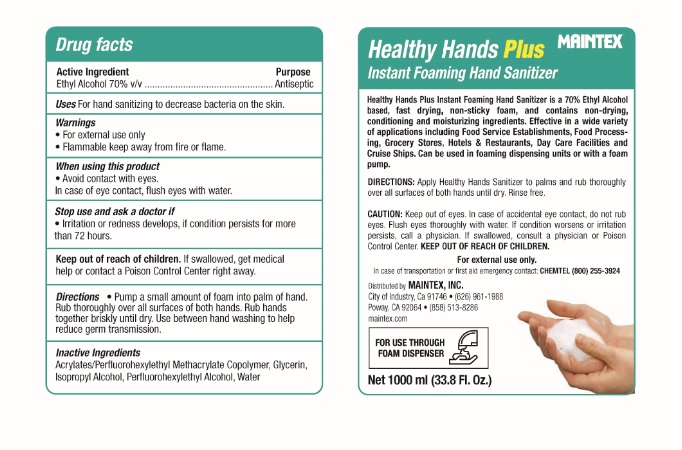
Healthy Hands Plus by Maintex Healthy Hands Plus
Healthy Hands Plus by
Drug Labeling and Warnings
Healthy Hands Plus by is a Otc medication manufactured, distributed, or labeled by Maintex. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
HEALTHY HANDS PLUS- ethyl alcohol liquid
Maintex
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Healthy Hands Plus
Inactive Ingredients
Acrylates/Perfluorohexylethyl Methacrylate Copolymer, Glycerin, Isopropyl Alcohol, Perfluorohexylethyl Alcohol, Water
Stop use and ask a doctor if
-Irritation or redness develops, if condition persists for more than 72 hours.
Keep out of reach of children.
Keep out of reach of children. If swallowed, get medical help
or contact a Poison Control Center immediately.
| HEALTHY HANDS PLUS
ethyl alcohol liquid |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Maintex (008317307) |
| Registrant - Maintex (008317307) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Maintex | 008317307 | manufacture(77617-015) , api manufacture(77617-015) , pack(77617-015) | |
Revised: 1/2022
Document Id: e9842f3d-d6f7-46f5-9ad8-4f687927defc
Set id: a0636b48-7002-4502-9e3f-6a973b9d9d2b
Version: 5
Effective Time: 20220118
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.