, or 100 mg of sildenafil equivalent to 35, 70, 105, 140 mg sildenafil citrate respectively for oral administration. In addition to the active ingredient, sildenafil citrate, each oral film contains the following inactive ingredients: Blue Videojet ink, FD&C Blue No.2, glycerin, grapefruit flavor, lemon flavor, maltodextrin, polysorbate 20, polyvinyl acetate dispersion, propylene glycol monocaprylate, sucralose, titanium dioxide. VYBRIQUE contains no ingredient made from a gluten-containing grain (wheat, barley, or rye).
-
12
CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The physiologic mechanism of erection of the penis involves release of nitric oxide (NO) in the corpus cavernosum during sexual stimulation. NO then activates the enzyme guanylate cyclase, which results in increased levels of cyclic guanosine monophosphate (cGMP), producing smooth muscle relaxation in the corpus cavernosum and allowing inflow of blood.
Sildenafil enhances the effect of NO by inhibiting phosphodiesterase type 5 (PDE5), which is responsible for degradation of cGMP in the corpus cavernosum. Sildenafil has no direct relaxant effect on isolated human corpus cavernosum. When sexual stimulation causes local release of NO, inhibition of PDE5 by sildenafil causes increased levels of cGMP in the corpus cavernosum, resulting in smooth muscle relaxation and inflow of blood to the corpus cavernosum. Sildenafil at recommended doses has no effect in the absence of sexual stimulation.
Binding Characteristics
Studies in vitro have shown that sildenafil is selective for PDE5. Its effect is more potent on PDE5 than on other known phosphodiesterases (10-fold for PDE6, >80-fold for PDE1, >700-fold for PDE2, PDE3, PDE4, PDE7, PDE8, PDE9, PDE10, and PDE11). Sildenafil is approximately 4,000-fold more selective for PDE5 compared to PDE3. PDE3 is involved in control of cardiac contractility. Sildenafil is only about 10-fold as potent for PDE5 compared to PDE6, an enzyme found in the retina which is involved in the phototransduction pathway of the retina. This lower selectivity is thought to be the basis for abnormalities related to color vision [seeClinical Pharmacology (12.2)].
In addition to human corpus cavernosum smooth muscle, PDE5 is also found in other tissues including platelets, vascular and visceral smooth muscle, and skeletal muscle, brain, heart, liver, kidney, lung, pancreas, prostate, bladder, testis, and seminal vesicle. The inhibition of PDE5 in some of these tissues by sildenafil may be the basis for the enhanced platelet antiaggregatory activity of NO observed in vitro, an inhibition of platelet thrombus formation in vivo and peripheral arterial-venous dilatation in vivo.
12.2 Pharmacodynamics
Effects ofSildenafil on Erectile Response:In eight double-blind, placebo-controlled crossover studies of patients with either organic or psychogenic erectile dysfunction, sexual stimulation resulted in improved erections, as assessed by an objective measurement of hardness and duration of erections (RigiScan®), after sildenafil administration compared with placebo. Most studies assessed the efficacy of sildenafil approximately 60 minutes post dose. The erectile response, as assessed by RigiScan®, generally increased with increasing sildenafil dose and plasma concentration. The time course of effect was examined in one study, showing an effect for up to 4 hours but the response was diminished compared to 2 hours.
Effects ofVYBRIQUE on Blood Pressure:
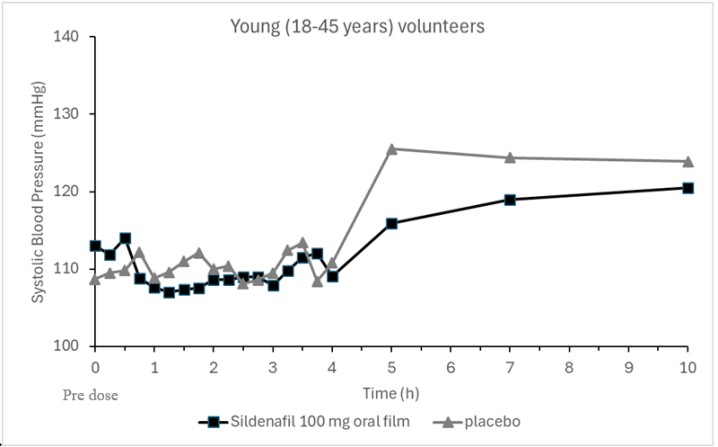
Single oral doses (50 mg, 75 mg, 100 mg) of VYBRIQUE administered to 12 healthy young volunteers produced decreases in supine blood pressure. The placebo-subtracted mean maximum decreases in systolic/diastolic blood pressure were 6.1/2.8 mmHg after a single 100 mg dose. Subjects were under fasting conditions and lay in the supine position for the first 4 hours post-dose and were not initially titrated. The decrease in supine blood pressure was most notable approximately 1-2 hours after dosing.
Figure1:Mean Change from Baseline inSupineSystolic Blood Pressure, Healthy Volunteers
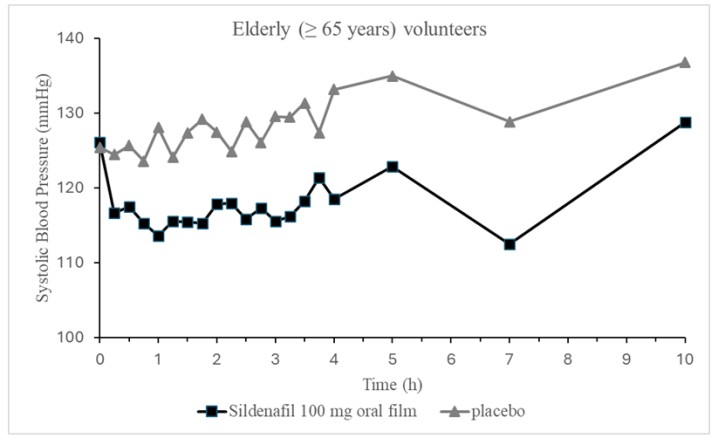
Single oral doses of 50 mg, 75 mg, 100 mg of VYBRIQUE administered to 12 elderly volunteers produced decreases in supine blood pressure. The placebo-subtracted maximum mean decreases in systolic/diastolic blood pressure were 13.7/8.5 mmHg after a single 100 mg dose. Subjects were under fasting conditions and lay in the supine position for the first 4 hours post-dose and were not initially titrated. The decrease in supine blood pressure was generally observed from 15 min to 4 h post-dose.
Figure2: Mean Change from Baseline inSupineSystolic Blood Pressure,Elderly
Effects ofSildenafil on Blood Pressure When Nitroglycerin is Subsequently Administered:Based on the pharmacokinetic profile of a single 100 mg sildenafil oral dose given to healthy normal volunteers, the plasma levels of sildenafil at 24 hours post dose are approximately 2 ng/mL (compared to peak plasma levels of approximately 440 ng/mL). In the following patients: age >65 years, hepatic impairment (e.g., cirrhosis), severe renal impairment (e.g., creatinine clearance <30 mL/min), and concomitant use of erythromycin or strong CYP3A4 inhibitors, plasma levels of sildenafil at 24 hours post dose have been found to be 3 to 8 times higher than those seen in healthy volunteers. Although plasma levels of sildenafil at 24 hours post dose are much lower than at peak concentration, it is unknown whether nitrates can be safely co-administered at this time point [see Contraindications (4.1)].
Effects ofSildenafil on Blood Pressure When Co-administered with Alpha-Blockers:Three double-blind, placebo-controlled, randomized, two-way crossover studies were conducted to assess the interaction of sildenafil with doxazosin, an alpha-adrenergic blocking agent.
Study 1:Sildenafil with Doxazosin
In the first study, a single oral dose of sildenafil 100 mg or matching placebo was administered in a 2-period crossover design to 4 generally healthy males with benign prostatic hyperplasia (BPH). Following at least 14 consecutive daily doses of doxazosin, sildenafil 100 mg or matching placebo was administered simultaneously with doxazosin. Following a review of the data from these first 4 subjects (details provided below), the sildenafil dose was reduced to 25 mg. Thereafter, 17 subjects were treated with sildenafil 25 mg or matching placebo in combination with doxazosin 4 mg (15 subjects) or doxazosin 8 mg (2 subjects). The mean subject age was 66.5 years.
For the 17 subjects who received sildenafil 25 mg and matching placebo, the placebo-subtracted mean maximum decreases from baseline (95% CI) in systolic blood pressure were as follows:
Placebo-subtracted mean maximum decreasein systolic blood pressure (mm Hg) Sildenafil 25 mg Supine 7.4 (-0.9 ,15.7) Standing 6.0 (-0.8, 12.8) The mean profiles of the change from baseline in standing systolic blood pressure in subjects treated with doxazosin in combination with 25 mg sildenafil or matching placebo are shown in Figure 3.
Figure3:Mean Standing Systolic Blood Pressure Change from Baseline
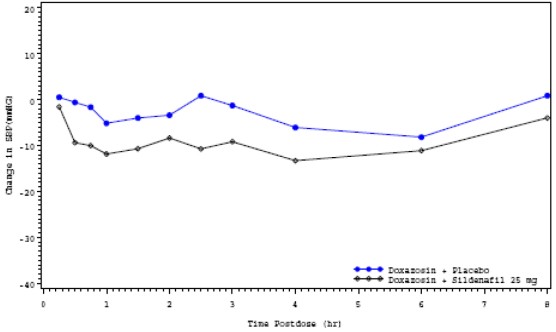
Blood pressure was measured immediately pre-dose and at 15, 30, 45 minutes, and 1, 1.5, 2, 2.5, 3, 4, 6 and 8 hours after sildenafil or matching placebo. Outliers were defined as subjects with a standing systolic blood pressure of <85 mmHg or a decrease from baseline in standing systolic blood pressure of >30 mmHg at one or more timepoints. There were no subjects treated with sildenafil 25 mg who had a standing SBP <85mmHg. There were three subjects with a decrease from baseline in standing systolic BP >30mmHg following sildenafil 25 mg, one subject with a decrease from baseline in standing systolic BP > 30 mmHg following placebo and two subjects with a decrease from baseline in standing systolic BP >30 mmHg following both sildenafil and placebo. No severe adverse events potentially related to blood pressure effects were reported in this group.
Of the four subjects who received sildenafil 100 mg in the first part of this study, a severe adverse event related to blood pressure effect was reported in one patient (postural hypotension that began 35 minutes after dosing with sildenafil with symptoms lasting for 8 hours), and mild adverse events potentially related to blood pressure effects were reported in two others (dizziness, headache and fatigue at 1 hour after dosing; and dizziness, lightheadedness and nausea at 4 hours after dosing). There were no reports of syncope among these patients. For these four subjects, the placebo-subtracted mean maximum decreases from baseline in supine and standing systolic blood pressures were 14.8 mmHg and 21.5 mmHg, respectively. Two of these subjects had a standing SBP <85mmHg. Both these subjects were protocol violators, one due to a low baseline standing SBP, and the other due to baseline orthostatic hypotension.
Study 2: Sildenafil with Doxazosin
In the second study, a single oral dose of sildenafil 50 mg or matching placebo was administered in a 2-period crossover design to 20 generally healthy males with BPH. Following at least 14 consecutive days of doxazosin, sildenafil 50 mg or matching placebo was administered simultaneously with doxazosin 4 mg (17 subjects) or with doxazosin 8 mg (3 subjects). The mean subject age in this study was 63.9 years.
Twenty subjects received sildenafil 50 mg, but only 19 subjects received matching placebo. One patient discontinued the study prematurely due to an adverse event of hypotension following dosing with sildenafil 50 mg. This patient had been taking minoxidil, a potent vasodilator, during the study.
For the 19 subjects who received both sildenafil and matching placebo, the placebo-subtracted mean maximum decreases from baseline (95% CI) in systolic blood pressure were as follows:
Placebo-subtracted mean maximum decrease in systolic blood pressure (mm Hg) Sildenafil50 mg (95% CI) Supine 9.08 (5.48, 12.68) Standing 11.62 (7.34, 15.90) The mean profiles of the change from baseline in standing systolic blood pressure in subjects treated with doxazosin in combination with 50 mg sildenafil or matching placebo are shown in Figure 4.
Figure4: Mean Standing Systolic Blood Pressure Change from Baseline
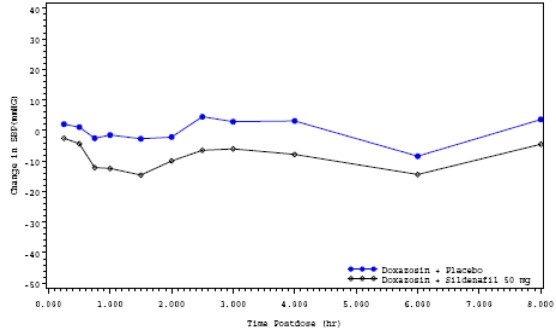
Blood pressure was measured after administration of sildenafil at the same times as those specified for the first doxazosin study. There were two subjects who had a standing SBP of <85 mmHg. In these two subjects, hypotension was reported as a moderately severe adverse event, beginning at approximately 1 hour after administration of sildenafil 50 mg and resolving after approximately 7.5 hours. There was one subject with a decrease from baseline in standing systolic BP >30mmHg following sildenafil 50 mg and one subject with a decrease from baseline in standing systolic BP >30 mmHg following both sildenafil 50 mg and placebo. There were no severe adverse events potentially related to blood pressure and no episodes of syncope reported in this study.
Study 3:Sildenafilwith Doxazosin
In the third study, a single oral dose of sildenafil 100 mg or matching placebo was administered in a 3-period crossover design to 20 generally healthy males with BPH. In dose period 1, subjects were administered open label doxazosin and a single dose of sildenafil 50 mg simultaneously, after at least 14 consecutive days of doxazosin. If a subject did not successfully complete this first dosing period, he was discontinued from the study. Subjects who had successfully completed the previous doxazosin interaction study (using sildenafil 50 mg), including no significant hemodynamic adverse events, were allowed to skip dose period 1. Treatment with doxazosin continued for at least 7 days after dose period 1. Thereafter, sildenafil 100 mg or matching placebo was administered simultaneously with doxazosin 4 mg (14 subjects) or doxazosin 8 mg (6 subjects) in standard crossover fashion. The mean subject age in this study was 66.4 years.
Twenty-five subjects were screened. Two were discontinued after study period 1: one failed to meet pre-dose screening qualifications and the other experienced symptomatic hypotension as a moderately severe adverse event 30 minutes after dosing with open-label sildenafil 50 mg. Of the twenty subjects who were ultimately assigned to treatment, a total of 13 subjects successfully completed dose period 1, and seven had successfully completed the previous doxazosin study (using sildenafil 50 mg).
For the 20 subjects who received sildenafil 100 mg and matching placebo, the placebo-subtracted mean maximum decreases from baseline (95% CI) in systolic blood pressure were as follows:
Placebo-subtracted mean maximum decrease in systolic blood pressure (mm Hg) Sildenafil100 mg Supine 7.9 (4.6, 11.1) Standing 4.3 (-1.8,10.3) The mean profiles of the change from baseline in standing systolic blood pressure in subjects treated with doxazosin in combination with 100 mg sildenafil citrate or matching placebo are shown in Figure 5.
Figure5: Mean Standing Systolic Blood Pressure Change from Baseline
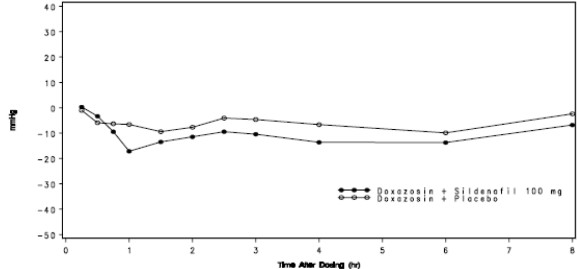
Blood pressure was measured after administration of sildenafil at the same times as those specified for the previous doxazosin studies. There were three subjects who had a standing SBP of <85 mmHg. All three were taking sildenafil 100 mg, and all three reported mild adverse events at the time of reductions in standing SBP, including vasodilation and lightheadedness. There were four subjects with a decrease from baseline in standing systolic BP >30 mmHg following sildenafil 100 mg, one subject with a decrease from baseline in standing systolic BP >30 mmHg following placebo and one subject with a decrease from baseline in standing systolic BP >30 mmHg following both sildenafil citrate and placebo. While there were no severe adverse events potentially related to blood pressure reported in this study, one subject reported moderate vasodilatation after both sildenafil 50 mg and 100 mg. There were no episodes of syncope reported in this study.
Effect ofSildenafilon Blood Pressure When Co-administered with Antihypertensives:When sildenafil 100 mg oral was co-administered with amlodipine, 5 mg or 10 mg oral, to hypertensive patients, the mean additional reduction on supine blood pressure was 8 mmHg systolic and 7 mmHg diastolic.
Effect ofSildenafilon Blood Pressure When Co-administered with Alcohol:Sildenafil 50 mg did not potentiate the hypotensive effect of alcohol (0.5 g/kg) in healthy volunteers with mean maximum blood alcohol levels of 0.08%. The maximum observed decrease in systolic blood pressure was -18.5 mmHg when sildenafil was co-administered with alcohol versus -17.4 mmHg when alcohol was administered alone. The maximum observed decrease in diastolic blood pressure was -17.2 mmHg when sildenafil was co-administered with alcohol versus -11.1 mmHg when alcohol was administered alone. There were no reports of postural dizziness or orthostatic hypotension. The maximum recommended dose of 100 mg sildenafil was not evaluated in this study [seeDrug Interactions (7.5)].
Effects ofSildenafil on Cardiac Parameters:Single oral doses of sildenafil up to 100 mg produced no clinically relevant changes in the ECGs of normal male volunteers.
Studies have produced relevant data on the effects of sildenafil citrate on cardiac output. In one small, open-label, uncontrolled, pilot study, eight patients with stable ischemic heart disease underwent Swan-Ganz catheterization. A total dose of 40 mg sildenafil was administered by four intravenous infusions.
The results from this pilot study are shown in Table 4; the mean resting systolic and diastolic blood pressures decreased by 7% and 10% compared to baseline in these patients. Mean resting values for right atrial pressure, pulmonary artery pressure, pulmonary artery occluded pressure and cardiac output decreased by 28%, 28%, 20% and 7% respectively. Even though this total dosage produced plasma sildenafil concentrations which were approximately 2 to 5 times higher than the mean maximum plasma concentrations following a single oral dose of 100 mg in healthy male volunteers, the hemodynamic response to exercise was preserved in these patients.
Table 4: Hemodynamic Data in Patients with Stable Ischemic Heart Disease after IntravenousAdministration of 40 mg of Sildenafil Means ± SD At rest After 4 minutes of exercise n Baseline
(B2)n Sildenafil
(D1)n Baseline n Sildenafil PAOP (mmHg) 8 8.1 ± 5.1 8 6.5 ± 4.3 8 36.0 ± 13.7 8 27.8 ± 15.3 Mean PAP (mmHg) 8 16.7 ± 4 8 12.1 ± 3.9 8 39.4 ± 12.9 8 31.7 ± 13.2 Mean RAP (mmHg) 7 5.7 ± 3.7 8 4.1 ± 3.7 - - - - Systolic SAP (mmHg) 8 150.4 ± 12.4 8 140.6 ± 16.5 8 199.5 ± 37.4 8 187.8 ± 30.0 Diastolic SAP (mmHg) 8 73.6 ± 7.8 8 65.9 ± 10 8 84.6 ± 9.7 8 79.5 ± 9.4 Cardiac output (L/min) 8 5.6 ± 0.9 8 5.2 ± 1.1 8 11.5 ± 2.4 8 10.2 ± 3.5 Heart rate (bpm) 8 67 ± 11.1 8 66.9 ± 12 8 101.9 ± 11.6 8 99.0 ± 20.4 In a double-blind study, 144 patients with erectile dysfunction and chronic stable angina limited by exercise, not receiving chronic oral nitrates, were randomized to a single dose of placebo or sildenafil 100 mg 1 hour prior to exercise testing. The primary endpoint was time to limiting angina in the evaluable cohort. The mean times (adjusted for baseline) to onset of limiting angina were 423.6 and 403.7 seconds for sildenafil (N=70) and placebo, respectively. These results demonstrated that the effect of sildenafil on the primary endpoint was statistically non-inferior to placebo.
Effects ofSildenafil on Vision:At single oral doses of 100 mg and 200 mg, transient dose-related impairment of color discrimination was detected using the Farnsworth-Munsell 100-hue test, with peak effects near the time of peak plasma levels. This finding is consistent with the inhibition of PDE6, which is involved in phototransduction in the retina. Subjects in the study reported this finding as difficulties in discriminating blue/green. An evaluation of visual function at doses up to twice the maximum recommended dose revealed no effects of sildenafil on visual acuity, intraocular pressure, or pupillometry.
Effects ofSildenafilon Sperm: There was no effect on sperm motility or morphology after single 100 mg oral doses of sildenafil in healthy volunteers.
12.3 Pharmacokinetics
Sildenafil is rapidly absorbed after oral administration, with a mean absolute bioavailability of 41% (range 25-63%). The pharmacokinetics of sildenafil are dose-proportional over the recommended dose range. It is eliminated predominantly by hepatic metabolism (mainly CYP3A4) and is converted to an active metabolite with properties similar to the parent, sildenafil. Both sildenafil and the metabolite have terminal half-lives of about 4 hours.
Figure6: Mean Sildenafil Plasma Concentrations in Healthy Male Volunteers
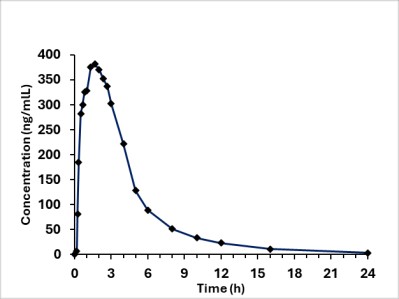
Absorption and Distribution:VYBRIQUE is rapidly absorbed. Maximum observed plasma concentrations are reached within 30 to 300 minutes (median 80 minutes) of oral dosing in the fasted state. After a single dose of 100 mg of VYBRIQUE in adult males, the mean (coefficient of variation, %) peak plasma concentration (Cmax) and area under the concentration curve (AUC) of sildenafil were 503.5 (37.7%) ng/mL and 1902.4 (36.3%) ng·h/mL, respectively.
When VYBRIQUE is taken with a high fat meal, the rate of absorption is reduced, with a mean delay in Tmax of 87 minutes and a mean reduction in Cmax of 45%. AUC is not impacted by food.
In a clinical study in 35 healthy males (18-55 years old) where VYBRIQUE 100 mg was administered in fed conditions with or without water, no clinically significant differences in pharmacokinetics of sildenafil and its major N-desmethyl metabolite was observed.
The mean steady state volume of distribution (Vss) for sildenafil is 105 L, indicating distribution into the tissues. Sildenafil and its major circulating N-desmethyl metabolite are both approximately 96% bound to plasma proteins. Protein binding is independent of total drug concentrations.
Based upon measurements of sildenafil in semen of healthy volunteers 90 minutes after dosing, less than 0.001% of the administered dose may appear in the semen of patients.
Metabolism and Excretion:Sildenafil is cleared predominantly by the CYP3A4 (major route) and CYP2C9 (minor route) hepatic microsomal isoenzymes. The major circulating metabolite results from N-desmethylation of sildenafil and is itself further metabolized. This metabolite has a PDE selectivity profile similar to sildenafil and an in vitropotency for PDE5 approximately 50% of the parent drug. Plasma concentrations of this metabolite are approximately 40% of those seen for sildenafil, so that the metabolite accounts for about 20% of sildenafil’s pharmacologic effects.
After either oral or intravenous administration, sildenafil is excreted as metabolites predominantly in the feces (approximately 80% of administered oral dose) and to a lesser extent in the urine (approximately 13% of the administered oral dose). Similar values for pharmacokinetic parameters were seen in normal volunteers and in the patient population, using a population pharmacokinetic approach.
Pharmacokinetics in Special Populations
Geriatrics:Healthy elderly volunteers (65 years or over) had a reduced clearance of sildenafil, resulting in approximately 84% and 107% higher plasma AUC values of sildenafil and its active N-desmethyl metabolite, respectively, compared to those seen in healthy younger volunteers (18-45 years). Due to age-differences in plasma protein binding, the corresponding increase in the AUC of free (unbound) sildenafil and its active N-desmethyl metabolite were 45% and 57%, respectively [see Dosage and Administration (2.3), and Use in Specific Populations (8.5)]
Renal Impairment:In volunteers with mild (CLcr=50-80 mL/min) and moderate (CLcr=30-49 mL/min) renal impairment, the pharmacokinetics of a single oral dose of sildenafil (50 mg) were not altered. In volunteers with severe (CLcr <30 mL/min) renal impairment, sildenafil clearance was reduced, resulting in approximately doubling of AUC and Cmax compared to age-matched volunteers with no renal impairment [see Dosage and Administration (2.3), and Use in Specific Populations (8.6)].
In addition, N-desmethyl metabolite AUC and Cmax values significantly increased by 200% and 79%, respectively in subjects with severe renal impairment compared to subjects with normal renal function.
Hepatic Impairment:In volunteers with hepatic impairment (Child-Pugh Class A and B), sildenafil clearance was reduced, resulting in increases in AUC (85%) and Cmax (47%) compared to age-matched volunteers with no hepatic impairment. The pharmacokinetics of sildenafil in patients with severely impaired hepatic function (Child-Pugh Class C) have not been studied [see Dosage and Administration (2.3), and Use in Specific Populations (8.7)].
Therefore, age >65, hepatic impairment and severe renal impairment are associated with increased plasma levels of sildenafil. A starting oral dose of 25 mg should be considered in those patients [see Dosage and Administration(2.3)].
Drug Interaction Studies
Effects of Other Drugs onSildenafil
Sildenafil metabolism is principally mediated by CYP3A4 (major route) and CYP2C9 (minor route). Therefore, inhibitors of these isoenzymes may reduce sildenafil clearance and inducers of these isoenzymes may increase sildenafil clearance. The concomitant use of erythromycin or strong CYP3A4 inhibitors (e.g., saquinavir, ketoconazole, itraconazole) as well as the nonspecific CYP inhibitor, cimetidine, is associated with increased plasma levels of sildenafil [see Dosage and Administration (2.2)].
In vivostudies:
Cimetidine (800 mg), a nonspecific CYP inhibitor, caused a 56% increase in plasma sildenafil concentrations when co-administered with sildenafil (50 mg) to healthy volunteers.
When a single 100 mg dose of sildenafil was administered with erythromycin, a moderate CYP3A4 inhibitor, at steady state (500 mg bid for 5 days), there was a 160% increase in sildenafil Cmax and an 182% increase in sildenafil AUC. In addition, in a study performed in healthy male volunteers, co-administration of the HIV protease inhibitor saquinavir, also a CYP3A4 inhibitor, at steady state (1200 mg tid) with sildenafil (100 mg single dose) resulted in a 140% increase in sildenafil Cmax and a 210% increase in sildenafil AUC. Sildenafil had no effect on saquinavir pharmacokinetics. A stronger CYP3A4 inhibitor such as ketoconazole or itraconazole could be expected to have greater effect than that seen with saquinavir. Population pharmacokinetic data from patients in clinical trials also indicated a reduction in sildenafil clearance when it was co-administered with CYP3A4 inhibitors (such as ketoconazole, erythromycin, or cimetidine) [see Dosage and Administration(2.2) and Drug Interactions (7.4)].
In another study in healthy male volunteers, co-administration with the HIV protease inhibitor ritonavir, which is a highly potent P450 inhibitor, at steady state (500 mg bid) with sildenafil (100 mg single dose) resulted in a 300% (4-fold) increase in sildenafil Cmax and a 1000% (11-fold) increase in sildenafil plasma AUC. At 24 hours the plasma levels of sildenafil were still approximately 200 ng/mL, compared to approximately 5 ng/mL when sildenafil was dosed alone. This is consistent with ritonavir’s marked effects on a broad range of P450 substrates. Sildenafil had no effect on ritonavir pharmacokinetics [see Dosage and Administration (2.2) and Drug Interactions (7.4)].
Although the interaction between other protease inhibitors and sildenafil has not been studied, their concomitant use is expected to increase sildenafil levels.
In a study of healthy male volunteers, co-administration of sildenafil at steady state (80 mg t.i.d.) with endothelin receptor antagonist bosentan (a moderate inducer of CYP3A4, CYP2C9 and possibly of CYP2C19) at steady state (125 mg b.i.d.) resulted in a 63% decrease of sildenafil AUC and a 55% decrease in sildenafil Cmax. Concomitant administration of strong CYP3A4 inducers, such as rifampin, is expected to cause greater decreases in plasma levels of sildenafil.
Single doses of antacid (magnesium hydroxide/aluminum hydroxide) did not affect the bioavailability of sildenafil citrate.
In healthy male volunteers, there was no evidence of a clinically significant effect of azithromycin (500 mg daily for 3 days) on the systemic exposure of sildenafil or its major circulating metabolite.
Pharmacokinetic data from patients in clinical trials showed no effect on sildenafil pharmacokinetics of CYP2C9 inhibitors (such as tolbutamide, warfarin), CYP2D6 inhibitors (such as selective serotonin reuptake inhibitors, tricyclic antidepressants), thiazide and related diuretics, ACE inhibitors, and calcium channel blockers. The AUC of the active metabolite, N-desmethyl sildenafil, was increased 62% by loop and potassium-sparing diuretics and 102% by nonspecific beta-blockers. These effects on the metabolite are not expected to be of clinical consequence.
Effects ofSildenafilon Other Drugs
In vitrostudies:
Sildenafil is a weak inhibitor of the CYP isoforms 1A2, 2C9, 2C19, 2D6, 2E1 and 3A4 (IC50 >150 μM). Given sildenafil peak plasma concentrations of approximately 1 μM after recommended doses, it is unlikely that sildenafil will alter the clearance of substrates of these isoenzymes.
In vivo studies:
No significant interactions were shown with tolbutamide (250 mg) or warfarin (40 mg), both of which are metabolized by CYP2C9.
In a study of healthy male volunteers, sildenafil (100 mg) did not affect the steady state pharmacokinetics of the HIV protease inhibitors, saquinavir and ritonavir, both of which are CYP3A4 substrates.
Sildenafil (50 mg) did not potentiate the increase in bleeding time caused by aspirin (150 mg).
Sildenafil at steady state, at a dose not approved for the treatment of erectile dysfunction (80 mg t.i.d.) resulted in a 50% increase in AUC and a 42% increase in Cmax of bosentan (125 mg b.i.d.).
-
13
NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Sildenafil was not carcinogenic when administered to rats for 24 months at a dose resulting in total systemic drug exposure (AUCs) for unbound sildenafil and its major metabolite of 20- and 38- times, for male and female rats, respectively, the exposures observed in human males given the Maximum Recommended Human Dose (MRHD) of 100 mg. Sildenafil was not carcinogenic when administered to mice for 18-21 months at dosages up to the Maximum Tolerated Dose (MTD) of 10 mg/kg/day, approximately 0.4 times the MRHD on a mg/m 2basis in a 50 kg subject.
Mutagenesis
Sildenafil was negative in in vitrobacterial and Chinese hamster ovary cell assays to detect mutagenicity, and invitrohuman lymphocytes and in vivomouse micronucleus assays to detect clastogenicity.
Impairment of Fertility
There was no impairment of fertility in rats given sildenafil up to 60 mg/kg/day for 36 days to females and 102 days to males, a dose producing an AUC value of more than 25 times the human male AUC.
-
14
CLINICAL STUDIES
Efficacy Results inVYBRIQUEClinical Study
The efficacy of VYBRIQUE in the treatment of erectile dysfunction was established in part on the basis of efficacy data from trials with the tablet formulation of sildenafil.
VYBRIQUE was evaluated in one randomized, double-blind, placebo-controlled, flexible-dose study (25 mg, 50 mg, 75 mg, 100 mg) over 12 weeks study to evaluate the erectile function of men with erectile dysfunction (ED). The co-primary efficacy endpoints included both a 4-week assessment of sexual function based on the International Index of Erectile Function (IIEF) questionnaire and an assessment of sexual function after each sexual intercourse based on the Sexual Encounter Profile (SEP) on the daily diary.
- The six-item, 30-point, erectile function (EF) domain of the IIEF was assessed at baseline, at follow-up visits, and at the end of the study reflecting subjects’ sexual experience during the past 4 weeks.
- The SEP Question 2 (“Were you able to insert your penis into your partner’s vagina”) and Question 3 (“Did your erection last long enough for you to have successful intercourse?”) were answered immediately after each sexual attempt with binary response (Yes=success, No=failure).
Sexual function data were recorded by patients in a daily diary.
A total of 475 patients were enrolled and received treatment with VYBRIQUE, including subjects with comorbidities such as diabetes, dyslipidemia, hypertension and obesity. Of these, 354 patients received VYBRIQUE and 121 patients received placebo without regard to food and alcohol concomitant intake. The initial dose was 50 mg for subjects under 65 years and 25 mg for subjects 65 years or older with dose adjustment permitted up to 50 mg (for subjects 65 years or older), 75 mg or 100 mg or down to 25 mg based on efficacy and tolerability. By the end of the study, 94 (26.6%) and 115 (32.5%) patients in the VYBRIQUE group were receiving 75 mg or 100 mg respectively. The efficacy of VYBRIQUE, administered at flexible doses of 25 mg, 50 mg, 75 mg and 100 mg for the treatment of ED, was superior to placebo. Efficacy results are shown in Figure 6. Subgroup analyses by age, race, and ethnicity showed efficacy results consistent with the overall population.
Table 5: Co-Primary Efficacy Results in a Randomized, Double-Blind, Placebo-Controlled, Flexible-Dose Study of VYBRIQUE IIEF-EF
Domain Score
Mean (SD)SEP Q2
Percentage (%) of ‘Yes’ Responses
Mean (SD)SEP Q3
Percentage (%) of ‘Yes’ Responses
Mean (SD)Sildenafil (N=354) Placebo (N=121) Sildenafil (N=354) Placebo (N=121) Sildenafil (N=354) Placebo (N=121) Baseline 12.9 (4.8) 12.7 (4.8) 42.7 (34.3) 44.2 (36.5) 11.5 (17.2) 8.9 (16.0) Week 12 24.3 (6.6) 21.0 (8.7) 82.1 (28.8) 68.6 (36.5) 69.7 (34.2) 49.9 (38.7) Change fromBaseline 11.5 (7.1) 8.2 (8.9) 39.5 (35.3) 24.2 (36.2) 58.2 (34.6) 41.3 (39.6) Sildenafil - Placebo IIEF-EF SEP Q2 SEP Q3 LS Means
[95% CI]3.3*
[1.6, 5.0]13.9%*
[7.9%, 19.9%]19.0%*
[11.8%, 26.3%]*Statistically significant differences vs. placebo; p-value <0.001 IIEF= International Index of Erectile Function, EF = Erectile Function, SEP Q2=Sexual Encounter Profile Question 2, SEP Q3 = Sexual Encounter Profile Question 3, N = Number of Subjects, SD = Standard Deviation, LS = Least Squares, CI = Confidence Interval.
Sildenafil was assessed in many clinical studies for its effect on the ability of men with ED to engage in sexual activity and in many cases specifically on the ability to achieve and maintain an erection sufficient for satisfactory sexual activity. Sildenafil was evaluated primarily at doses of 25 mg, 50 mg and 100 mg in 21 randomized, double-blind, placebo-controlled trials of up to 6 months in duration, using a variety of study designs (fixed dose, titration, parallel, crossover). Sildenafil was administered to more than 3,000 patients aged 19 to 87 years, with ED of various etiologies (organic, psychogenic, mixed) with a mean duration of 5 years. sildenafil citrate demonstrated statistically significant improvement compared to placebo in all 21 studies. The studies that established benefit demonstrated improvements in success rates for sexual intercourse compared with placebo.
Sildenafil Efficacy Results from Controlled Clinical Studies
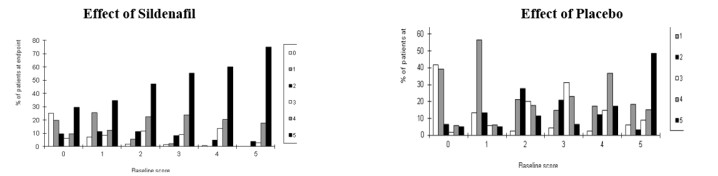
The effect of sildenafil on one of the major end points, maintenance of erections after penetration, is shown in Figure 7, for the pooled results of 5 sildenafil fixed-dose, dose-response studies of greater than one-month duration, showing response according to baseline function. Results with all doses have been pooled, but scores showed greater improvement at the 50 and 100 mg doses than at the 25 mg dose. The pattern of responses was similar for the other principal question, the ability to achieve an erection sufficient for intercourse. The titration studies, in which most patients received 100 mg, showed similar results. Figure 7 shows that regardless of the baseline levels of function, subsequent function in patients treated with sildenafil was better than that seen in patients treated with placebo. At the same time, on-treatment function was better in treated patients who were less impaired at baseline.
Figure7: Effect of Sildenafil and Placebo on Maintenance of Erection by Baseline Score
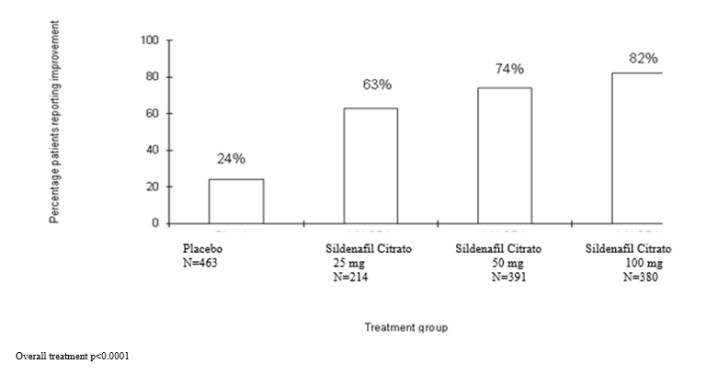
The frequency of patients reporting improvement of erections in response to a global question in four of the sildenafil randomized, double-blind, parallel, placebo-controlled fixed dose studies (1797 patients) of 12 to 24 weeks duration is shown in Figure 8. These patients had erectile dysfunction at baseline that was characterized by median categorical scores of 2 (a few times) on principal IIEF questions. Erectile dysfunction was attributed to organic (58%; generally not characterized, but including diabetes and excluding spinal cord injury), psychogenic (17%), or mixed (24%) etiologies. Sixty-three percent, 74%, and 82% of the patients on 25 mg, 50 mg and 100 mg of sildenafil, respectively, reported an improvement in their erections, compared to 24% on placebo. In the titration studies (n=644) (with most patients eventually receiving 100 mg), results were similar.
Figure8: Percentageof Patients Reportingan Improvement in Erections
The patients in the sildenafil studies had varying degrees of ED. One-third to one-half of the subjects in these studies reported successful intercourse at least once during a 4-week, treatment-free run-in period.
In many of the sildenafil studies, of both fixed dose and titration designs, daily diaries were kept by patients. In these studies, involving about 1600 patients, analyses of patient diaries showed no effect of sildenafil on rates of attempted intercourse (about 2 per week), but there was clear treatment-related improvement in sexual function: per patient weekly success rates averaged 1.3 on 50-100 mg of sildenafil vs 0.4 on placebo; similarly, group mean success rates (total successes divided by total attempts) were about 66% on sildenafil citrate vs about 20% on placebo.
During 3 to 6 months of double-blind treatment or longer-term (1 year), open-label sildenafil studies, few patients withdrew from active treatment for any reason, including lack of effectiveness. At the end of the long-term study, 88% of patients reported that sildenafil improved their erections.
Men with untreated ED had relatively low baseline scores for all aspects of sexual function measured (again using a 5-point scale) in the IIEF. Sildenafil improved these aspects of sexual function: frequency, firmness and maintenance of erections; frequency of orgasm; frequency and level of desire; frequency, satisfaction and enjoyment of intercourse; and overall relationship satisfaction.
One randomized, double-blind, flexible-dose, placebo-controlled sildenafil study included only patients with erectile dysfunction attributed to complications of diabetes mellitus (n=268). As in the other titration studies, patients were started on 50 mg and allowed to adjust the dose up to 100 mg or down to 25 mg of sildenafil; all patients, however, were receiving 50 mg or 100 mg at the end of the study. There were highly statistically significant improvements on the two principal IIEF questions (frequency of successful penetration during sexual activity and maintenance of erections after penetration) on sildenafil compared to placebo. On a global improvement question, 57% of sildenafil patients reported improved erections versus 10% on placebo. Diary data indicated that on sildenafil, 48% of intercourse attempts were successful versus 12% on placebo.
One randomized, double-blind, placebo-controlled, crossover, flexible-dose (up to 100 mg) sildenafil study of patients with erectile dysfunction resulting from spinal cord injury (n=178) was conducted. The changes from baseline in scoring on the two endpoint questions (frequency of successful penetration during sexual activity and maintenance of erections after penetration) were highly statistically significantly in favor of sildenafil. On a global improvement question, 83% of patients reported improved erections on sildenafil versus 12% on placebo. Diary data indicated that 59% of attempts at sexual intercourse were successful on sildenafil compared to 13% on placebo. Across all trials, sildenafil improved the erections of 43% of radical prostatectomy patients compared to 15% on placebo.
Subgroup analyses of responses to a global improvement question in patients with psychogenic etiology in two fixed-dose sildenafil studies (total n=179) and two sildenafil titration studies (total n=149) showed 84% of sildenafil patients reported improvement in erections compared with 26% of placebo. The changes from baseline in scoring on the two endpoint questions (frequency of successful penetration during sexual activity and maintenance of erections after penetration) were highly statistically significantly in favor of sildenafil .Diary data in two of the studies (n=178) showed rates of successful intercourse per attempt of 70% for sildenafil and 29% for placebo.
Efficacy Results in Subpopulations in Controlled Clinical Studies
A review of population subgroups in sildenafil studies demonstrated efficacy regardless of baseline severity, etiology, race and age. Sildenafil was effective in a broad range of ED patients, including those with a history of coronary artery disease, hypertension, other cardiac disease, peripheral vascular disease, diabetes mellitus, depression, coronary artery bypass graft (CABG), radical prostatectomy, transurethral resection of the prostate (TURP) and spinal cord injury, and in patients taking antidepressants/antipsychotics and antihypertensives/diuretics.
- The six-item, 30-point, erectile function (EF) domain of the IIEF was assessed at baseline, at follow-up visits, and at the end of the study reflecting subjects’ sexual experience during the past 4 weeks.
-
16
HOW SUPPLIED/STORAGE AND HANDLING
VYBRIQUE is supplied as oral films in individually sealed foil pouches in four dosage strengths. Each carton contains 4 or 8 pouches. The product is an opaque light blue, thin, flexible oral film with film imprint code, and characteristic lemon and grapefruit flavor with the following dimensions:
Strengths(mg) of sildenafil FilmImprint Code Film Dimensions (shape) NDC forCarton of4 Pouches
NDC forCarton of8 Pouches
25 mg S25 30 mm x 15 mm (rectangular) 71858-0205-5 71858-0205-6 50 mg S50 30 mm x 30 mm
(square)71858-0210-5 71858-0210-6 75 mg S75 30 mm x 45 mm (rectangular) 71858-0215-5 71858-0215-6 100 mg S100 45 mm x 40 mm (rectangular) 71858-0220-5 71858-0220-6 Recommended Storage:Store at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
-
17
PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling ( Patient Information)
Administration Instructions
Advise patients to place VYBRIQUE directly onto the tongue where it will disintegrate and can then be swallowed with saliva without the need for water or other liquids. Advise patients not to cut or chew VYBRIQUE [seeDosage and Administration (2.4) ].
Nitrates
Physicians should discuss with patients the contraindication of VYBRIQUE with regular and/or intermittent use of nitric oxide donors, such as organic nitrates or organic nitrites in any form [see Contraindications (4.1)].
Guanylate Cyclase(GC) Stimulators
Physicians should discuss with patients the contraindication of VYBRIQUE with use of guanylate cyclase stimulators such as riociguat [see Contraindications (4.3)].
Concomitant Use with Drugs Which Lower Blood Pressure
Physicians should advise patients of the potential for VYBRIQUE to augment the blood pressure lowering effect of alpha-blockers and antihypertensive medications. Concomitant administration of VYBRIQUE and an alpha blocker may lead to symptomatic hypotension in some patients. Therefore, when VYBRIQUE is co-administered with alpha-blockers, patients should be stable on alpha-blocker therapy prior to initiating VYBRIQUE treatment and VYBRIQUE should be initiated at the lowest dose [see Warnings and Precautions (5.5)].
CardiovascularRiskConsiderations
Physicians should discuss with patients the potential cardiac risk of sexual activity in patients with preexisting cardiovascular risk factors. Patients who experience symptoms (e.g., angina pectoris, dizziness, nausea) upon initiation of sexual activity should be advised to refrain from further activity and should discuss the episode with their physician [see Warnings and Precautions (5.1)].
Sudden Loss of Vision
Physicians should advise patients to stop use of all PDE5 inhibitors, including VYBRIQUE and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision including possible permanent loss of vision, that has been reported rarely post-marketing in temporal association with the use of all PDE5 inhibitors, including sildenafil. Physicians should discuss with patients the increased risk of NAION in individuals who have already experienced NAION in one eye. Physicians should also discuss with patients the increased risk of NAION among the general population in patients with a “crowded” optic disc, although evidence is insufficient to support screening of prospective users of PDE5 inhibitor, including VYBRIQUE, for this uncommon condition [see Warnings and Precautions (5.3),Adverse Reactions (6.2)].
Sudden Hearing Loss
Physicians should advise patients to stop taking PDE5 inhibitors, including VYBRIQUE and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including VYBRIQUE. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Warnings and Precautions (5.4),Adverse Reactions (6.2)].
Priapism
Physicians should warn patients that prolonged erections greater than 4 hours and priapism (painful erections greater than 6 hours in duration) have been reported infrequently since market approval of sildenafil. In the event of an erection that persists longer than 4 hours, the patient should seek immediate medical assistance. If priapism is not treated immediately, penile tissue damage and permanent loss of potency may result [see Warnings and Precautions (5.2)].
Avoid Use with other PDE5 Inhibitors
Physicians should inform patients not to take VYBRIQUE with other PDE5 inhibitors, including pulmonary arterial hypertension (PAH) treatments containing sildenafil. The safety and efficacy of VYBRIQUE with other PDE5 inhibitors have not been studied [see Warnings and Precautions (5.7)].
Sexually Transmitted Disease
The use of VYBRIQUE offers no protection against sexually transmitted diseases. Counseling of patients about the protective measures necessary to guard against sexually transmitted diseases, including the Human Immunodeficiency Virus (HIV), should be considered [see Warnings and Precautions (5.9)].
Distributed by:
IBSA Pharma Inc,
Parsippany, NJ 07054 USA -
PATIENT PACKAGE INSERT
Patient Information
VYBRIQUETM (vī-BRĒK)
(sildenafil)
Oral FilmWhat is the most important information I should know aboutVYBRIQUE?
VYBRIQUEcan cause your blood pressure to drop suddenly to an unsafe level if it is taken with certain other medicines.Do not take VYBRIQUE if you take any other medicines called nitrates. Nitrates are used to treat chest pain (angina). A sudden drop in blood pressure can cause you to feel dizzy, faint, or have a heart attack or stroke.
Do not take VYBRIQUE if you take medicines called guanylate cyclase stimulators which include:
- Riociguat a medicine that treats pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension.
Stop sexual activity and get medical help right away if you get symptoms such as chest pain, dizziness, or nausea during sex. Sexual activity can put an extra strain on your heart, especially if your heart is already weak from a heart attack or heart disease. VYBRIQUE does not protect you or your partner from getting sexually transmitted diseases, including Human Immunodeficiency Virus (HIV).What isVYBRIQUE?
VYBRIQUE is a prescription medicine used to treat erectile dysfunction (ED).
VYBRIQUE is not for use in women or children.
It is not known if VYBRIQUE is safe and effective in women or children under 18 years of age.Do not takeVYBRIQUE if you: - take medicines called nitrates.
- take any medicines called guanylate cyclase stimulators.
- are allergic to sildenafil or any of the ingredients in VYBRIQUE. See the end of this Patient information leaflet for a complete list of ingredients in VYBRIQUE.
Before taking VYBRIQUE, tell your healthcare provider about all of your medical conditions, including if you:
- have or have had heart problems such as a heart attack, irregular heartbeat, angina, chest pain, narrowing of the aortic valve or heart failure.
- have had heart surgery within the last 6 months.
- have pulmonary hypertension.
- have had a stroke.
- have low blood pressure, or high blood pressure that is not controlled.
- have a deformed penis shape.
- have had an erection that lasted for more than 4 hours.
- have problems with your blood cells such as sickle cell anemia, multiple myeloma, or leukemia.
- have retinitis pigmentosa, a rare genetic (runs in families) eye disease.
- have ever had severe vision loss, including an eye problem called non-arteritic anterior ischemic optic neuropathy (NAION).
- have bleeding problems.
- have or have had stomach ulcers.
- have liver problems.
- have kidney problems or are having kidney dialysis.
VYBRIQUE may affect the way other medicines work, and other medicines may affect the way VYBRIQUE works causing side effects. Especially tell your healthcare provider if you take any of the following:
- medicines called nitrates (see
“What is the most important information I should know aboutVYBRIQUE?”)
- medicines called guanylate cyclase stimulators, such as riociguat (see “
What is the most important information I should know about VYBRIQUE?”)
- medicines called alpha blockers such as doxazosin. Alpha-blockers are sometimes prescribed for prostate problems or high blood pressure. In some patients, the use of VYBRIQUE with alpha-blockers can lead to a drop-in blood pressure or to fainting.
- medicines called HIV protease inhibitors, such as ritonavir and saquinavir.
- some types of oral antifungal medicines, such as ketoconazole and itraconazole.
- some types of antibiotics, such as erythromycin.
- other medicines that treat high blood pressure.
- other medicines or treatments for ED.
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine.How should I takeVYBRIQUE?
- Take VYBRIQUE exactly as your healthcare provider tells you to take it.
- Your healthcare provider will tell you how much VYBRIQUE to take and when to take it.
- Your healthcare provider may change your dose if needed.
- Take VYBRIQUE by mouth about 1 hour before sexual activity. You may take VYBRIQUE between 30 minutes to 4 hours before sexual activity if needed.
- VYBRIQUE can be taken with or without food.
- Do not cut or chew VYBRIQUE.
-
Do nottake VYBRIQUE more than 1 time a day.
- If you take too much VYBRIQUE, call your healthcare provider or go to the nearest hospital emergency room right away.
Important information you need to know before using VYBRIQUE- Do not remove the film from the pouch unless ready for immediate use.
- Do not handle the oral film with wet hands.
a) Take the pouch and locate the notch on the side of the pouch. 
b) Gently tear along the dotted line to open the pouch. 
c) Gently peel both parts of the pouch. Now you can hold each side of the pouch between your thumb and your index finger. 
d) Take the oral film with dry fingers out of the pouch and put it onto your tongue. It will dissolve in your mouth and can be swallowed with saliva. 
What are the possible side effects ofVYBRIQUE?
VYBRIQUE can cause serious side effects including:
- See
“What is the most important information I should know about VYBRIQUE?”
-
an erection that will not go away (priapism).If you have an erection that lasts more than 4 hours, get medical help right away. If it is not treated right away, priapism can permanently damage your penis.
-
sudden vision loss in one or both eyes.Sudden vision loss in 1 or both eyes can be a sign of a serious eye problem called non-arteritic anterior ischemic optic neuropathy (NAION). It is uncertain whether PDE5 inhibitors directly cause the vision loss. Stop taking VYBRIQUE and call your healthcare provider right away if you have sudden vision loss in 1 or both eyes.
- sudden hearing decrease or hearing loss.Some people may also have ringing in their ears (tinnitus) or dizziness. If you have these symptoms, stop taking VYBRIQUE and contact a healthcare provider right away.
- headache
- flushing
- upset stomach
- stuffy or runny nose
- dizziness
- abnormal vision, such as changes in color and blurred vision
- back pain
- muscle pain
- nausea
- rash
These are not all of the possible side effects of VYBRIQUE. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I storeVYBRIQUE?
- Store VYBRIQUE at room temperature between 68°F to 77°F (20°C to 25°C).
General information about the safe and effective use ofVYBRIQUE.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use VYBRIQUE for a condition for which it was not prescribed. Do not give VYBRIQUE to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about VYBRIQUE that is written for health professionals.What are the ingredients inVYBRIQUE?
Active ingredient:sildenafil
Inactive ingredients: Blue Videojet ink, FD&C Blue No.2, glycerin, grapefruit flavor, lemon flavor, maltodextrin, polysorbate 20, polyvinyl acetate dispersion, propylene glycol monocaprylate, sucralose, titanium dioxide.
Contains no ingredient made from a gluten-containing grain (wheat, barley or rye).
Manufactured by: ALTERGON Italia Srl, via Zona Industriale, Morra de Sanctis, Avellino 83040, Italy
Manufactured for: IBSA Institut Biochimique SA, CH-6912 Pazzallo, Switzerland
Distributed by: IBSA Pharma Inc, Parsippany, NJ 07054 USAThis Patient Information has been approved by the U.S. Food and Drug Administration Approved: 12/2025
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
VYBRIQUE
sildenafil citrate filmProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71858-0205 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SILDENAFIL CITRATE (UNII: BW9B0ZE037) (SILDENAFIL - UNII:3M7OB98Y7H) SILDENAFIL 25 mg Inactive Ingredients Ingredient Name Strength MALTODEXTRIN (UNII: 7CVR7L4A2D) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL MONOCAPRYLATE (UNII: RT9P9S09QI) SUCRALOSE (UNII: 96K6UQ3ZD4) POLYSORBATE 20 (UNII: 7T1F30V5YH) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) Product Characteristics Color Score Shape Size Flavor LEMON, GRAPEFRUIT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71858-0205-2 1 in 1 POUCH; Type 0: Not a Combination Product 02/15/2026 2 NDC: 71858-0205-4 1 in 1 POUCH; Type 0: Not a Combination Product 02/15/2026 3 NDC: 71858-0205-5 4 in 1 CARTON 02/15/2026 3 1 in 1 POUCH; Type 0: Not a Combination Product 4 NDC: 71858-0205-6 8 in 1 CARTON 02/15/2026 4 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA210858 02/15/2026 VYBRIQUE
sildenafil citrate filmProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71858-0210 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SILDENAFIL CITRATE (UNII: BW9B0ZE037) (SILDENAFIL - UNII:3M7OB98Y7H) SILDENAFIL 50 mg Inactive Ingredients Ingredient Name Strength MALTODEXTRIN (UNII: 7CVR7L4A2D) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL MONOCAPRYLATE (UNII: RT9P9S09QI) SUCRALOSE (UNII: 96K6UQ3ZD4) POLYSORBATE 20 (UNII: 7T1F30V5YH) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) Product Characteristics Color Score Shape Size Flavor LEMON, GRAPEFRUIT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71858-0210-1 2 in 1 CARTON 02/15/2026 1 1 in 1 POUCH; Type 0: Not a Combination Product 2 NDC: 71858-0210-2 1 in 1 POUCH; Type 0: Not a Combination Product 02/15/2026 3 NDC: 71858-0210-4 1 in 1 POUCH; Type 0: Not a Combination Product 02/15/2026 4 NDC: 71858-0210-5 4 in 1 CARTON 02/15/2026 4 1 in 1 POUCH; Type 0: Not a Combination Product 5 NDC: 71858-0210-6 8 in 1 CARTON 02/15/2026 5 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA210858 02/15/2026 VYBRIQUE
sildenafil citrate filmProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71858-0215 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SILDENAFIL CITRATE (UNII: BW9B0ZE037) (SILDENAFIL - UNII:3M7OB98Y7H) SILDENAFIL 75 mg Inactive Ingredients Ingredient Name Strength MALTODEXTRIN (UNII: 7CVR7L4A2D) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL MONOCAPRYLATE (UNII: RT9P9S09QI) SUCRALOSE (UNII: 96K6UQ3ZD4) POLYSORBATE 20 (UNII: 7T1F30V5YH) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) Product Characteristics Color Score Shape Size Flavor LEMON, GRAPEFRUIT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71858-0215-1 2 in 1 CARTON 02/15/2026 1 1 in 1 POUCH; Type 0: Not a Combination Product 2 NDC: 71858-0215-2 1 in 1 POUCH; Type 0: Not a Combination Product 02/15/2026 3 NDC: 71858-0215-4 1 in 1 POUCH; Type 0: Not a Combination Product 02/15/2026 4 NDC: 71858-0215-5 4 in 1 CARTON 02/15/2026 4 1 in 1 POUCH; Type 0: Not a Combination Product 5 NDC: 71858-0215-6 8 in 1 CARTON 02/15/2026 5 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA210858 02/15/2026 VYBRIQUE
sildenafil citrate filmProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71858-0220 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SILDENAFIL CITRATE (UNII: BW9B0ZE037) (SILDENAFIL - UNII:3M7OB98Y7H) SILDENAFIL 100 mg Inactive Ingredients Ingredient Name Strength MALTODEXTRIN (UNII: 7CVR7L4A2D) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL MONOCAPRYLATE (UNII: RT9P9S09QI) SUCRALOSE (UNII: 96K6UQ3ZD4) POLYSORBATE 20 (UNII: 7T1F30V5YH) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) Product Characteristics Color Score Shape Size Flavor LEMON, GRAPEFRUIT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71858-0220-1 2 in 1 CARTON 02/15/2026 1 1 in 1 POUCH; Type 0: Not a Combination Product 2 NDC: 71858-0220-2 1 in 1 POUCH; Type 0: Not a Combination Product 02/15/2026 3 NDC: 71858-0220-4 1 in 1 POUCH; Type 0: Not a Combination Product 02/15/2026 4 NDC: 71858-0220-5 4 in 1 CARTON 02/15/2026 4 1 in 1 POUCH; Type 0: Not a Combination Product 5 NDC: 71858-0220-6 8 in 1 CARTON 02/15/2026 5 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA210858 02/15/2026 Labeler - IBSA Pharma Inc. (081215551)
Trademark Results [Vybrique]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 VYBRIQUE 97486068 not registered Live/Pending |
IBSA INSTITUT BIOCHIMIQUE SA 2022-07-01 |
 VYBRIQUE 88346833 not registered Live/Pending |
IBSA INSTITUT BIOCHIMIQUE SA 2019-03-19 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.