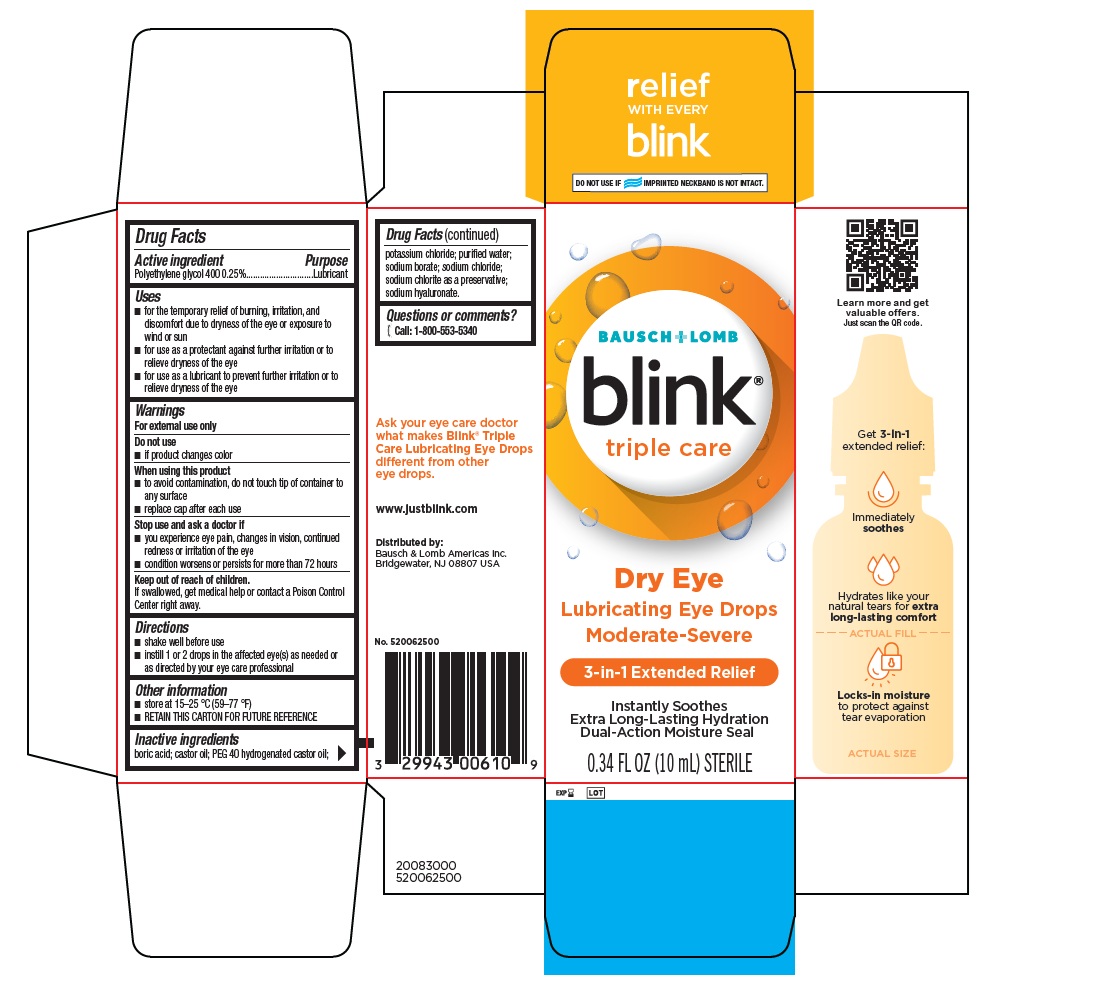
BLINK TRIPLE CARE- polyethylene glycol 400 solution/ drops
BLINK TRIPLE CARE by
Drug Labeling and Warnings
BLINK TRIPLE CARE by is a Otc medication manufactured, distributed, or labeled by Johnson & Johnson Surgical Vision, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Uses
-
Warnings
For external use only
Do not use
- if product changes color
When using this product
- to avoid contamination, do not touch tip of container to any surface
- replace cap after each use
- Directions
- Other Information
- Inactive Ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BLINK TRIPLE CARE
polyethylene glycol 400 solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 29943-006 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) (POLYETHYLENE GLYCOL 400 - UNII:B697894SGQ) POLYETHYLENE GLYCOL 400 2.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength BORIC ACID (UNII: R57ZHV85D4) CASTOR OIL (UNII: D5340Y2I9G) POLYOXYL 40 HYDROGENATED CASTOR OIL (UNII: 7YC686GQ8F) POTASSIUM CHLORIDE (UNII: 660YQ98I10) WATER (UNII: 059QF0KO0R) SODIUM BORATE (UNII: 91MBZ8H3QO) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CHLORITE (UNII: G538EBV4VF) HYALURONATE SODIUM (UNII: YSE9PPT4TH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 29943-006-10 10 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 05/01/2022 2 NDC: 29943-006-25 2.5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 05/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M018 05/01/2022 Labeler - Bausch & Lomb Incorporated (196603781) Establishment Name Address ID/FEI Business Operations Bausch & Lomb Incorporated Greenville, South Carolina 114406598 manufacture(29943-006)
Trademark Results [BLINK TRIPLE CARE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 BLINK TRIPLE CARE 88818805 not registered Live/Pending |
JOHNSON & JOHNSON SURGICAL VISION, INC. 2020-03-03 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.