Alcohol Prep Pads by ALCOVE BRANDS Alcohol Prep Pads
Alcohol Prep Pads by
Drug Labeling and Warnings
Alcohol Prep Pads by is a Otc medication manufactured, distributed, or labeled by ALCOVE BRANDS. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
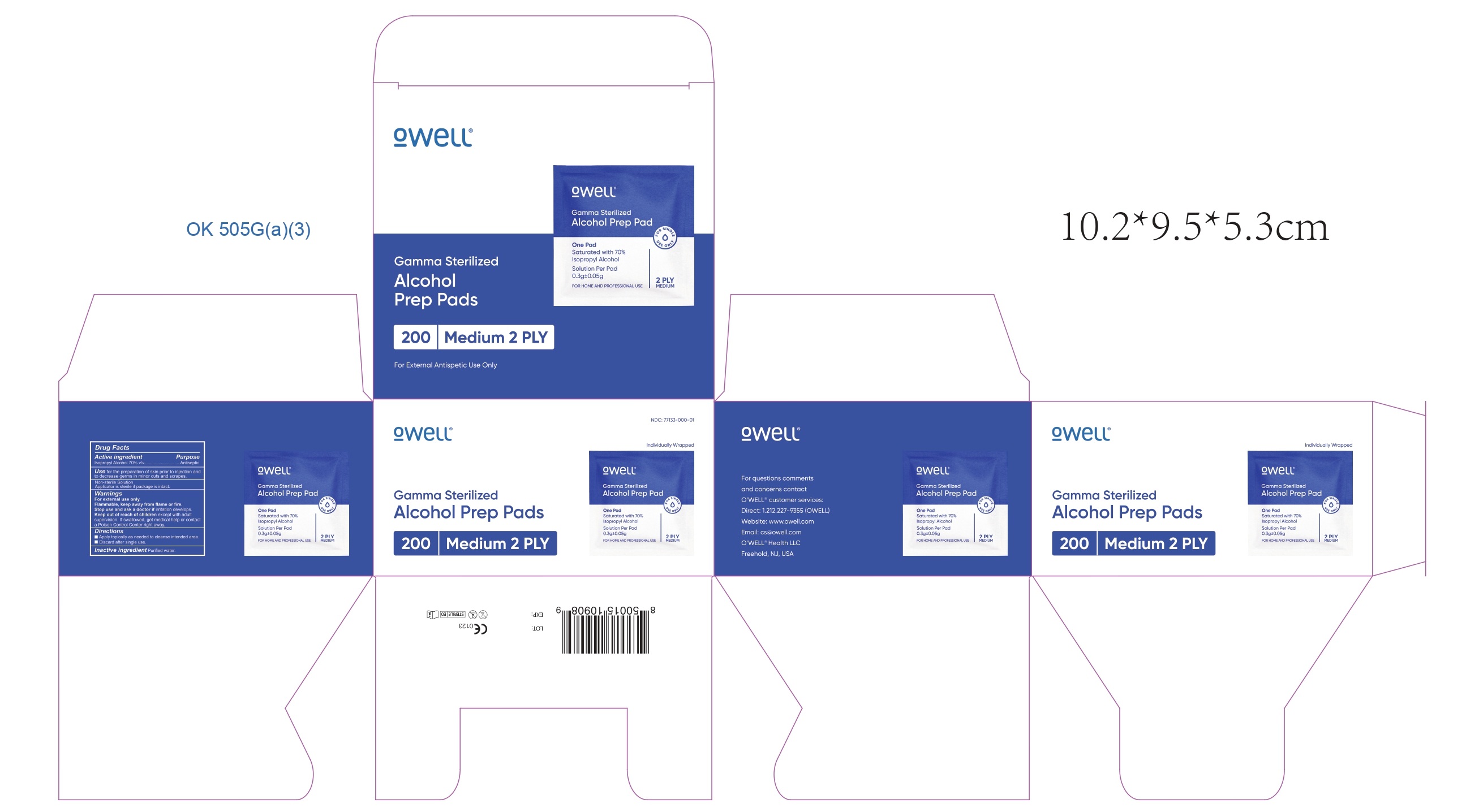
ALCOHOL PREP PADS- isopropyl alcohol cloth
ALCOVE BRANDS
----------
Alcohol Prep Pads
Use
for the preparation of skin prior to injection and to decrease germs in minor cuts and scrapes.
Non-sterile Solution
Applicator is sterile if package is intact.
| ALCOHOL PREP PADS
isopropyl alcohol cloth |
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - ALCOVE BRANDS (052943257) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Shandong Lianfa Medical Plastic Products Co. Ltd. | 421330121 | manufacture(77133-000) | |
Revised: 2/2026
Document Id: 4bae6c9a-d67d-3992-e063-6394a90a3758
Set id: 96b235af-7a11-4b52-8cc2-c5bfb2ad9115
Version: 11
Effective Time: 20260225
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.