5% Minoxidil Topical Solution
5% Minoxidil Topical Solution by
Drug Labeling and Warnings
5% Minoxidil Topical Solution by is a Otc medication manufactured, distributed, or labeled by Gabar Health Sciences Corp.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
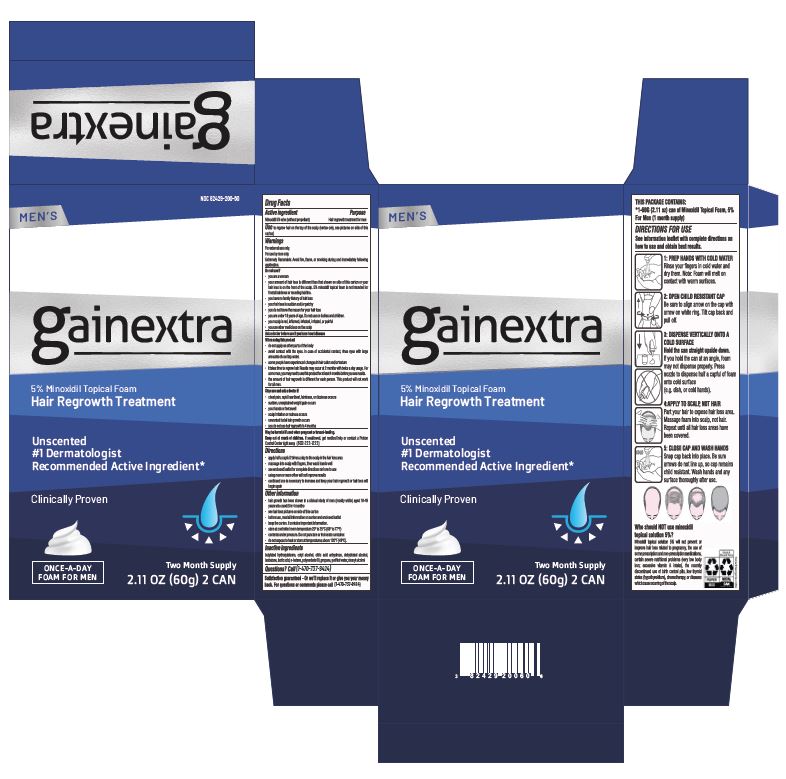
5% MINOXIDIL TOPICAL SOLUTION- minoxidil solution
Gabar Health Sciences Corp.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
5% Minoxidil Topical Solution
Warnings
For external use only.
For use by men only.
Extremely Flammable: Avoid fire, flame, or smoking during and immediately following application.
Do not use if
- you are a woman
- your amount of hair loss is different than that shown on side of this carton or your hair loss is on the front of the scalp 5% minoxidil topical foam is not intended for frontal baldmess or receding hairline.
- you have no family history of hair loss
- your hair loss is sudden and/or patchy
- you do not know the reason for your hair loss
- you are under 18 years of age. Do not use on babies and children.
- your scalp is red, inflamed, infected, irritated, or painful
- you use other medicines on the scalp
When using this product
- do not apply on other parts of the body
- avoid contact with the eyes. In case of accidental contact, rinse eyes with large amounts of cool tap water.
- some people have experienced changes in hair color and/or texture
- it takes time to regrow hair. Results may occur at 2 months with twice a day usage. For some men, you may need to use this product for at least 4 months before you see results
- the amount of hair regrowth is different for each person. This product will not work for all men.
Stop use and ask a doctor if
- chest pain, rapid heartbeat, faintness, or dizziness occurs
- sudden, unexplained weight gain occurs
- your hands or feet swell
- scalp irritation or redness occurs
- unwanted facial hair growth occurs
- you do not see hair regrowth in 4 months
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Directions
- apply half a capful 2 times a day to the scalp in the hair loss area
- massage into scalp with fingers, then wash hands well
- see enclosed leaflet for complete directons on how to use
- using more or more often will not improve results
- continued use is necessary to increase and keep your hair regrowth or hair loss will begin again
Other information
- hair growth has been shown in a clinical study of men (mostly white) aged 18-49 years who used it for 4 months
- see hair loss picture on side of this carton
- before use, read all information on carton and enclosed leaflet
- keep the carton. It contains important information.
- store at controlled room temperature 20º to 25ºC (68º to 77ºF)
- contents under pressure. Do not puncture or incinerate container.
- do not expose to heat or store at temperature above 120ºF (49ºC).
Inactive ingredients
butylated hydroxytoluene, cetyl alcohol, citric acid anhydrous, dehydrated alcohol, isobutane, lactic acid, n-butane, polysorbate 60, propane, purified water, stearyl alcohol
Questions?
- Call 1-470-737-9424
Manufactured and distributed by:
Gabar Health Sciences Corp., Atlanta, GA 30385
PRINCIPAL DISPLAY PANEL
Minoxidil Topical Aerosol, 5% (For Men)
HAIR REGROWTH TREATMENT
Reactivates hair follicles to stimulate regrowth
Clinically proven to help regrow hair
#1 Dermatologist Recommended Active Ingredient
Easy-to-use-foam unscented
3 MONTH SUPPLY
3 - 60 g (2.11 OZ) CANS
NOT FOR USE BY WOMEN
| 5% MINOXIDIL TOPICAL SOLUTION
minoxidil solution |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - Gabar Health Sciences Corp. (118401847) |
| Registrant - Gabar Health Sciences Corp. (118401847) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Gabar Health Sciences Corp. | 118401847 | manufacture(82429-200) | |