Allergy Relief by Preferred Pharmaceuticals, Inc. Drug Facts
Allergy Relief by
Drug Labeling and Warnings
Allergy Relief by is a Otc medication manufactured, distributed, or labeled by Preferred Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
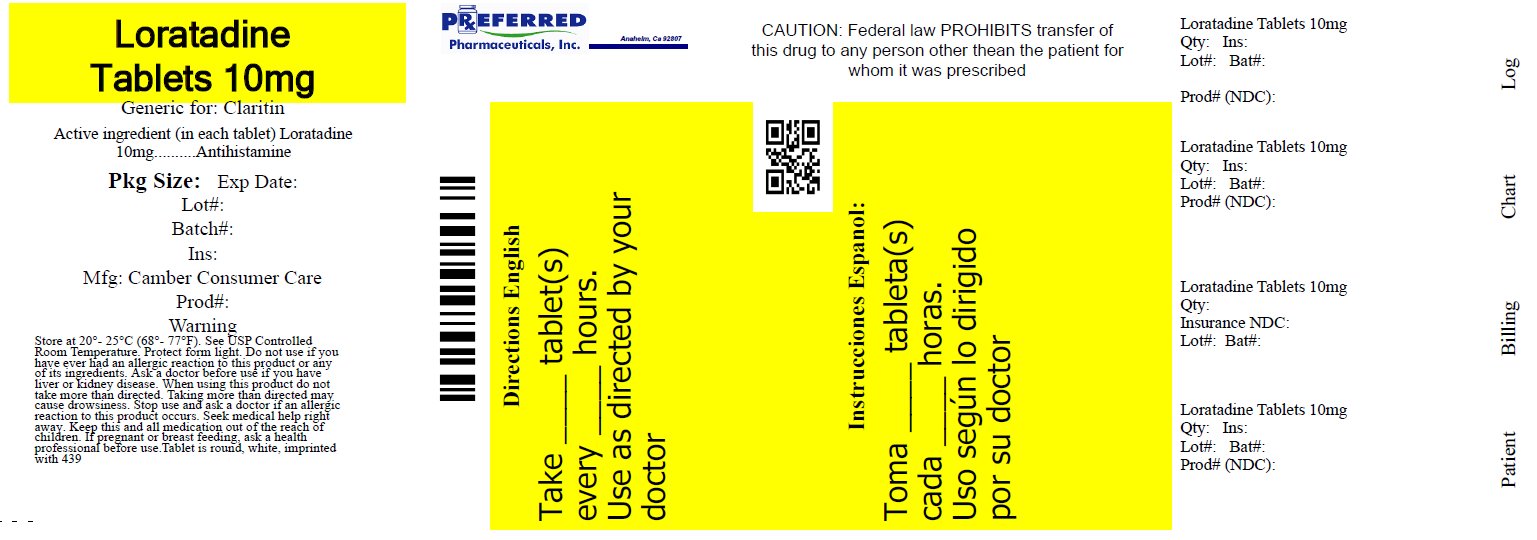
ALLERGY RELIEF NON DROWSY- loratadine tablet
Preferred Pharmaceuticals, Inc.
----------
Drug Facts
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Warnings
Ask a doctor before use if you have
liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product,
do not take more than directed. Taking more than directed may cause drowsiness.
Directions
|
adults and children 6 years and over |
1 tablet daily; not more than 1 tablet in 24 hours |
|
children under 6 years of age |
ask a doctor |
|
consumers with liver or kidney disease |
ask a doctor |
Other information
- store at 20º-25ºC (68º-77ºF) (see UPS Controlled Room Temperature)
- protect from light
Inactive ingredients
lactose monohydrate, magnesium stearate, microcrystalline cellulose, sodium starch glycolate
Principal Display Panel
†Compare to the active ingredient in Claritin® 24 Hour
Non-drowsy*
Allergy Relief
Loratadine Tablets, USP 10 mg / Antihistamine
Indoor & outdoor allergies
24 Hour Relief of:
- Sneezing
- Runny Nose
- Itchy, Watery Eyes
- Itchy Nose or Throat
Tablets
Gluten-Free
*When taken as directed. See Drug Facts panel.
†This product is not manufactured or distributed by Bayer Healthcare LLC, distributor of Claritin® 24 Hour
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAPS IS BROKEN OR MISSING.
Distributed by:
Camber Consumer Care Inc., Piscataway, NJ 08854, USA
Repackaged By: Preferred Pharmaceuticals Inc.
| ALLERGY RELIEF
NON DROWSY
loratadine tablet |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Labeler - Preferred Pharmaceuticals, Inc. (791119022) |
| Registrant - Preferred Pharmaceuticals, Inc. (791119022) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Preferred Pharmaceuticals, Inc. | 791119022 | REPACK(68788-7769) | |
Trademark Results [Allergy Relief]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ALLERGY RELIEF 98236984 not registered Live/Pending |
Dmytro Kononenko 2023-10-24 |
 ALLERGY RELIEF 90457167 not registered Live/Pending |
American Textile Company, Inc. 2021-01-10 |
 ALLERGY RELIEF 78838437 3358249 Live/Registered |
Meshbesher Health Corporation 2006-03-16 |
 ALLERGY RELIEF 76619855 3066888 Live/Registered |
AMERICAN TEXTILE COMPANY 2004-11-09 |
 ALLERGY RELIEF 74668018 not registered Dead/Abandoned |
NaturaLife Corporation 1995-05-01 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.