SINUS CONGESTION AND PAIN NIGHTTIME- acetaminophen, chlorpheniramine maleate and phenylephrine hcl tablet, film coated
Sinus Congestion and Pain by
Drug Labeling and Warnings
Sinus Congestion and Pain by is a Otc medication manufactured, distributed, or labeled by FRED'S, INC., LNK International, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredients (in each caplet)
- Purpose
-
Uses
- temporarily relieves these symptoms of hay fever or other respiratory allergies, and the common cold:
- headache
- nasal congestion
- runny nose and sneezing
- minor aches and pains
- sinus congestion and pressure
- headache
- temporarily reduces fever
- promotes sinus drainage
- helps clear nasal passages
- helps decongest sinus openings and passages
- temporarily relieves these symptoms of hay fever or other respiratory allergies, and the common cold:
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 4,000 mg in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- blisters
- rash
- skin reddening
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- difficulty in urination due to enlargement of the prostate gland
- liver disease
- diabetes
- glaucoma
- high blood pressure
- a breathing problem such as emphysema or chronic bronchitis
- heart disease
- thyroid disease
Ask a doctor or pharmacist before use if you are
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers
When using this product
- do not exceed recommended dosage
- excitability may occur, especially in children
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic beverages
- use caution when driving a motor vehicle or operating machinery
- may cause drowsiness
- more than 4,000 mg in 24 hours, which is the maximum daily amount
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
fred's
PharmacyNDC: 55315-455-08
Nighttime
SINUS
CONGESTION & PAIN
Acetaminophen, Chlorpheniramine maleate,
Phenylephrine HClPain Reliever/Fever Reducer, Antihistamine, Nasal Decongestant
Pseudoephedrine Free
Headache Sinus Pressure,
Runny Nose Nasal Congestion24 Cool Caplets with Cool Blast Flavor
Compare to the active ingredients in:
Tylenol® Sinus Congestion & Pain Nighttime*TAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN, BROKEN
OR SHOWS ANY SIGNS OF TAMPERINGDISTRIBUTED BY: fred's, Inc.
4300 NEW GETWELL RD, MEMPHIS, TN 38118
www.fredsinc.com100% satisfaction
guaranteed
*This product is not manufactured or distributed by McNeil
Consumer Healthcare, owner of the registered trademark
Tylenol® Sinus Congestion & Pain Nighttime.
50844 REV0616G45508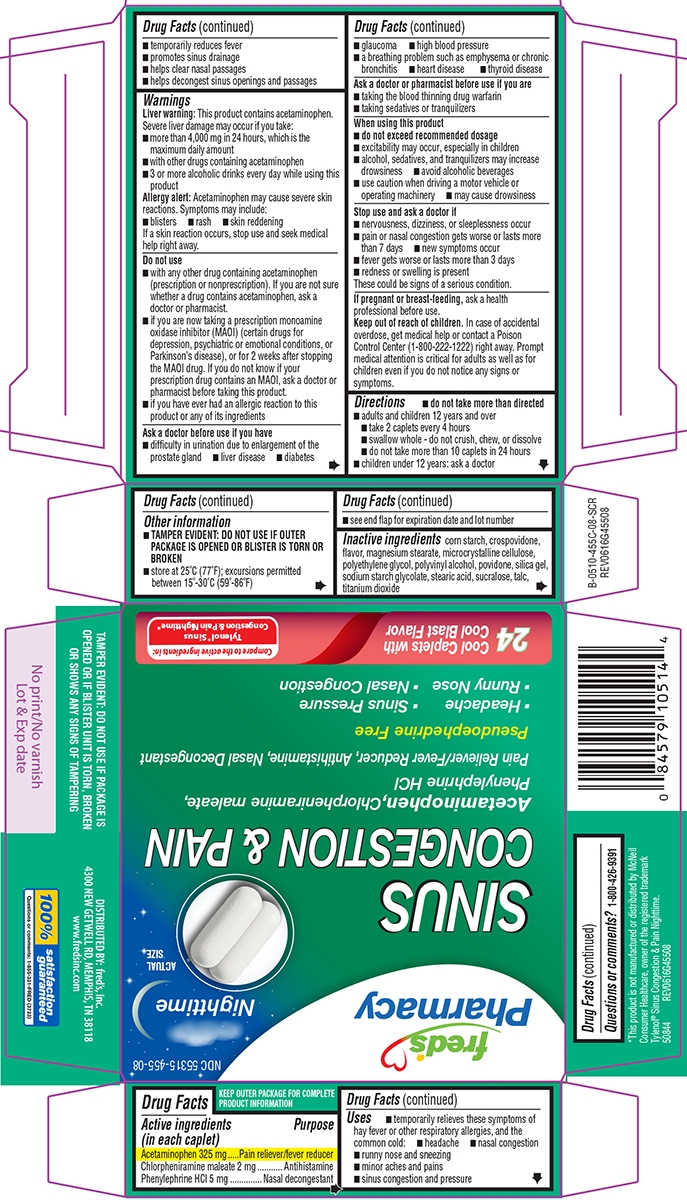
Freds 44-455C
-
INGREDIENTS AND APPEARANCE
SINUS CONGESTION AND PAIN NIGHTTIME
acetaminophen, chlorpheniramine maleate and phenylephrine hcl tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 55315-455 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 2 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STEARIC ACID (UNII: 4ELV7Z65AP) SUCRALOSE (UNII: 96K6UQ3ZD4) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STARCH, CORN (UNII: O8232NY3SJ) POVIDONE (UNII: FZ989GH94E) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) TALC (UNII: 7SEV7J4R1U) CROSPOVIDONE (UNII: 2S7830E561) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score no score Shape OVAL Size 17mm Flavor MINT Imprint Code 44;455 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 55315-455-08 2 in 1 CARTON 06/28/2005 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part341 06/28/2005 Labeler - FRED'S, INC. (005866116) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 PACK(55315-455) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 MANUFACTURE(55315-455) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 PACK(55315-455) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 PACK(55315-455) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 868734088 PACK(55315-455)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.