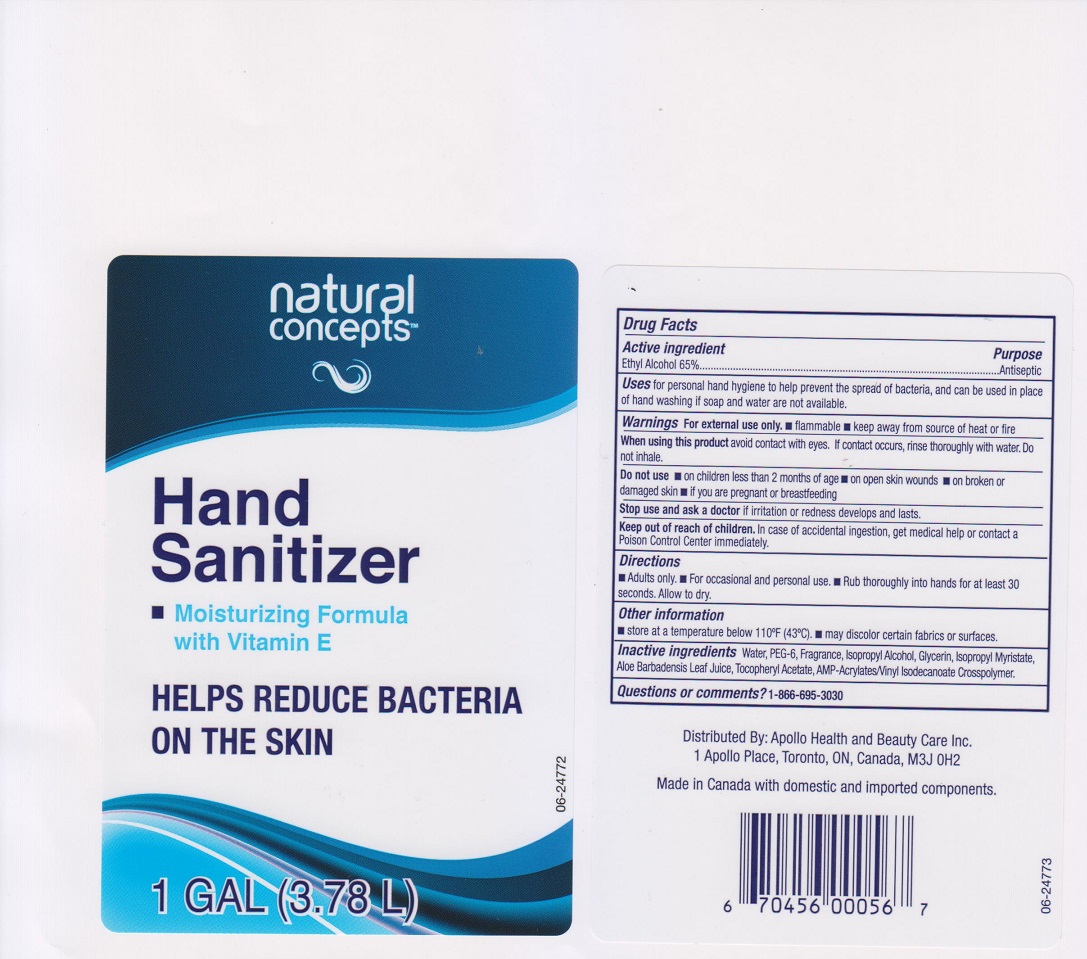
Natural Concepts Hand Sanitizer by Apollo Health and Beauty Care Inc. Drug Facts
Natural Concepts Hand Sanitizer by
Drug Labeling and Warnings
Natural Concepts Hand Sanitizer by is a Otc medication manufactured, distributed, or labeled by Apollo Health and Beauty Care Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
NATURAL CONCEPTS HAND SANITIZER- ethyl alcohol gel
Apollo Health and Beauty Care Inc.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Drug Facts
Uses
for personal hand hygiene to help prevent the spread of bacteria, and can be used in place of hand washing if soap and water are not available.
Warnings
For external use only
- flammable
- keep away from source of heat or fire
Directions
- Adults only
- for occasional and personal use
- rub thoroughly into hands for at least 30 seconds. Allow to dry.
Other information
- store at a temperature below 110°F (43°C)
- may discolor certain fabrics or surfaces
| NATURAL CONCEPTS HAND SANITIZER
ethyl alcohol gel |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Apollo Health and Beauty Care Inc. (201901209) |
| Registrant - Apollo Health and Beauty Care Inc. (201901209) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Apollo Health and Beauty Care Inc. | 201901209 | manufacture(63148-516) | |
Revised: 2/2025
Document Id: 2d180956-5f20-0491-e063-6394a90abdbf
Set id: 9f7abd46-dfa1-4893-a248-afced94686db
Version: 2
Effective Time: 20250201