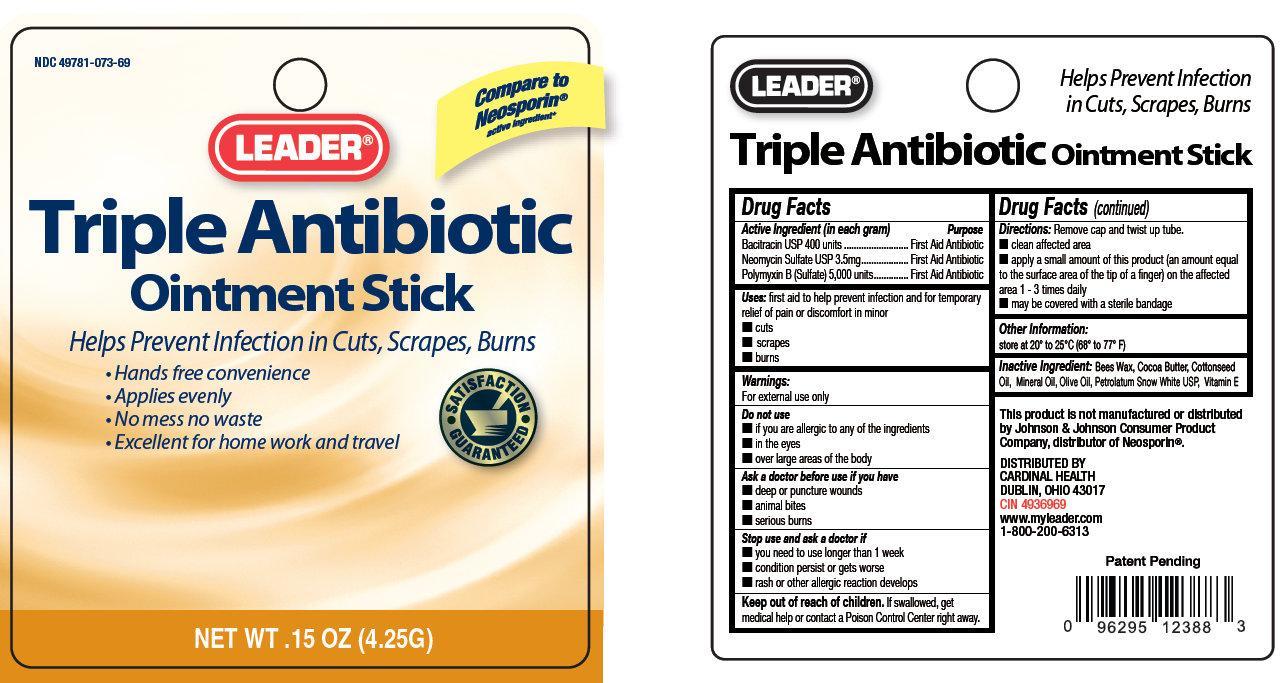
LEADER Triple Antibiotic Ointment Stick
LEADER Triple Antibiotic by
Drug Labeling and Warnings
LEADER Triple Antibiotic by is a Otc medication manufactured, distributed, or labeled by Cardinal Health, Filltech USA, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
LEADER TRIPLE ANTIBIOTIC- bacitracin zinc, neomycin sulfate, polymyxin b sulfate stick
Cardinal Health
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
LEADER Triple Antibiotic Ointment Stick
Active ingredient (in each gram)
Bacitracin USP 400 units
Neomycin Sulfate USP 3.5 mg
Polymyxin B (Sulfate) 5,000 units
Uses:
first aid to help prevent infection and for temporary relief of pain or discomfort in minor
- cuts
- scrapes
- burns
Warnings
For external use only
Directions:
- clean the affected area
- apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the affected area 1-3 times daily
- may be covered with a sterile bandage
| LEADER TRIPLE ANTIBIOTIC
bacitracin zinc, neomycin sulfate, polymyxin b sulfate stick |
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
| Labeler - Cardinal Health (097537435) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Filltech USA, LLC | 926433855 | manufacture(49781-073) | |
Revised: 11/2019
Document Id: 982f3baf-4a58-a7c4-e053-2995a90a1553
Set id: 92834fdb-7713-46b2-85d0-38fb7c2c6955
Version: 5
Effective Time: 20191125
Cardinal Health