Tobramycin by Lifestar Pharma LLC / Mankind Pharma Limited TOBRAMYCIN solution
Tobramycin by
Drug Labeling and Warnings
Tobramycin by is a Prescription medication manufactured, distributed, or labeled by Lifestar Pharma LLC, Mankind Pharma Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use TOBRAMYCIN INHALATION SOLUTION safely and effectively. See full prescribing information for TOBRAMYCIN INHALATION SOLUTION.
TOBRAMYCIN inhalation solution for oral inhalation use
Initial U.S. Approval: 1980
RECENT MAJOR CHANGES
Warnings and Precautions, Ototoxicity (5.1) 2/2023
INDICATIONS AND USAGE
Tobramycin inhalation solution is an inhaled aminoglycoside antibacterial indicated for the management of cystic fibrosis patients with Pseudomonas aeruginosa. (1) Safety and efficacy have not been demonstrated in patients under the age of six years, patients with a forced expiratory volume in one second (FEV1) less than 40% or greater than 80% predicted, or patients colonized with Burkholderia cepacia. (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Inhalation Solution: 300 mg tobramycin per 4 mL solution in a single-dose ampule. (16)
CONTRAINDICATIONS
Tobramycin inhalation solution is contraindicated in patients with a known hypersensitivity to any aminoglycoside. (4)
WARNINGS AND PRECAUTIONS
- Caution should be exercised when prescribing tobramycin inhalation solution to patients with known or suspected auditory, vestibular, renal, or neuromuscular dysfunction. (5.1, 5.2, 5.3 and 5.5)
- Aminoglycoside may aggravate muscle weakness because of a potential curare-like effect on neuromuscular function. (5.3)
- Bronchospasm can occur with inhalation of tobramycin inhalation solution. (5.4)
- Audiograms, serum concentration, and renal function should be monitored as appropriate. (5.2 and 5.5)
- Fetal harm can occur when aminoglycosides are administered to a pregnant woman. Apprise women of the potential hazard to the fetus. (5.6)
ADVERSE REACTIONS
Common adverse reactions (more than 5%) occurring more frequently in tobramycin inhalation solution patients are forced expiratory volume decreased, rales, red blood cell sedimentation rate increased, and dysphonia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Lifestar Pharma LLC at 1-888-995-4337 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Concurrent and/or sequential use of tobramycin inhalation solution with other drugs with neurotoxic, nephrotoxic or ototoxic potential should be avoided. (7.1)
- Tobramycin inhalation solution should not be administered concomitantly with ethacrynic acid, furosemide, urea, or intravenous mannitol. (7.2)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
2.2 Administration Instructions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Ototoxicity
5.2 Nephrotoxicity
5.3 Neuromuscular Disorders
5.4 Bronchospasm
5.5 Laboratory Tests
5.6 Embryo-Fetal Toxicity
5.7 Concomitant Use of Systemic Aminoglycosides
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Drugs with Neurotoxic, Nephrotoxic, or Ototoxic Potential
7.2 Diuretics
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
17.1 Ototoxicity
17.2 Bronchospasm
17.3 Risks Associated with Aminoglycosides
17.4 Laboratory Tests
17.5 Embryo-Fetal Toxicity
17.6 Administration
17.7 Storage Instructions
Instructions for Use
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
Tobramycin inhalation solution is indicated for the management of cystic fibrosis patients with Pseudomonas aeruginosa. Safety and efficacy have not been demonstrated in patients under the age of six years, patients with FEV1 less than 40% or greater than 80% predicted, or patients colonized with Burkholderia cepacia [see Clinical Studies (14)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
Tobramycin inhalation solution is for oral inhalation only [see Dosage and Administration (2.2)]. The recommended dosage of tobramycin inhalation solution for patients six years of age and older is to administer one single-dose ampule (300 mg/4 mL) twice daily by oral inhalation in repeated cycles of 28 days on drug, followed by 28 days off drug. The doses should be taken as close to 12 hours apart as possible and not less than 6 hours apart.
The 300 mg/4 mL dose of tobramycin inhalation solution is the same for patients regardless of age or weight. Tobramycin inhalation solution has not been studied in patients less than six years old.
If patients miss a dose, they should take it as soon as possible anytime up to 6 hours prior to their next scheduled dose. If less than 6 hours remain before the next dose, wait until their next scheduled dose.
2.2 Administration Instructions
Tobramycin inhalation solution is administered by oral inhalation using a hand-held PARI LC PLUS Reusable Nebulizer with a PARI Vios Air compressor over an approximately 15 minute period and until sputtering from the output of the nebulizer has occurred for at least one minute. Tobramycin inhalation solution should not be diluted or mixed with dornase alfa or other medications in the nebulizer. Tobramycin inhalation solution is not for subcutaneous, intravenous, or intrathecal administration.
Further patient instructions on how to administer tobramycin inhalation solution are provided in the Patient's Instructions for Use [see Patient Counseling Information (17)].
Tobramycin inhalation solution should not be used if it is cloudy, if there are particles in the solution, or if it has been stored at room temperature for more than 28 days.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Ototoxicity
Ototoxicity with use of tobramycin inhalation solution
Caution should be exercised when prescribing tobramycin inhalation solution to patients with known or suspected auditory or vestibular dysfunction.
Findings related to ototoxicity as measured by audiometric evaluations and auditory adverse event reports were similar between tobramycin inhalation solution and placebo in controlled clinical trials. Hearing loss was reported in two (1.1%) tobramycin inhalation solution-treated patients and in one (0.9%) placebo-treated patient during clinical studies. Additionally, dizziness and vertigo, both of which may be manifestations of vestibular forms of ototoxicity, were observed in similar numbers of tobramycin inhalation solution- and placebo-treated patients. Dizziness occurred in two (1.1%) tobramycin inhalation solution-treated patients and one (0.9%) placebo-treated patient and vertigo occurred in two (1.1%) tobramycin inhalation solution-treated patients versus no placebo patients in clinical studies. None of the tobramycin inhalation solution patients discontinued their therapy due to hearing loss, dizziness or vertigo.
Tinnitus may be a sentinel symptom of ototoxicity. No reports of tinnitus occurred in patients during clinical studies with tobramycin inhalation solution, but because it has been observed with inhaled tobramycin solutions [see Adverse Reactions (6.2)], onset of this symptom warrants caution. Ototoxicity, manifested as both auditory and vestibular toxicity, has been reported with parenteral aminoglycosides. Vestibular toxicity may be manifested by vertigo, ataxia or dizziness. Patients with known or suspected auditory or vestibular dysfunction should be closely monitored when taking tobramycin inhalation solution. Monitoring may include obtaining audiometric evaluations and serum tobramycin levels. If ototoxicity is noted, the patient should be managed as medically appropriate, including potentially discontinuing tobramycin inhalation solution.
Risk of Ototoxicity Due to Mitochondrial DNA Variants
Cases of ototoxicity with aminoglycosides have been observed in patients with certain variants in the mitochondrially encoded 12S rRNA gene (MT-RNR1), particularly the m.1555A>G variant. Ototoxicity occurred in some patients even when their aminoglycoside serum levels were within the recommended range. Mitochondrial DNA variants are present in less than 1% of the general US population, and the proportion of the variant carriers who may develop ototoxicity as well as the severity of ototoxicity is unknown. In case of known maternal history of ototoxicity due to aminoglycoside use or a known mitochondrial DNA variant in the patient, consider alternative treatments other than aminoglycosides unless the increased risk of permanent hearing loss is outweighed by the severity of infection and lack of safe and effective alternative therapies.
5.2 Nephrotoxicity
Caution should be exercised when prescribing tobramycin inhalation solution to patients with known or suspected renal dysfunction.
Nephrotoxicity was not seen during tobramycin inhalation solution clinical studies but has been associated with aminoglycosides as a class. Patients with known or suspected renal dysfunction or taking concomitant nephrotoxic drugs along with tobramycin inhalation solution should have serum concentrations of tobramycin and laboratory measurements of renal function obtained at the discretion of the treating physician. If nephrotoxicity develops, the patient should be managed as medically appropriate, including potentially discontinuing tobramycin inhalation solution until serum concentrations fall below 2 mcg/mL.
Twenty-six (14%) tobramycin inhalation solution patients and 15 (13%) placebo patients had increases in serum creatinine of at least 50% over baseline. Follow-up values were obtained for 17 of the 26 tobramycin inhalation solution patients, all of which decreased to serum creatinine values that were within normal laboratory ranges. Patients who experience an increase in serum creatinine during treatment with tobramycin inhalation solution should have their renal function closely monitored.
5.3 Neuromuscular Disorders
Tobramycin inhalation solution should be used cautiously in patients with muscular disorders.
Aminoglycosides, including tobramycin, may aggravate muscle weakness because of a potential curare-like effect on neuromuscular function. Neuromuscular blockade, respiratory failure, and prolonged respiratory paralysis may occur more commonly in patients with underlying neuromuscular disorders, such as myasthenia gravis or Parkinson's disease. Prolonged respiratory paralysis may also occur in patients receiving concomitant neuromuscular blocking agents. If neuromuscular blockade occurs, it may be reversed by the administration of calcium salts but mechanical assistance may be necessary.
5.4 Bronchospasm
Bronchospasm has been reported with inhalation of tobramycin. In clinical studies with tobramycin inhalation solution, bronchospasm was observed in one (0.5%) tobramycin inhalation solution-treated patient and in no placebo-treated patients. Wheezing occurred in ten (5%) tobramycin inhalation solution-treated patients and four (4%) placebo-treated patients. Bronchospasm and wheezing should be treated as medically appropriate.
5.5 Laboratory Tests
Clinical studies of inhaled tobramycin solutions did not identify hearing loss using audiometric tests which evaluated hearing up to 8000 Hz. Physicians should consider an audiogram for patients who show any evidence of auditory dysfunction, or who are at increased risk for auditory dysfunction. Tinnitus may be a sentinel symptom of ototoxicity, and therefore the onset of this symptom warrants caution.
Serum Concentrations
In patients with normal renal function treated with tobramycin inhalation solution, serum tobramycin concentrations range from approximately 0.06-1.89 mcg/mL one hour after dose administration and do not require routine monitoring. Serum concentrations of tobramycin in patients with renal dysfunction or patients treated with concomitant parenteral tobramycin should be monitored at the discretion of the treating physician [see Clinical Pharmacology (12.3)].
The serum concentration of tobramycin should only be monitored through venipuncture and not finger prick blood sampling. Contamination of the skin of the fingers with tobramycin may lead to falsely increased measurements of serum levels of the drug. This contamination cannot be completely avoided by hand washing before testing.
Renal Function
The clinical studies of tobramycin inhalation solution did not reveal any imbalance in the percentage of patients who experienced at least a 50% rise in serum creatinine from baseline in either the tobramycin inhalation solution group (n=26, 14%) or the placebo group (n=15, 13%). Laboratory tests of urine and renal function should be conducted at the discretion of the treating physician.
5.6 Embryo-Fetal Toxicity
Aminoglycosides can cause fetal harm when administered to a pregnant woman. Aminoglycosides cross the placenta, and streptomycin has been associated with several reports of total, irreversible, bilateral congenital deafness in pediatric patients exposed in utero. However, systemic absorption of tobramycin following inhaled administration is expected to be minimal [see Clinical Pharmacology (12.3)]. Patients who use tobramycin inhalation solution during pregnancy, or become pregnant while taking tobramycin inhalation solution should be apprised of the potential hazard to the fetus [see Use in Specific Populations (8.1)].
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of drugs cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to tobramycin inhalation solution in two placebo-controlled studies in 305 cystic fibrosis patients. Patients receiving tobramycin inhalation solution ranged in age from 6 to 31 years.
In Study 1, an eight week study, 29 patients received tobramycin inhalation solution versus 30 patients who received placebo for a total of four weeks on drug and four weeks off drug. All patients were ≤ 30 years of age (mean age 12.6 years) and 46% were females. 52.5% of patients were 6 to 12 years of age while 30.5% of patients were 13-17 years old. Only 16.5% of patients were adults (> 17 years old). Eighty percent (80%) of patients were chronically colonized with Pseudomonas aeruginosa while 20.3% of patients were initially or intermittently colonized with Pseudomonas aeruginosa during the study.
More patients in the placebo group discontinued/dropped out of Study 1 than in the tobramycin inhalation solution group (23% [7/30] vs 3.4% [1/29], respectively). Five patients in the placebo group compared to none in the tobramycin inhalation solution group discontinued/dropped out because of treatment-emergent adverse events (TEAEs) such as pulmonary exacerbations and respiratory disorders.
In Study 2, a 24 week study, 161 patients received tobramycin inhalation solution versus 85 patients who received placebo in alternating four week on-off cycles for three cycles. All patients were ≤ 46 years of age (mean age 14.8 years) and 45% were females. 41% of patients were 6-12 years old while 29% of patients were 13-17 years old. Only 30% were adults (>17 years). Eighty-seven percent (87%) of patients were chronically colonized with P. aeruginosa. Only 13% were either initially or intermittently colonized with P. aeruginosa during the study.
More patients in the placebo group discontinued/dropped out of Study 2 than in the tobramycin inhalation solution group (9.4% [8/85] vs 4.3% [7/161], respectively). Of these, 3 patients in the tobramycin inhalation solution group (1.9%) compared to 2 patients in the placebo group (2.4%) withdrew due to a TEAE. The most common TEAEs causing patients to discontinue from the study drug are respiratory, thoracic, and mediastinal disorders.
The most common adverse experiences reported were respiratory disorders, consistent with the underlying disease in the patient population being evaluated and these were similarly distributed between both tobramycin inhalation solution- and placebo-treated patients. The following adverse reactions were reported in at least 5% of tobramycin inhalation solution -treated patients and at rates ≥ 2% more common compared to the placebo-treated patients: decreased forced expiratory volume, rales, red blood cell sedimentation rate increased, and dysphonia (Table 1).
Table 1: Patients with Selected Treatment-Emergent Adverse Reactions Occurring in ≥ 2% of Tobramycin Inhalation Solution Patients
Adverse Reactions
Tobramycin Inhalation Solution N=190
(%)
Placebo N=115
(%)
Forced expiratory volume decreased
59 (31%)
33 (29%)
Rales
36 (19%)
18 (16%)
Red blood cell sedimentation rate increased
16 (8%)
6 (5%)
Dysphonia
11 (6%)
2 (2%)
Wheezing
10 (5%)
4 (4%)
Epistaxis
6 (3%)
0
Pharyngolaryngeal pain
5 (3%)
2 (2%)
Bronchitis
5 (3%)
1 (1%)
Tonsillitis
4 (2%)
0
Diarrhea
3 (2%)
1 (1%)
Eosinophilia
3 (2%)
0
Immunoglobulins increased
3 (2%)
0
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of tobramycin inhalation solution. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Ear and labyrinth disorders: Hearing loss, Tinnitus [see Warnings and Precautions (5.1)]
Skin and subcutaneous tissue disorders: Hypersensitivity, pruritus, urticaria, rash
Nervous system disorders: Aphonia, dysgeusia
Respiratory, thoracic, and mediastinal disorders: Bronchospasm [see Warnings and Precautions (5.4)], oropharyngeal pain
Metabolism and Nutrition Disorders: Decreased appetite
-
7 DRUG INTERACTIONS
7.1 Drugs with Neurotoxic, Nephrotoxic, or Ototoxic Potential
Concurrent and/or sequential use of tobramycin inhalation solution with other drugs with neurotoxic, nephrotoxic, or ototoxic potential should be avoided.
7.2 Diuretics
Some diuretics can enhance aminoglycoside toxicity by altering antibiotic concentrations in serum and tissue. Therefore, tobramycin inhalation solution should not be administered concomitantly with ethacrynic acid, furosemide, urea, or intravenous mannitol. The interaction between inhaled mannitol and tobramycin inhalation solution has not been evaluated.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Aminoglycosides can cause fetal harm. Published literature reports that use of streptomycin, an aminoglycoside, can cause total, irreversible, bilateral congenital deafness when administered to a pregnant woman [Warnings and Precautions (5.6)]. Although there are no available data on use of tobramycin inhalation solution in pregnant women to be able to inform a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes, systemic absorption of tobramycin following inhaled administration is expected to be minimal [see Clinical Pharmacology (12.3)]. There are risks to the mother associated with cystic fibrosis in pregnancy (see Clinical Considerations). In animal reproduction studies with subcutaneous administration of tobramycin in pregnant rats and rabbits during organogenesis there were no adverse developmental outcomes; however, ototoxicity was not evaluated in the offspring from these studies (see Data). Advise pregnant women of the potential risk to a fetus.
The estimated background risk of major birth defects and miscarriage for the indicated populations are unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Cystic fibrosis may increase the risk for preterm delivery.
Data
Animal Data
No reproduction toxicology studies have been conducted with inhaled tobramycin. However, subcutaneous administration of tobramycin at doses of up to 100 (rat) or 20 (rabbit) mg/kg/day during organogenesis was not associated with adverse developmental outcomes. Subcutaneous doses of tobramycin ≥ 40mg/kg/day were severely maternally toxic to rabbits and precluded the evaluation of adverse developmental outcomes. Ototoxicity was not evaluated in offspring during nonclinical reproductive toxicity studies with tobramycin.
8.2 Lactation
There are no data on the presence of tobramycin in either human or animal milk, the effects on the breastfed infant, or the effects on milk production following oral inhalation of tobramycin inhalation solution. Limited published data on other formulations of tobramycin in lactating women indicate that tobramycin is present in human milk. However, systemic absorption of tobramycin following inhaled administration is expected to be minimal [see Clinical Pharmacology (12.3)]. Tobramycin may cause alteration in the intestinal flora of the breastfeeding infant (see Clinical Considerations). The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for tobramycin inhalation solution and any potential adverse effects on the breastfed child from tobramycin inhalation solution or from the underlying maternal condition.
Clinical Considerations
Tobramycin may cause intestinal flora alteration. Advise a woman to monitor the breastfed infant for loose or bloody stools and candidiasis (thrush, diaper rash).
8.4 Pediatric Use
The safety and efficacy of tobramycin inhalation solution have not been studied in pediatric cystic fibrosis patients under six years of age.
8.5 Geriatric Use
Clinical studies of tobramycin inhalation solution did not include patients aged 65 years and over. Tobramycin is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, it may be useful to monitor renal function [see Warnings and Precautions (5.2, 5.5)].
8.6 Renal Impairment
Tobramycin is primarily excreted unchanged in the urine and renal function is expected to affect the exposure of tobramycin. The risk of adverse reactions to this drug may be greater in patients with impaired renal function. Patients with serum creatinine > 2 mg/dL and blood urea nitrogen (BUN) > 40 mg/dL have not been included in clinical studies and there are no data in this population to support a recommendation for or against dose adjustment [see Warnings and Precautions (5.2, 5.5)].
Serum concentrations of tobramycin in patients with renal dysfunction, or patients treated with concomitant parenteral tobramycin should be monitored at the discretion of the treating physician.
-
10 OVERDOSAGE
No overdoses have been reported with tobramycin inhalation solution in clinical trials. Signs and symptoms of acute toxicity from overdosage of intravenous tobramycin might include dizziness, tinnitus, vertigo, loss of high-tone hearing acuity, respiratory failure, neuromuscular blockade, and renal impairment. Administration by inhalation results in low systemic bioavailability of tobramycin. Tobramycin is not significantly absorbed following oral administration. Tobramycin serum concentrations may be helpful in monitoring overdosage.
In all cases of suspected overdosage, physicians should contact the Regional Poison Control Center for information about effective treatment. In the case of any overdosage, the possibility of drug interactions with alterations in drug disposition should be considered.
-
11 DESCRIPTION
Tobramycin inhalation solution, USP is a sterile, a clear, colorless or slight yellow to pale yellow color, non-pyrogenic, aqueous solution with pH and salinity adjusted. Tobramycin inhalation solution, USP is administered by a compressed air driven reusable nebulizer. The chemical formula for tobramycin, USP is C18H37N5O9 and the molecular weight is 467.52. Tobramycin, USP is O-3-amino-3-deoxy-α-D-glucopyranosyl-(1→4)-O-[2,6-diamino-2,3,6-trideoxy-α-D-ribo-hexopyranosyl-(1→6)]-2-deoxy-L-streptamine.
The structural formula for tobramycin, USP is:
Each single-dose 4 mL ampule of tobramycin inhalation solution, USP contains one 300 mg dose of tobramycin, with sodium chloride and sulfuric acid in water for injection. Sulfuric acid and sodium hydroxide are used, as needed, to adjust the pH to 5.0. Nitrogen is used for sparging, filling and pouching. The formulation contains no preservatives.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Tobramycin inhalation solution is an aminoglycoside antibacterial [see Clinical Pharmacology (12.4)].
12.3 Pharmacokinetics
Tobramycin inhalation solution contains tobramycin, a cationic polar molecule that does not readily cross epithelial membranes.1The bioavailability of tobramycin inhalation solution may vary because of individual differences in nebulizer performance and airway pathology.2
Sputum Concentrations
Thirty minutes after inhalation of the first 300 mg dose of tobramycin inhalation solution, the maximum geometric mean concentration of tobramycin was 814 mcg/g (ranging from 23 to 2843 mcg/g) in sputum. High variability of tobramycin concentration in sputum was observed. Three hours after inhalation started, sputum tobramycin concentrations declined to approximately 15% of those observed at 30 minutes. After four weeks of therapy with tobramycin inhalation solution average mean sputum tobramycin concentrations obtained 10 minutes following administration were 717 mcg/g.
Distribution
Following administration of tobramycin inhalation solution, tobramycin remains concentrated primarily in the airways. Binding of tobramycin to serum proteins is negligible.
Elimination
Tobramycin is not metabolized. The elimination half-life of tobramycin from serum is approximately two hours after intravenous (IV) administration. The elimination half-life following the inhalation of tobramycin inhalation solution is approximately 4.4 hours. Assuming tobramycin absorbed following inhalation behaves similarly to tobramycin following intravenous administration, systemically absorbed tobramycin is eliminated principally by glomerular filtration. Unabsorbed tobramycin following inhalation is likely eliminated in expectorated sputum.
12.4 Microbiology
Tobramycin, an aminoglycoside antibacterial, acts primarily by disrupting protein synthesis in the bacterial cell which eventually leads to death of the cell. Tobramycin has activity against a wide range of gram-negative bacteria including P. aeruginosa. It is bactericidal at or above the minimal inhibitory concentration (MIC) needed to inhibit growth of bacteria.
Mechanism of Resistance
The predominant mechanism of resistance to tobramycin in P. aeruginosa isolated from CF patients is impermeability and to a lesser extent enzymatic modification and other mechanisms which cumulatively lead to decreased susceptibility of P. aeruginosa to tobramycin.
Cross Resistance
Cross resistance between aminoglycosides exists but the cross resistance is variable.
Development of Resistance
Treatment for six months with tobramycin inhalation solution in one clinical trial did not affect the susceptibility of the majority of P. aeruginosa isolates tested; however, increases in minimal inhibitory concentrations (MIC) were noted in some patients. The clinical significance of this information has not been clearly established in the treatment of cystic fibrosis patients.
Susceptibility Testing
The clinical microbiology laboratory should provide cumulative results of the in vitro susceptibility test results for antimicrobial drugs used in local hospitals and practice areas to the physicians as periodic reports that describe the susceptibility profile of nosocomial and community-acquired pathogens. These reports should aid the physician in selecting the most effective antimicrobial.
Susceptibility Testing Techniques
Dilution Techniques
Quantitative methods can be used to determine the minimum inhibitory concentration (MIC) of tobramycin that will inhibit the growth of the bacteria being tested. The MIC provides an estimate of the susceptibility of bacteria to tobramycin. The MIC should be determined using a standardized procedure.3, 5Standardized procedures are based on a dilution method (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of tobramycin powder.
Diffusion Techniques
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. One such standardized procedure requires the use of standardized inoculum concentrations.4, 5This procedure uses paper disks impregnated with 10 mcg of tobramycin to test the susceptibility of bacteria to tobramycin.
Susceptibility Test Interpretive Criteria
In vitro susceptibility test interpretive criteria for inhaled tobramycin have not been determined. The relation of the in vitro MIC and/or disk diffusion susceptibility test results to clinical efficacy of inhaled tobramycin against the bacteria tested should be monitored.
Quality Control Parameters for Susceptibility Testing
In vitro susceptibility test quality control parameters exist for tobramycin so that laboratories that test the susceptibility of bacterial isolates to tobramycin can determine if the susceptibility test is performing correctly. Standardized dilution techniques and diffusion methods require the use of laboratory control bacteria to monitor the technical aspects of the laboratory procedures. Standard tobramycin powder should provide the following MIC and a 10 mcg tobramycin disk should produce the following zone diameters with the indicated quality control strains (Table 2).
Table 2: Acceptable Quality Control Ranges for Tobramycin
Bacteria
MIC Range (mcg/mL)
Disk Diffusion Zone Diameter (mm)
Pseudomonas æruginosa ATCC 27853
0.25-1
19-25
No trends in the treatment-emergent isolation of other bacterial respiratory pathogens such as Burkholderia cepacia, Stenotrophomonas maltophilia, Achromobacter xylosoxidans, or Staphylococcus aureus were observed in clinical trials of tobramycin inhalation solution relative to placebo. There was a slight increase in isolation of Candida spp in sputum at the end of the tobramycin inhalation solution treatment cycle in clinical trials.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A two-year rat inhalation toxicology study to assess carcinogenic potential of an inhaled solution of tobramycin has been completed. Rats were exposed to tobramycin for up to 1.5 hours per day for 95 weeks. Serum levels of tobramycin up to 35 mcg/mL were measured in rats, 35x the average 1 mcg/mL exposure levels observed in cystic fibrosis patients in clinical trials. There was no drug-related increase in the incidence of any variety of tumors.
Additionally, tobramycin has been evaluated for genotoxicity in a battery of in vitro and in vivo tests. The Ames bacterial reversion test, conducted with five tester strains, failed to show a significant increase in revertants with or without metabolic activation in all strains. Tobramycin was negative in the mouse lymphoma forward mutation assay, did not induce chromosomal aberrations in Chinese hamster ovary cells, and was negative in the mouse micronucleus test.
Subcutaneous administration of up to 100 mg/kg of tobramycin did not affect mating behavior or cause impairment of fertility in male or female rats.
-
14 CLINICAL STUDIES
Two, double-blind, randomized, placebo-controlled, parallel group clinical studies (Study 1 and Study 2), which randomized and dosed 306 patients, were conducted in cystic fibrosis patients with P. aeruginosa. The osmolality of the drug formulation used in these studies differed from the to-be-marketed product. To rely upon the efficacy and safety established in the placebo-controlled studies, an additional study was conducted as a bridge to the to-be-marketed drug. The bridging study assessed the efficacy and tolerability of aerosolized Tobramycin Inhalation Solution with osmolality similar to tobramycin inhalation solution over a 4-week treatment in 324 patients with cystic fibrosis. Results of this study showed that the Tobramycin Inhalation Solution in this study had similar efficacy as that seen in the placebo-controlled studies.
The compressors in the placebo-controlled studies and the bridging study differed from the PARI VIOS compressor to be used with tobramycin inhalation solution. In vitro cascade impaction studies demonstrated that the various compressors used in the clinical trials delivered equivalent doses and respirable fractions of the to-be-marketed tobramycin inhalation solution and TOBI with the marketed compressor (PARI VIOS) when used with the same nebulizer (PARI LC Plus Reusable nebulizer).
All subjects enrolled in both efficacy studies had baseline FEV1 % predicted ≥ 40% and ≤80% (mean baseline FEV1 of 60% of predicted normal) and infected with P. aeruginosa. Subjects who were less than 6 years of age, or who had a baseline creatinine of ≥ 1.5 mg/dL, or who had Burkholderia cepacia isolated from sputum were excluded. A total of 190 patients, 29 in Study 1 and 161 in Study 2, received tobramycin inhalation solution therapy on an outpatient basis. Of these, 55% were males and 45% were females. Eighty-two (43.2%) patients were between 6 and 12 years of age, 54 (28.4%) patients were between 13 and 17 years of age, and the remaining 54 (28.4%) patients were greater than 17 years of age. Of the patients who received tobramycin inhalation solution, only 89.7% of patients in Study 1 had at least one concomitant medication, while all patients in Study 2 also received at least one concomitant medication. These concomitant medications include mucolytics, steroidal and nonsteroidal anti-inflammatory drugs, bronchodilators, rehabilitative physiotherapies and if necessary, antibiotics for bacterial infections other than P. aeruginosa.
Study 1
Study 1 was a double-blind, single cycle study that randomized 59 patients to receive tobramycin inhalation solution (n=29) or placebo (n=30) for one cycle of treatment (28 days on treatment followed by 28 days off treatment). All patients were ≤ 30 years of age (mean age 12.6 years) and 46% were females. All randomized patients were included in the primary analysis except for one patient who had missing baseline information.
Tobramycin inhalation solution significantly improved lung function compared with placebo as measured by the absolute change in FEV1 % predicted from baseline to the end of Cycle 1 dosing in the primary analysis population. Treatment with tobramycin inhalation solution and placebo resulted in absolute increases in FEV1 % predicted of 16% and 5%, respectively (LS mean difference = 11%; 95% CI: 3, 19; p=0.003). This analysis is adjusted for the covariate of baseline FEV1 % predicted, using multiple imputation for missing data. Figure 1 shows the average change in FEV1 % predicted over eight weeks.
Study 2
Study 2 was a randomized, double-blind, 3-cycle, placebo-controlled trial. A total of 247 eligible patients were randomized 2:1 to receive three cycles of tobramycin inhalation solution (n=161) or placebo (n=86). As in Study 1, each cycle comprised 28 days on treatment followed by 28 days off treatment. All patients were ≤46 years of age (mean age 14.8 years) and 44.9% were females. In this study, two randomized patients in the placebo group were not included in the primary efficacy analysis; one withdrew consent without taking any trial medication and the other withdrew due to an adverse drug reaction.
Tobramycin inhalation solution significantly improved lung function compared with placebo as measured by the absolute change in FEV1 % predicted from baseline to the end of Cycle 3 "ON" period. Treatment with tobramycin inhalation solution and placebo resulted in absolute increases in FEV1 % predicted of 7% and 1%, respectively (LS mean difference = 6%; 95% CI: 3, 10; p<0.001). This analysis is adjusted for the covariate of baseline FEV1 % predicted, using multiple imputation for missing data. Figure 1 shows the average change in FEV1 % predicted over 24 weeks from Study 2.
Figure 1: FEV1 % of Predicted Normal–Absolute Change from Baseline (Adjusted mean)–ITT Population
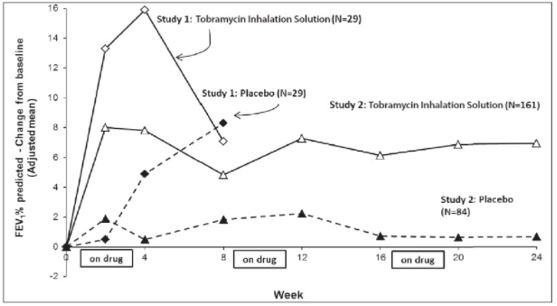
In Study 2, 9.9% of patients treated with tobramycin inhalation solution and 24.7% of patients who received placebo had unplanned hospitalizations due to the disease.
Also in Study 2, 6.2% of patients treated with tobramycin inhalation solution and 16.5% of placebo patients received parenteral tobramycin.
-
15 REFERENCES
- Neu HC. Tobramycin: an overview. [Review]. J Infect Dis. 1976; Suppl 134:S3-19.
- Weber A, Smith A, Williams-Warren J, et al. Nebulizer delivery of tobramycin to the lower respiratory tract. Pediatr Pulmonol. 1994; 17(5):331-9.
- Clinical and laboratory Standards Institute (CLSI) Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically: Approved Standard -9th edition. CLSI document M07-A9. CLSI 940 West Valley Rd. Suite 1400, Wayne PA 19087-1898. 2012
- CLSI Performance Standards for Antimicrobial Susceptibility Testing: 22nd Informational supplement CLSI document M100-S22. CLSI 2012.
- CLSI Performance standards for Antimicrobial Disk Susceptibility Tests: Approved standard -11th ed. CLSI document M02-A11. CLSI 2012.
- PARI Vios Aerosol Delivery System with LC Plus Nebulizer: Instructions for Use. PARI Respiratory Equipment, Inc. 2010; 310D0028 Rev A 6-10.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Tobramycin inhalation solution, USP 300 mg/4 mL is supplied as a sterile, a clear, colorless or slight yellow to pale yellow color, non-pyrogenic, aqueous solution and is available as follows:
- NDC: 70756-617-56: 4 mL single-dose ampule (carton of 14 foil pouches each containing four ampules)
16.2 Storage and Handling
Tobramycin inhalation solution, USP should be stored under refrigeration at 2°C to 8°C (36°F to 46°F). Upon removal from the refrigerator, or if refrigeration is unavailable, tobramycin inhalation solution, USP pouches (opened or unopened) may be stored at room temperature [up to 25°C (77°F)] for up to 28 days. Tobramycin inhalation solution, USP should not be used beyond the expiration date stamped on the ampule when stored under refrigeration 2°C to 8°C (36°F to 46°F) or beyond 28 days when stored at room temperature [up to 25°C (77°F)].
Tobramycin inhalation solution, USP ampules should not be exposed to intense light. Tobramycin inhalation solution, USP is light sensitive; unopened ampules should be returned to the foil pouch. The solution in the ampule is colorless to pale yellow, but may darken with age if not stored in the refrigerator; however, the color change does not indicate any change in the quality of the product as long as it is stored within the recommended storage conditions.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Information for Patients
Information on the long term efficacy and safety of tobramycin inhalation solution is limited. There is no information in patients with severe cystic fibrosis (FEV1 < 40% predicted).
Patients should be advised to complete a full 28-day course of tobramycin inhalation solution, even if they are feeling better. After 28 days of therapy, patients should stop tobramycin inhalation solution therapy for the next 28 days, and then resume therapy for the next 28 day on and 28 day off cycle.
For patients taking several different inhaled medications and/or performing chest physiotherapy, advise the patient regarding the order they should take the therapies. It is recommended that tobramycin inhalation solution be taken last.
Tobramycin inhalation solution is to be used with the PARI LC PLUS reusable nebulizer and the PARI VIOS air compressor. Refer to the manufacturer's instructions for care and use of the nebulizer and compressor.
17.1 Ototoxicity
Inform patients that ototoxicity, as measured by complaints of hearing loss or tinnitus, was reported by patients treated with tobramycin. Physicians should consider an audiogram at baseline, particularly for patients at increased risk of auditory dysfunction.
If a patient reports tinnitus or hearing loss during tobramycin inhalation solution therapy, the physician should refer that patient for audiological assessment.
Patients should be reminded that vestibular toxicity may manifest as vertigo, ataxia, or dizziness.
17.3 Risks Associated with Aminoglycosides
Inform patients of adverse reactions associated with aminoglycosides such as nephrotoxicity and neuromuscular disorders.
17.4 Laboratory Tests
Inform patients of the need to monitor hearing, serum concentrations of tobramycin, or renal function as necessary during treatment with tobramycin inhalation solution.
17.5 Embryo-Fetal Toxicity
Inform patients that aminoglycosides can cause fetal harm when administered to a pregnant woman. Advise them to inform their doctor if they are pregnant, become pregnant, or plan to become pregnant.
17.6 Administration
Patients should be informed about what to do in the event they miss a dose of tobramycin inhalation solution:
- In case a dose of tobramycin inhalation solution is missed and there are at least 6 hours until the next dose, patients should be instructed to take the prescribed dose of tobramycin inhalation solution as soon as possible. Otherwise, the missed dose should not be taken and the patient should resume the usual dosing schedule.
- Patients should be advised to contact their healthcare provider if they have questions.
17.7 Storage Instructions
You should store tobramycin inhalation solution ampules in a refrigerator (36°F to 46°F or 2°C to 8°C). However, when you don't have a refrigerator available (e.g., transporting your tobramycin inhalation solution), you may store the foil pouches (opened or unopened) at room temperature (up to 77°F/25°C) for up to 28 days.
Tobramycin inhalation solution is light sensitive; unopened ampules should be returned to the foil pouch. Avoid exposing tobramycin inhalation solution ampules to intense light. Unrefrigerated tobramycin inhalation solution, which is normally colorless to pale yellow, may darken with age; however, the color change does not indicate any change in the quality of the product.
You should not use tobramycin inhalation solution if it is cloudy, if there are particles in the solution, or if it has been stored at room temperature for more than 28 days. You should not use tobramycin inhalation solution beyond the expiration date stamped on the ampule.
Manufactured for:
Lifestar Pharma LLC
1200 MacArthur Blvd.
Mahwah, NJ 07430 USA
Made in India
Revised: February 2023, V-04
All trademarks referenced herein are the property of their prospective owners
-
SPL PATIENT PACKAGE INSERT
Patient Information
Tobramycin Inhalation Solution
( toh-bruh- mahy -sin )
for oral inhalation use
What is tobramycin inhalation solution?
Tobramycin inhalation solution is a prescription medicine that is used to treat people with cystic fibrosis who have a bacterial infection called Pseudomonas aeruginosa . Tobramycin inhalation solution contains an antibacterial medicine called tobramycin (an aminoglycoside).
It is not known if tobramycin inhalation solution is safe and effective:
- in children under 6 years of age
- in people who have decreased lung volume or a forced expiratory volume in one second (FEV1) less than 40% or greater than 80% predicted
- in people who are colonized with a bacterium called Burkholderia cepacia
Do not take tobramycin inhalation solution if you are allergic to tobramycin, any of the ingredients in tobramycin inhalation solution, or to any other aminoglycoside antibacterial.
See the end of this Patient Information for a complete list of ingredients in tobramycin inhalation solution.
Before you take tobramycin inhalation solution, tell your healthcare provider about all of your medical conditions, including if you:
- have or have had hearing problems (including noises in your ears such as ringing or hissing), hearing loss, or your mother has had hearing problems after taking an aminoglycoside.
- have been told you have certain gene variants (a change in the gene) related to hearing abnormalities inherited from your mother.
- have dizziness
- have or have had kidney problems
- have or have had problems with muscle weakness such as myasthenia gravis or Parkinson's disease
- have or have had breathing problems such as wheezing, coughing, or chest tightness
- are pregnant or plan to become pregnant. Tobramycin inhalation solution is in a class of medicines that can harm your unborn baby and may be connected with complete deafness in babies at birth. The deafness affects both ears and cannot be changed.
- are breastfeeding or plan to breastfeed. It is not known if tobramycin passes into your breast milk. Tobramycin, the medicine in tobramycin inhalation solution may cause the following symptoms in your breastfed baby:
- loose or bloody stools
- yeast infection in the mouth or throat (thrush)
- diaper rash
- are receiving aminoglycoside therapy by injection or through a vein (intravenous) while taking tobramycin inhalation solution. Your blood levels of tobramycin will be checked.
How should I take Tobramycin Inhalation Solution?
- See the step-by-step Instructions for Use at the end of this Patient Information leaflet about the right way to take your tobramycin inhalation solution.
- Take tobramycin inhalation solution exactly as your healthcare provider tells you to. Do not change your dose or stop taking tobramycin inhalation solution unless your healthcare provider tells you to.
- The usual dose of tobramycin inhalation solution for adults and children over 6 years of age is:
- 1 single-dose ampule of tobramycin inhalation solution inhaled 2 times each day using your hand-held PARI LC PLUS Reusable Nebulizer with a PARI Vios air compressor.
- Each dose of tobramycin inhalation solution should be taken as close to 12 hours apart as possible.
- You should not take your dose of tobramycin inhalation solution less than 6 hours apart.
- Tobramycin inhalation solution is taken as a breathing treatment (inhalation) with a hand-held PARI LC Reusable Nebulizer with a PARI Vios air compressor. Do not use any other nebulizer for your tobramycin inhalation solution treatment.
- Do not mix or dilute tobramycin inhalation solution with dornase alfa or other medicines in your nebulizer system.
- Each treatment of tobramycin inhalation solution should take about 15 minutes.
- Tobramycin inhalation solution should be inhaled while you are sitting or standing upright and breathing normally through the mouthpiece of the nebulizer. Nose clips may help you to breathe through your mouth.
- If you forget to take tobramycin inhalation solution and there are at least 6 hours to your next dose, take your dose as soon as you can. Otherwise, wait for your next dose. Do not double the dose to make up for the missed dose.
- After taking tobramycin inhalation solution for 28 days, you should stop taking it and wait 28 days. After you have stopped taking tobramycin inhalation solution for 28 days, you should start taking tobramycin inhalation solution again for 28 days. Complete the full 28-day course even if you are feeling better. It is important that you keep to the 28-day on, 28-day off cycle.
- other medicines that may harm your nervous system, kidneys, or hearing
- "water pills" (diuretics) such as ethacrynic acid, furosemide, or intravenous mannitol
- Urea
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
- If you take too much tobramycin inhalation solution, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of Tobramycin inhalation solution?
Tobramycin inhalation solution can cause serious side effects, including:
- hearing loss or ringing in the ears (ototoxicity). Some people who were treated with tobramycin, the medicine in tobramycin inhalation solution had hearing loss or ringing in the ears. Tell your healthcare provider right away if you have hearing loss or hear noises in your ears (such as ringing or hissing), or if you develop vertigo, dizziness, or difficulty with balance.
- worsening kidney problems (nephrotoxicity). Your healthcare provider may do a blood test and urine test to check how your kidneys are working while you are taking tobramycin inhalation solution.
- worsening muscle weakness (neuromuscular disorder). Tobramycin inhalation solution can cause muscle weakness to get worse in people who already have problems with muscle weakness (myasthenia gravis or Parkinson's disease).
- severe breathing problems (bronchospasm). Tell your healthcare provider right away if you get any of these symptoms of bronchospasm while taking tobramycin inhalation solution:
o coughing and chest tightness
The most common side effects of tobramycin inhalation solution include:
- worsening of lung problems or cystic fibrosis
- noisy breathing (rales)
- abnormal red blood cell activity
- changes in your voice (hoarseness)
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of tobramycin inhalation solution.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use tobramycin inhalation solution for a condition for which it was not prescribed. Do not give tobramycin inhalation solution to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your pharmacist or healthcare provider for more information about tobramycin inhalation solution that is written for health professionals.
What are the ingredients in tobramycin inhalation solution?
Active ingredient: tobramycin
Inactive ingredients: sodium chloride, sulfuric acid in water for injection, and sodium hydroxide (for pH adjustment)
What is Psuedomonas aeriguinosa?
It is a very common bacterium that infects the lungs of nearly everyone with cystic fibrosis at some time during their lives. Some people do not get this infection until later in their lives, while others get it very young. It is one of the most damaging bacteria for people with cystic fibrosis. If the infection is not properly managed, it will continue to damage your lungs causing further problems to your breathing.
For more information, go to www.lifestarpharma.com or call 1-888-995-4337.
This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 02/2023, V-03
Instructions for Use
Tobramycin inhalation solution
(toh-bruh-mahy-sin)
Follow the instructions below for taking tobramycin inhalation solution. If you have any questions, ask your healthcare provider or pharmacist.
Tobramycin inhalation solution is available as a 28-day supply containing 56 ampules including 14 foil pouches. Each foil pouch contains 4 tobramycin inhalation solution ampules.
Supplies you will need to take tobramycin inhalation solution (See Figure A):
- 1 ampule of tobramycin inhalation solution
- PARI LC PLUS reusable nebulizer
- PARI Vios compressor
- tubing to connect the nebulizer and compressor
- clean paper or cloth towels
- nose clips (optional)
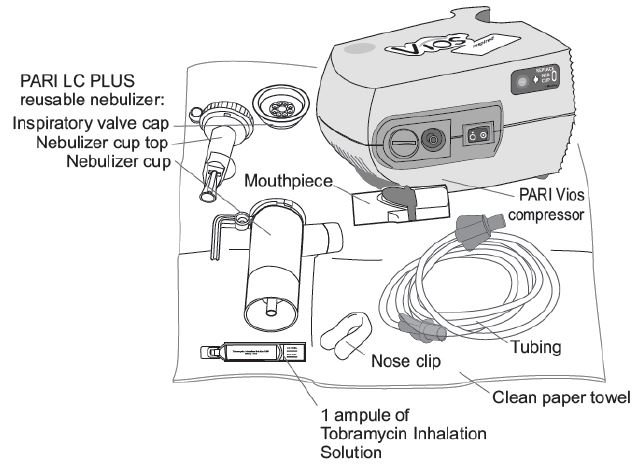
Tobramycin inhalation solution is used only in a PARI LC PLUS re-usable Nebulizer connected to a PARI LC PLUS Vios air compressor. Make sure you know how to use your nebulizer machine before you use it to breathe in tobramycin inhalation solution.
Do not mix tobramycin inhalation solution with other medicines in your nebulizer.
Tobramycin inhalation solution comes in a sealed foil pouch. Do not open a sealed pouch until you are ready to use a dose of tobramycin inhalation solution. After opening the pouch, unused ready-to-use ampules should be returned to, and stored in, the pouch.
Getting ready:
- Put your PARI LC PLUS Reusable Nebulizer Top and Bottom (Nebulizer Cup) Assembly, Inspiratory Valve Cap, Mouthpiece with Valve, and Tubing on a clean and dry surface.
- Wash your hands with soap and water.
Preparing your tobramycin inhalation solution dose:
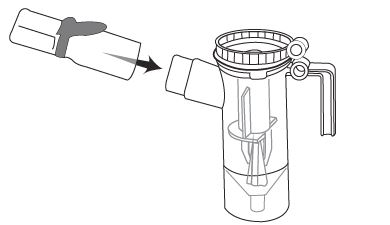
Step 1: Open foil pouch. (See Figure B)
Step 2: Separate 1 ampule by gently pulling apart at the bottom tabs (See Figure C) and use it right away.
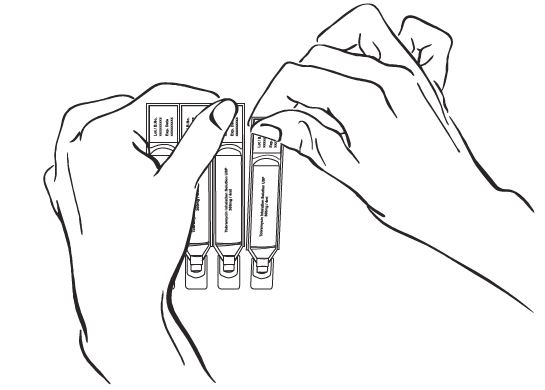
Step 3: Hold the bottom tab on the tobramycin inhalation solution ampule with 1 hand (See Figure D). With your other hand, hold the top of the ampule and twist off the top of the ampule (See Figure D).
- Do not squeeze the ampule until you are ready to squeeze all the medicine into the Nebulizer Cup.
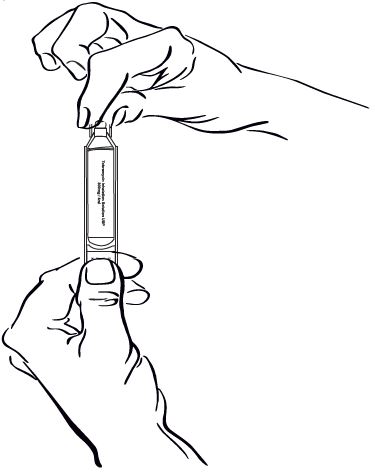
Step 4: Hold the Nebulizer Cup and twist off the Nebulizer Cup Top in a counter-clockwise direction (See Figure E). Set the Top aside on a clean, dry surface.
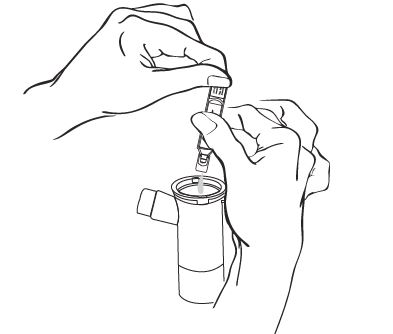
Step 5: Squeeze all of the medicine from the ampule into the Nebulizer Cup (See Figure F).
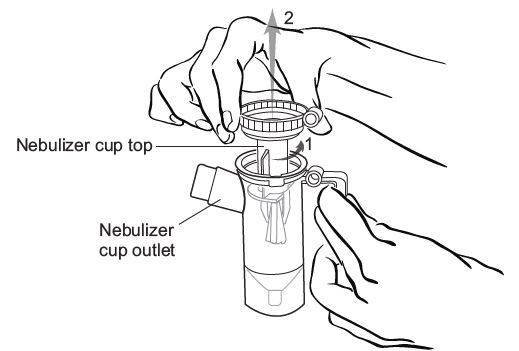
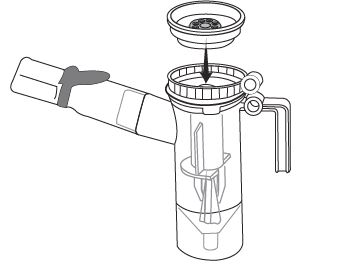
Step 6: Line up the semi-circle on the Nebulizer Cup Top with the Nebulizer Cup Outlet and twist on the Nebulizer Cup Top in a clock-wise direction until it is tight. (See Figure G).
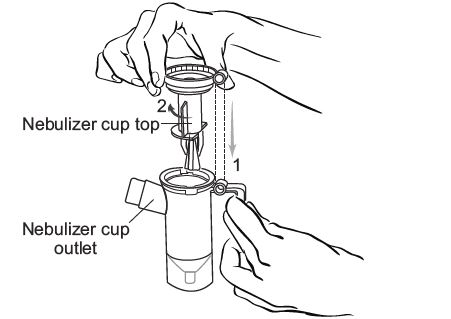
Step 7: Push the mouthpiece straight onto the Nebulizer Cup Outlet (See Figure H).
Step 8: Firmly push the Inspiratory Valve Cap straight down onto the Nebulizer Cup Top (See Figure I). The Inspiratory Valve Cap should fit tightly.
Step 9: Connect 1 end of the tubing to the compressor air outlet. The tubing should fit tightly (See Figure J).
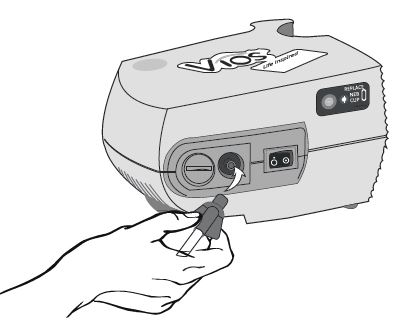
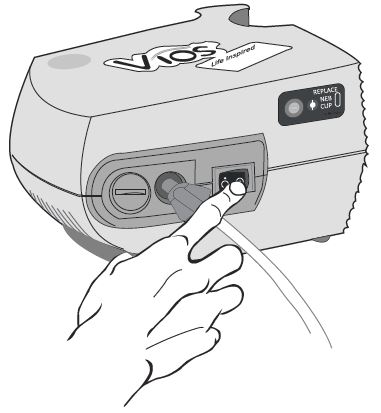
Step 10: Plug your compressor plug into an electrical outlet (See Figure K).
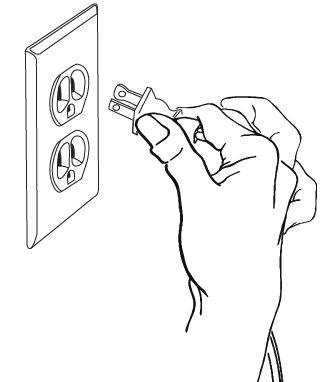
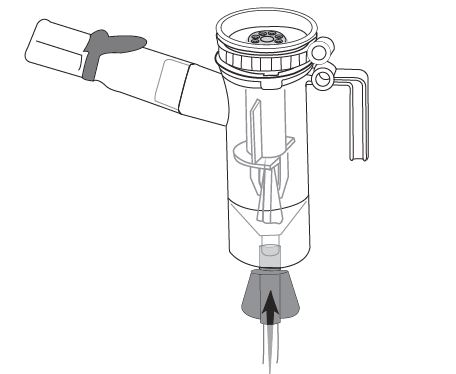
Step 11: Hold the Nebulizer Cup upright and firmly push the free end of the tubing straight up onto the Air Intake on the bottom of the Nebulizer Cup (See Figure L). Make sure to keep the Nebulizer Cup upright.
Giving your Tobramycin inhalation solution dose:
Step 12: Turn on the compressor (Figure M) and check the Mouthpiece. You should see a steady mist coming from the Mouthpiece (Figure N).
- If you do not see a steady mist coming from the mouthpiece, check all tubing connections and make sure that the compressor is working the right way.
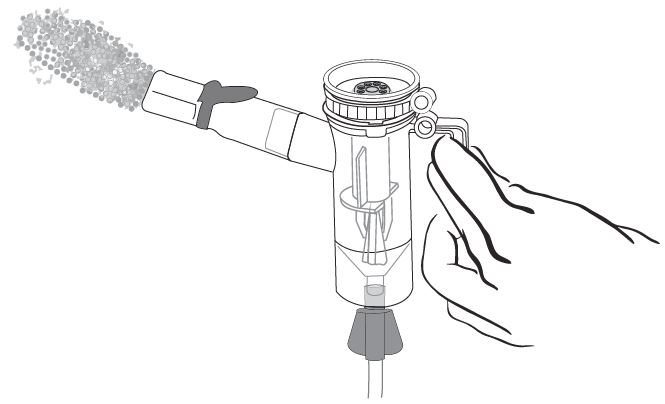
Step 13: Sit or stand in a comfortable, upright position that will let you breathe normally. Place the Mouthpiece between your teeth and on top of your tongue and breathe normally only through your mouth (See Figure O).
- Nose clips may help you breathe only through your mouth and not through your nose.
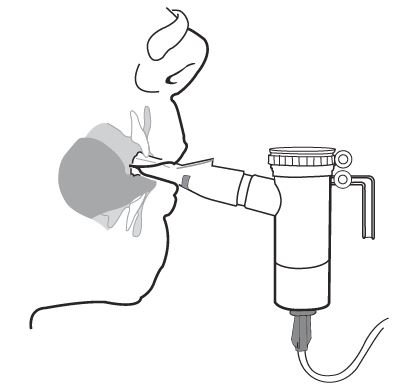
Step 14: Keep breathing in your tobramycin inhalation solution dose for at least 15 minutes. You will know that you have received your full dose of medicine when you hear a "spitting noise" coming from the Mouthpiece for at least 1 minute and the Nebulizer Cup is empty.
After your Tobramycin Inhalation Solution Dose:
Step 15: Clean and disinfect your nebulizer (see manufacturer's instructions).
Care and Use of Your PARI Vios®Compressor
Follow the manufacturer's instructions for care and use of your compressor.
How should I store Tobramycin inhalation solution?
- Store tobramycin inhalation solution in the refrigerator at 36°F to 46°F (2°C to 8°C) until needed.
- After removing from the refrigerator, or if refrigeration is not available, tobramycin inhalation solution foil pouches (opened or unopened) may be stored at room temperature up to 77°F (25°C) for up to 28 days.
- If tobramycin inhalation solution is not stored in the refrigerator but at room temperature up to 77°F (25°C) it may turn dark. If tobramycin inhalation solution turns dark, it does not change how well tobramycin inhalation solution works. Tobramycin inhalation solution can still be used as long as it is stored at room temperature up to 77°F (25°C).
- Do not use tobramycin inhalation solution after the expiration date printed on the ampule.
- Keep tobramycin inhalation solution ampules in the foil pouch and away from light.
- Return unopened ampules to the foil pouch
- Keep tobramycin inhalation solution and all medicines out of the reach of children.
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Manufactured for:
Lifestar Pharma LLC
1200 MacArthur Blvd.
Mahwah, NJ 07430 USA
Made in India
All trademarks referenced herein are the property of their prospective owners
Revised: July 2022, V-03
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 70756-617-44
Tobramycin Inhalation
Solution, USP
300 mg/4 mL
For Oral Inhalation Only by Nebulizer
Single-Dose Only. Discard Each Ampule
After One Use.
Each foil pouch contains 4 ampules.
Storage: Store in a Refrigerator at 2°C to 8°C
(36°F to 46°F). Protect from Intense Light.
Tobramycin inhalation solution is light sensitive;
unopened ampules should be returned to
the foil pouch.
Rx Only
NDC: 70756-617-56
Tobramycin Inhalation
Solution, USP
300 mg/4 mL
For Oral Inhalation Only by Nebulizer
Single-Dose Only. Discard Each Ampule After One Use.
Storage: Store in a Refrigerator at 2°C to 8°C (36°F to 46°F).
Tobramycin inhalation solution is light sensitive;
unopened ampules should be returned to the foil pouch.
Each carton contains 56 Single-Dose Ampules (28-Day Supply)
Rx Only

-
INGREDIENTS AND APPEARANCE
TOBRAMYCIN
tobramycin solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 70756-617 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TOBRAMYCIN (UNII: VZ8RRZ51VK) (TOBRAMYCIN - UNII:VZ8RRZ51VK) TOBRAMYCIN 300 mg in 4 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SULFURIC ACID (UNII: O40UQP6WCF) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 70756-617-56 14 in 1 CARTON 09/23/2022 1 NDC: 70756-617-44 4 in 1 POUCH 1 4 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA216725 09/23/2022 Labeler - Lifestar Pharma LLC (080268943) Registrant - Mankind Pharma Limited (915834068) Establishment Name Address ID/FEI Business Operations Mankind Pharma Limited 916512493 MANUFACTURE(70756-617) , ANALYSIS(70756-617) , PACK(70756-617)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.