GANIRELIX ACETATE injection
Ganirelix Acetate by
Drug Labeling and Warnings
Ganirelix Acetate by is a Prescription medication manufactured, distributed, or labeled by Amphastar Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
DESCRIPTION
Ganirelix Acetate Injection is a synthetic decapeptide with high antagonistic activity against naturally occurring gonadotropin-releasing hormone (GnRH). Ganirelix Acetate is derived from native GnRH with substitutions of amino acids at positions 1, 2, 3, 6, 8, and 10 to form the following molecular formula of the peptide: N-acetyl-3-(2-naphthyl)-D-alanyl-4-chloro-D-phenylalanyl-3-(3-pyridyl)-D-alanyl-L-seryl-L-tyrosyl-N9,N10-diethyl-D-homoarginyl-L-leucyl-N9,N10-diethyl-L-homoarginyl-L-prolyl-D-alanylamide acetate. The molecular weight for Ganirelix Acetate is 1570.4 as an anhydrous free base. The structural formula is as follows:
Ganirelix Acetate
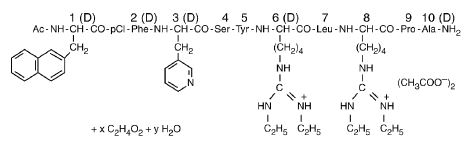
Ganirelix Acetate Injection is supplied as a colorless, sterile, ready-to-use, aqueous solution intended for SUBCUTANEOUS administration only. Each single dose, sterile, prefilled syringe contains 250 mcg/0.5 mL of Ganirelix Acetate, 0.1 mg glacial acetic acid, 23.5 mg mannitol, and water for injection adjusted to pH 5.0 with acetic acid, NF and/or sodium hydroxide, NF.
-
CLINICAL PHARMACOLOGY
The pulsatile release of GnRH stimulates the synthesis and secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH). The frequency of LH pulses in the mid and late follicular phase is approximately 1 pulse per hour. These pulses can be detected as transient rises in serum LH. At midcycle, a large increase in GnRH release results in an LH surge. The midcycle LH surge initiates several physiologic actions including: ovulation, resumption of meiosis in the oocyte, and luteinization. Luteinization results in a rise in serum progesterone with an accompanying decrease in estradiol levels.
Ganirelix Acetate acts by competitively blocking the GnRH receptors on the pituitary gonadotroph and subsequent transduction pathway. It induces a rapid, reversible suppression of gonadotropin secretion. The suppression of pituitary LH secretion by Ganirelix Acetate is more pronounced than that of FSH. An initial release of endogenous gonadotropins has not been detected with Ganirelix Acetate, which is consistent with an antagonist effect. Upon discontinuation of Ganirelix Acetate, pituitary LH and FSH levels are fully recovered within 48 hours.
Pharmacokinetics
The pharmacokinetic parameters of single and multiple injections of Ganirelix Acetate Injection in healthy adult females are summarized in Table I. Steady-state serum concentrations are reached after 3 days of treatment. The pharmacokinetics of Ganirelix Acetate are dose-proportional in the dose range of 125 to 500 mcg.
TABLE I: Mean (SD) pharmacokinetic parameters of 250 mcg of Ganirelix Acetate following a single subcutaneous (SC) injection (n=15) and daily SC injections (n=15) for seven days. tmax
ht1/2
hCmax
ng/mLAUC
ng∙h/mLCL/F
L/hVd/F
Ltmax Time to maximum concentration t1/2 Elimination half-life Cmax Maximum serum concentration AUC Area under the curve; Single dose: AUC0–∞; multiple dose: AUC0–24 Vd Volume of distribution CL Clearance = Dose/AUC0–∞ F Absolute bioavailability - * Based on intravenous administration
Ganirelix Acetate
single dose1.1 (0.3) 12.8 (4.3) 14.8 (3.2) 96 (12) 2.4 (0.2)* 43.7 (11.4)* Ganirelix Acetate
multiple dose1.1 (0.2) 16.2 (1.6) 11.2 (2.4) 77.1 (9.8) 3.3 (0.4) 76.5 (10.3) Absorption
Ganirelix Acetate is rapidly absorbed following subcutaneous injection with maximum serum concentrations reached approximately one hour after dosing. The mean absolute bioavailability of Ganirelix Acetate following a single 250 mcg subcutaneous injection to healthy female volunteers is 91.1%.Distribution
The mean (SD) volume of distribution of Ganirelix Acetate in healthy females following intravenous administration of a single 250-mcg dose is 43.7 (11.4) liters (L). In vitro protein binding to human plasma is 81.9%.Metabolism
Following single-dose intravenous administration of radiolabeled Ganirelix Acetate to healthy female volunteers, Ganirelix Acetate is the major compound present in the plasma (50–70% of total radioactivity in the plasma) up to 4 hours and urine (17.1–18.4% of administered dose) up to 24 hours. Ganirelix Acetate is not found in the feces. The 1–4 peptide and 1–6 peptide of Ganirelix Acetate are the primary metabolites observed in the feces.Excretion
On average, 97.2% of the total radiolabeled Ganirelix Acetate dose is recovered in the feces and urine (75.1% and 22.1%, respectively) over 288 h following intravenous single dose administration of 1 mg [14C]-Ganirelix Acetate. Urinary excretion is virtually complete in 24 h, whereas fecal excretion starts to plateau 192 h after dosing.Special Populations
The pharmacokinetics of Ganirelix Acetate Injection have not been determined in special populations such as geriatric, pediatric, renally impaired and hepatically impaired patients (see PRECAUTIONS).
Drug-Drug Interactions
Formal in vivo or in vitro drug-drug interaction studies have not been conducted (see PRECAUTIONS). Since Ganirelix Acetate can suppress the secretion of pituitary gonadotropins, dose adjustments of exogenous gonadotropins may be necessary when used during controlled ovarian hyperstimulation (COH).Clinical Studies
The efficacy of Ganirelix Acetate Injection was established in two adequate and well-controlled clinical studies which included women with normal endocrine and pelvic ultrasound parameters. The studies intended to exclude subjects with polycystic ovary syndrome (PCOS) and subjects with low or no ovarian reserve. One cycle of study medication was administered to each randomized subject. For both studies, the adminadministration of exogenous recombinant FSH [Follistim® (follitropin beta for injection)] 150 IU daily was initiated on the morning of Day 2 or 3 of a natural menstrual cycle. Ganirelix Acetate Injection was administered on the morning of Day 7 or 8 (Day 6 of recombinant FSH administration). The dose of recombinant FSH administered was adjusted according to individual responses starting on the day of initiation of Ganirelix Acetate. Both recombinant FSH and Ganirelix Acetate were continued daily until at least three follicles were 17 mm or greater in diameter at which time hCG [Pregnyl® (chorionic gonadotropin for injection, USP)] was administered. Following hCG administration, Ganirelix Acetate and recombinant FSH administration were discontinued. Oocyte retrieval, followed by in vitro fertilization (IVF) or intracytoplasmic sperm injection (ICSI), was subsequently performed.
In a multicenter, double-blind, randomized, dose-finding study, the safety and efficacy of Ganirelix Acetate Injection were evaluated for the prevention of LH surges in women undergoing COH with recombinant FSH. Ganirelix Acetate Injection doses ranging from 62.5 mcg to 2000 mcg and recombinant FSH were administered to 332 patients undergoing COH for IVF (see TABLE II). Median serum LH on the day of hCG administration decreased with increasing doses of Ganirelix Acetate. Median serum E2 (17ß-estradiol) on the day of hCG administration was 1475, 1110, and 1160 pg/mL for the 62.5-, 125-, and 250-mcg doses, respectively. Lower peak serum E2 levels of 823, 703, and 441 pg/mL were seen at higher doses of Ganirelix Acetate 500, 1000, and 2000 mcg, respectively. The highest pregnancy and implantation rates were achieved with the 250-mcg dose of Ganirelix Acetate Injection as summarized in Table II.
TABLE II: Results from the multicenter, double-blind, randomized, dose-finding study to assess the efficacy of Ganirelix Acetate Injection to prevent premature LH surges in women undergoing COH with recombinant FSH. Daily dose (mcg) of Ganirelix Acetate Injection 62.5 mcg 125 mcg 250 mcg 500 mcg 1000 mcg 2000 mcg (Protocol 38602) - * ET: Embryo Transfer
- † Following initiation of Ganirelix Acetate therapy. Includes subjects who have complied with daily injections
- ‡ Median values
- § As evidenced by ultrasound at 5–6 weeks following ET
- ¶ Mean (standard deviation)
No. subjects receiving Ganirelix Acetate 31 66 70 69 66 30 No. subjects with ET* 27 61 62 54 61 27 No. of subjects with LH rise ≥ 10 mIU/mL† 4 6 1 0 0 0 Serum LH (mIU/mL) on day of hCG‡ 3.6 2.5 1.7 1.0 0.6 0.3 5th–95th percentiles 0.6–19.9 0.6–11.4 < 0.25–6.4 0.4–4.7 < 0.25–2.2 < 0.25–0.8 Serum E2 (pg/mL) on day of hCG‡ 1475 1110 1160 823 703 441 5th–95th percentiles 645–3720 424–3780 384–3910 279–2720 284–2360 166–1940 Vital pregnancy rate§ per attempt, n (%) 7 (22.6) 17 (25.8) 25 (35.7) 8 (11.6) 9 (13.6) 2 (6.7) per transfer, n (%) 7 (25.9) 17 (27.9) 25 (40.3) 8 (14.8) 9 (14.8) 2 (7.4) Implantation rate (%)¶ 14.2 (26.8) 16.3 (30.5) 21.9 (30.6) 9.0 (23.7) 8.5 (21.7) 4.9 (20.1) Transient LH rises alone were not deleterious to achieving pregnancy with Ganirelix Acetate at doses of 125 mcg (3/6 subjects) and 250 mcg (1/1 subjects). In addition, none of the subjects with LH rises ≥10 mIU/mL had premature luteinization indicated by a serum progesterone above 2 ng/mL.
A multicenter, open-label, randomized study was conducted to assess the efficacy and safety of Ganirelix Acetate Injection in women undergoing COH. Follicular phase treatment with Ganirelix Acetate 250 mcg was studied using a luteal phase GnRH agonist as a reference treatment. A total of 463 subjects were treated with Ganirelix Acetate by subcutaneous injection once daily starting on Day 6 of recombinant FSH treatment. Recombinant FSH was maintained at 150 IU for the first 5 days of ovarian stimulation and was then adjusted by the investigator on the sixth day of gonadotropin use according to individual responses. The results for the Ganirelix Acetate arm are summarized in Table III.
TABLE III: Results from the multicenter, open-label, randomized study to assess the efficacy and safety of Ganirelix Acetate Injection in women undergoing COH. Ganirelix Acetate 250 mcg (Protocol 38607) Some centers were limited to the transfer of ≤ 2 embryos based on local practice standards - * Restricted to subjects with hCG injection
- † Mean (standard deviation)
- ‡ Median values
- § Following initiation of Ganirelix Acetate therapy
- ¶ ET: Embryo Transfer
- # As evidenced by ultrasound at 12–16 weeks following ET
- Þ Includes one patient who achieved pregnancy with intrauterine induction
No. subjects treated 463 Duration of GnRH analog (days)*† 5.4 (2.0) Duration of recombinant FSH (days)*† 9.6 (2.0) Serum E2 (pg/mL) on day of hCG‡
5th–95th percentiles1190
373–3105Serum LH (mIU/mL) on day of hCG‡
5th–95th percentiles1.6
0.6–6.9No. of subjects with LH rise ≥ 10 mIU/mL§ 13 No. of follicles > 11 mm*† 10.7 (5.3) No. of subjects with oocyte retrieval 440 No. of oocytes† 8.7 (5.6) Fertilization rate 62.1% No. subjects with ET¶ 399 No. of embryos transferred† 2.2 (0.6) No. of embryos† 6.0 (4.5) Ongoing pregnancy rate#* per attempt, n (%)Þ 94 (20.3) per transfer, n (%) 93 (23.3) Implantation rate (%)† 15.7 (29) The mean number of days of Ganirelix Acetate treatment was 5.4 (2–14).
LH Surges
The midcycle LH surge initiates several physiologic actions including: ovulation, resumption of meiosis in the oocyte, and luteinization. In 463 subjects administered Ganirelix Acetate Injection 250 mcg, a premature LH surge prior to hCG administration, (LH rise ≥ 10 mIU/mL with a significant rise in serum progesterone > 2 ng/mL, or a significant decline in serum estradiol) occurred in less than 1% of subjects. - INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Ganirelix Acetate Injection is contraindicated under the following conditions:
- Known hypersensitivity to Ganirelix Acetate or to any of its components.
- Known hypersensitivity to GnRH or any other GnRH analog.
- Known or suspected pregnancy (see PRECAUTIONS).
-
WARNINGS
Ganirelix Acetate Injection should be prescribed by physicians who are experienced in infertility treatment. Before starting treatment with Ganirelix Acetate, pregnancy must be excluded. Safe use of Ganirelix Acetate during pregnancy has not been established (see CONTRAINDICATIONS and PRECAUTIONS).
-
PRECAUTIONS
General
Special care should be taken in women with signs and symptoms of active allergic conditions. Cases of hypersensitivity reactions, including anaphylactoid reactions, have been reported, as early as with the first dose, during post-marketing surveillance (see ADVERSE REACTIONS). In the absence of clinical experience, Ganirelix Acetate treatment is not advised in women with severe allergic conditions.
Information for Patients
Prior to therapy with Ganirelix Acetate Injection, patients should be informed of the duration of treatment and monitoring procedures that will be required. The risk of possible adverse reactions should be discussed (see ADVERSE REACTIONS).
Ganirelix Acetate should not be prescribed if the patient is pregnant.
Laboratory Tests
A neutrophil count ≥ 8.3 ( x 109/L) was noted in 11.9% (up to 16.8 x 109/L) of all subjects treated within the adequate and well-controlled clinical trials. In addition, downward shifts within the Ganirelix Acetate Injection group were observed for hematocrit and total bilirubin. The clinical significance of these findings was not determined.
Carcinogenesis and Mutagenesis, Impairment of Fertility
Long-term toxicity studies in animals have not been performed with Ganirelix Acetate Injection to evaluate the carcinogenic potential of the drug. Ganirelix Acetate did not induce a mutagenic response in the Ames test (S. typhimurium and E. coli) or produce chromosomal aberrations in in vitro assay using Chinese Hamster Ovary cells.
Pregnancy
Ganirelix Acetate Injection is contraindicated in pregnant women. When administered from Day 7 to near term to pregnant rats and rabbits at doses up to 10 and 30 mcg/day (approximately 0.4 to 3.2 times the human dose based on body surface area), Ganirelix Acetate increased the incidence of litter resorption. There was no increase in fetal abnormalities. No treatment-related changes in fertility, physical, or behavioral characteristics were observed in the offspring of female rats treated with Ganirelix Acetate during pregnancy and lactation.
The effects on fetal resorption are logical consequences of the alteration in hormonal levels brought about by the antigonadotropic properties of this drug and could result in fetal loss in humans. Therefore, this drug should not be used in pregnant women (see CONTRAINDICATIONS).
-
ADVERSE REACTIONS
The safety of Ganirelix Acetate Injection was evaluated in two randomized, parallel-group, multicenter controlled clinical studies. Treatment duration for Ganirelix Acetate ranged from 1 to 14 days. Table IV represents adverse events (AEs) from first day of Ganirelix Acetate administration until confirmation of pregnancy by ultrasound at an incidence of ≥ 1% in Ganirelix Acetate-treated subjects without regard to causality.
TABLE IV: Incidence of common adverse events (Incidence ≥ 1% in Ganirelix Acetate-treated subjects). Completed controlled clinical studies (All-subjects-treated group). Adverse Events Occurring in ≥ 1% Ganirelix Acetate N=794
% (n)Abdominal Pain (gynecological) 4.8 (38) Death Fetal 3.7 (29) Headache 3.0 (24) Ovarian Hyperstimulation Syndrome 2.4 (19) Vaginal Bleeding 1.8 (14) Injection Site Reaction 1.1 (9) Nausea 1.1 (9) Abdominal Pain (gastrointestinal) 1.0 (8) During post-marketing surveillance, rare cases of hypersensitivity reactions, including anaphylactoid reactions, have been reported, as early as with the first dose (see PRECAUTIONS).
Congenital Anomalies
Ongoing clinical follow-up studies of 283 newborns of women administered Ganirelix Acetate Injection were reviewed. There were three neonates with major congenital anomalies and 18 neonates with minor congenital anomalies. The major congenital anomalies were: hydrocephalus/meningocele, omphalocele, and Beckwith-Wiedemann Syndrome. The minor congenital anomalies were: nevus, skin tags, sacral sinus, hemangioma, torticollis/asymmetric skull, talipes, supernumerary digit finger, hip subluxation, torticollis/high palate, occiput/abnormal hand crease, hernia umbilicalis, hernia inguinalis, hydrocele, undescended testis, and hydronephrosis. The causal relationship between these congenital anomalies and Ganirelix Acetate is unknown. Multiple factors, genetic and others (including, but not limited to ICSI, IVF, gonadotropins, progesterone) may confound ART (Assisted Reproductive Technology) procedures. - OVERDOSAGE
-
DOSAGE AND ADMINISTRATION
After initiating FSH therapy on Day 2 or 3 of the cycle, Ganirelix Acetate Injection 250 mcg may be administered subcutaneously once daily during the mid to late portion of the follicular phase. By taking advantage of endogenous pituitary FSH secretion, the requirement for exogenously administered FSH may be reduced. Treatment with Ganirelix Acetate should be continued daily until the day of hCG administration. When a sufficient number of follicles of adequate size are present, as assessed by ultrasound, final maturation of follicles is induced by administering hCG. The administration of hCG should be withheld in cases where the ovaries are abnormally enlarged on the last day of FSH therapy to reduce the chance of developing OHSS (Ovarian Hyperstimulation Syndrome).
Directions for Using Ganirelix Acetate Injection
- Ganirelix Acetate Injection is supplied in a single dose, sterile, prefilled syringe and is intended for SUBCUTANEOUS administration only.
- Wash hands thoroughly with soap and water.
- The most convenient sites for SUBCUTANEOUS injection are in the abdomen around the navel or upper thigh.
- The injection site should be swabbed with a disinfectant to remove any surface bacteria. Clean about two inches around the point where the needle will be inserted and let the disinfectant dry for at least one minute before proceeding.
- With syringe held upward, remove needle cover.
- Pinch up a large area of skin between the finger and thumb. Vary the injection site a little with each injection.
- The needle should be inserted at the base of the pinched-up skin at an angle of 45-90° to the skin surface.
- When the needle is correctly positioned, it will be difficult to draw back on the plunger. If any blood is drawn into the syringe, the needle tip has penetrated a vein or artery. If this happens, withdraw the needle slightly and reposition the needle without removing it from the skin. Alternatively, remove the needle and use a new, sterile, prefilled syringe. Cover the injection site with a swab containing disinfectant and apply pressure; the site should stop bleeding within one or two minutes.
- Once the needle is correctly placed, depress the plunger slowly and steadily, so the solution is correctly injected and the skin is not damaged.
- Pull the syringe out quickly and apply pressure to the site with a swab containing disinfectant.
- Use the sterile, prefilled syringe only once. Discard the unused portion and dispose of it properly.
-
HOW SUPPLIED
Ganirelix Acetate Injection is supplied in:
Disposable, ready for use, single dose, sterile, prefilled 1 mL glass syringes containing 250 mcg/0.5 mL
aqueous solution of Ganirelix Acetate closed with a rubber piston that is not made of natural rubber latex.
Each Ganirelix Acetate sterile, prefilled syringe is affixed with a 27 gauge x ½-inch needle closed by a
needle shield. (See PRECAUTIONS, General.) This syringe is not made with natural rubber latex.Single syringe NDC: 0548-5001-00 Stock No. 5001
Storage
Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [see USP Controlled Room Temperature]. Protect from light. - SPL UNCLASSIFIED SECTION
- Carton
-
INGREDIENTS AND APPEARANCE
GANIRELIX ACETATE
ganirelix acetate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0548-5001 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GANIRELIX ACETATE (UNII: 56U7906FQW) (GANIRELIX - UNII:IX503L9WN0) GANIRELIX ACETATE 250 ug in 0.5 mL Inactive Ingredients Ingredient Name Strength ACETIC ACID (UNII: Q40Q9N063P) 0.1 mg in 0.5 mL MANNITOL (UNII: 3OWL53L36A) 23.5 mg in 0.5 mL WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0548-5001-00 1 in 1 CARTON 06/20/2022 1 0.5 mL in 1 SYRINGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA212613 06/20/2022 Labeler - Amphastar Pharmaceuticals, Inc. (024736733) Establishment Name Address ID/FEI Business Operations Amphastar Pharmaceuticals, Inc. 024736733 manufacture(0548-5001) , analysis(0548-5001)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
