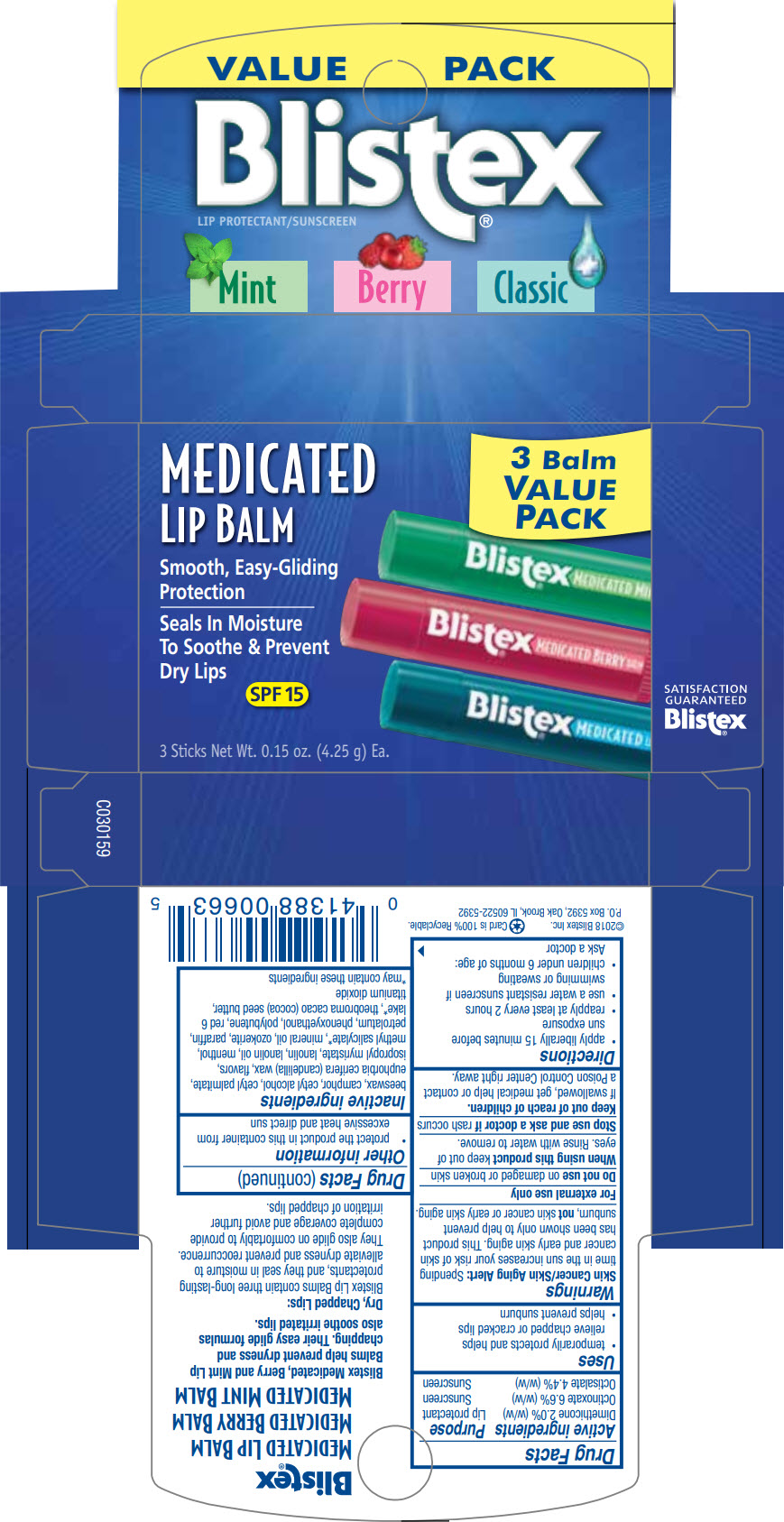
BLISTEX ASSORTED MEDICATED BALM VALUE PACK (BLISTEX MEDICATED BERRY LIP BALM, BLISTEX MEDICATED LIP BALM, BLISTEX MEDICATED MINT LIP BALM)- dimethicone, padimate o, oxybenzone, dimethicone, octinoxate, and octisalate
Blistex Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
| Active ingredients | Purpose |
| Dimethicone 2.0% (w/w) | Lip protectant |
| Octinoxate 6.6% (w/w) | Sunscreen |
| Octisalate 4.4% (w/w) | Sunscreen |
Uses
- temporarily protects and helps relieve chapped or cracked lips
- helps prevent sunburn
Warnings
Skin Cancer/Skin Aging Alert
Spending time in the sun increases your risk of skin cancer and early skin aging. This product has been shown only to help prevent sunburn, not skin cancer or early skin aging.
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash occurs
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- apply liberally 15 minutes before sun exposure
- reapply at least every 2 hours
- use a water resistant sunscreen if swimming or sweating
- children under 6 months of age: Ask a doctor
Other information
- protect the product in this container from excessive heat and direct sun
Inactive ingredients
beeswax, camphor, cetyl alcohol, cetyl palmitate, euphorbia cerifera (candelilla) wax, flavors, isopropyl myristate, lanolin, lanolin oil, menthol, methyl salicylate1, mineral oil, ozokerite, paraffin, petrolatum, phenoxyethanol, polybutene, red 6 lake1, theobroma cacao (cocoa) seed butter, titanium dioxide
1
may contain these ingredients
PRINCIPAL DISPLAY PANEL - Kit Carton
VALUE PACK
Blistex®
LIP PROTECTANT/SUNSCREEN
Mint Berry Classic
MEDICATED
LIP BALM
3 Balm
VALUE
PACK
Smooth, Easy-Gliding
Protection
Seals In Moisture
To Soothe & Prevent
Dry Lips
SPF 15
3 Sticks Net Wt. 0.15 oz. (4.25 g) Ea.