Chlorzoxazone by AXIS Pharmaceutics Inc CHLORZOXAZONE tablet
Chlorzoxazone by
Drug Labeling and Warnings
Chlorzoxazone by is a Prescription medication manufactured, distributed, or labeled by AXIS Pharmaceutics Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
DESCRIPTION
Chlorzoxazone USP is a centrally acting skeletal muscle relaxant, available as tablets of 500 mg for oral administration. Its chemical name is 5-Chloro-2-benzoxazolinone, and its structural formula is:
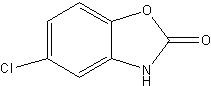
C7H4CINO2
MW 169.57
Chlorzoxazone USP is a white or practically white, practically odorless, crystalline powder. Chlorzoxazone is slightly soluble in water; sparingly soluble in alcohol, in isopropyl alcohol, and in methanol; soluble in solutions of alkali hydroxides and ammonia.
Chlorzoxazone tablets contain the inactive ingredients Pregelatinized Starch, Croscarmellose Sodium, Microcrystalline Cellulose, Docusate Sodium, Sodium Benzoate, Colloidal Silicon dioxide, Magnesium Stearate.
-
CLINICAL PHARMACOLOGY
Chlorzoxazone is a centrally-acting agent for painful musculoskeletal conditions. Data available from animal experiments as well as human study indicate that chlorzoxazone acts primarily at the level of the spinal cord and subcortical areas of the brain where it inhibits multisynaptic reflex arcs involved in producing and maintaining skeletal muscle spasm of varied etiology. The clinical result is a reduction of the skeletal muscle spasm with relief of pain and increased mobility of the involved muscles. Blood levels of chlorzoxazone can be detected in people during the first 30 minutes and peak levels may be reached, in the majority of the subjects, in about 1 to 2 hours after oral administration of chlorzoxazone. Chlorzoxazone is rapidly metabolized and is excreted in the urine, primarily in a conjugated form as the glucuronide. Less than one percent of a dose of chlorzoxazone is excreted unchanged in the urine in 24 hours.
-
INDICATIONS & USAGE
Chlorzoxazone is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute, painful musculoskeletal conditions. The mode of action of this drug has not been clearly identified, but may be related to its sedative properties. Chlorzoxazone does not directly relax tense skeletal muscles in man.
- CONTRAINDICATIONS
-
WARNINGS
Serious (including fatal) hepatocellular toxicity has been reported rarely in patients receiving chlorzoxazone. The mechanism is unknown but appears to be idiosyncratic and unpredictable. Factors predisposing patients to this rare event are not known. Patients should be instructed to report early signs and/or symptoms of hepatotoxicity such as fever, rash, anorexia, nausea, vomiting, fatigue, right upper quadrant pain, dark urine, or jaundice. Chlorzoxazone should be discontinued immediately and a physician consulted if any of these signs or symptoms develop. Chlorzoxazone use should also be discontinued if a patient develops abnormal liver enzymes (e.g., AST, ALT, alkaline phosphatase and bilirubin.)
The concomitant use of alcohol or other central nervous system depressants may have an additive effect.
Usage in Pregnancy
The safe use of chlorzoxazone has not been established with respect to the possible adverse effects upon fetal development. Therefore, it should be used in women of childbearing potential only when, in the judgment of the physician, the potential benefits outweigh the possible risks.
-
PRECAUTIONS
Chlorzoxazone should be used with caution in patients with known allergies or with a history of allergic reactions to drugs. If a sensitivity reaction occurs such as urticaria, redness, or itching of the skin, the drug should be stopped.
If any symptoms suggestive of liver dysfunction are observed, the drug should be discontinued -
ADVERSE REACTIONS
Chlorzoxazone containing products are usually well tolerated. It is possible in rare instances that chlorzoxazone may have been associated with gastrointestinal bleeding. Drowsiness, dizziness, lightheadedness, malaise, or over-stimulation may be noted by an occasional patient. Rarely, allergic- type skin rashes, petechiae, or ecchymoses may develop during treatment. Angioneurotic edema or anaphylactic reactions are extremely rare. There is no evidence that the drug will cause renal damage. Rarely, a patient may note discoloration of the urine resulting from a phenolic metabolite of chlorzoxazone. This finding is of no known clinical significance.
To report SUSPECTED ADVERSE EVENTS, contact AXIS Pharmaceutics Inc at 1-800-906- 6939 or FDA at 1-800- FDA-1088 or http://www.fda.gov/ for voluntary reporting of adverse reactions.
-
OVERDOSAGE
Symptoms
Initially, gastrointestinal disturbances such as nausea, vomiting, or diarrhea together with drowsiness, dizziness, lightheadedness or headache may occur. Early in the course there may be malaise or sluggishness followed by marked loss of muscle tone, making voluntary movement impossible. The deep tendon reflexes may be decreased or absent. The sensorium remains intact, and there is no peripheral loss of sensation. Respiratory depression may occur with rapid, irregular respiration and intercostal and substernal retraction. The blood pressure is lowered, but shock has not been observed.
Treatment
Gastric lavage or induction of emesis should be carried out, followed by administration of activated charcoal. Thereafter, treatment is entirely supportive. If respirations are depressed, oxygen and artificial respiration should be employed and a patent airway assured by use of an oropharyngeal airway or endotracheal tube. Hypotension may be counteracted by use of dextran, plasma, concentrated albumin or a vasopressor agent such as norepinephrine. Cholinergic drugs or analeptic drugs are of no value and should not be used
- DOSAGE & ADMINISTRATION
-
HOW SUPPLIED
Chlorzoxazone Tablets, USP 500 mg for Oral administration are supplied as follows,
White to off-white capsule shaped tablets debossed with N085 on the bisected side:NDC 73007-013-03 100 Tablets
NDC: 73007-013-02 200 Tablets
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense contents with a child-resistant closure (as required) and in a tight container as defined in the USP/NF.
Manufactured for:
AXIS Pharmaceutics Inc
118 ADAMS ST STE 304B,
FAIRMONT, WV, 26554
Revised 01/2020 - Product label
-
INGREDIENTS AND APPEARANCE
CHLORZOXAZONE
chlorzoxazone tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 73007-013 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORZOXAZONE (UNII: H0DE420U8G) (CHLORZOXAZONE - UNII:H0DE420U8G) CHLORZOXAZONE 500 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) DOCUSATE SODIUM (UNII: F05Q2T2JA0) SODIUM BENZOATE (UNII: OJ245FE5EU) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color WHITE (White to off white) Score 2 pieces Shape CAPSULE (Capsule shaped tablet) Size 17mm Flavor Imprint Code n085 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73007-013-03 100 in 1 BOTTLE; Type 0: Not a Combination Product 01/22/2020 2 NDC: 73007-013-02 200 in 1 BOTTLE; Type 0: Not a Combination Product 01/22/2020 03/16/2020 3 NDC: 73007-013-04 2 in 1 PACKAGE 01/22/2020 3 100 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA212254 01/22/2020 Labeler - AXIS Pharmaceutics Inc (116938414)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
