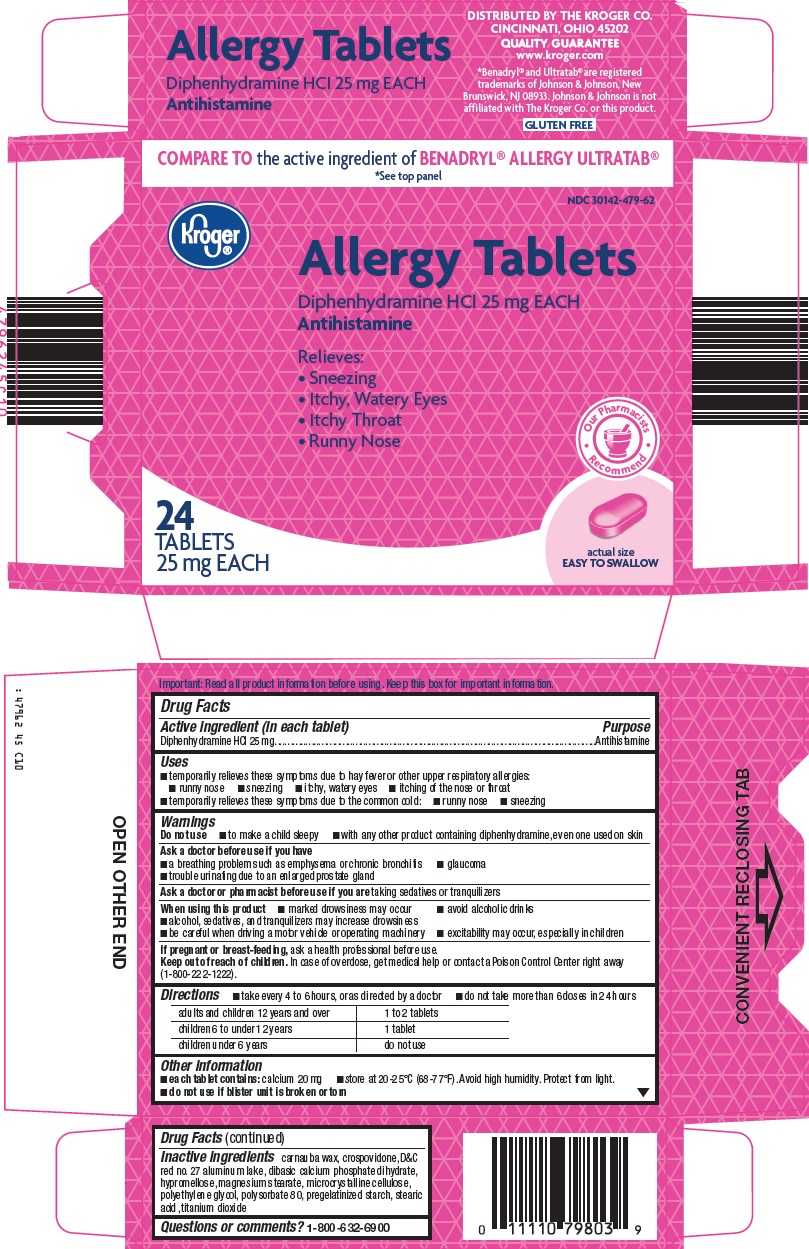
ALLERGY- diphenhydramine hydrochloride tablet
Allergy by
Drug Labeling and Warnings
Allergy by is a Otc medication manufactured, distributed, or labeled by Kroger Company. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ALLERGY
diphenhydramine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 30142-479 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 25 mg Inactive Ingredients Ingredient Name Strength CARNAUBA WAX (UNII: R12CBM0EIZ) CROSPOVIDONE (15 MPA.S AT 5%) (UNII: 68401960MK) D&C RED NO. 27 (UNII: 2LRS185U6K) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) Product Characteristics Color PINK (dark) Score no score Shape OVAL Size 10mm Flavor Imprint Code L479 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 30142-479-62 24 in 1 CARTON 09/15/1987 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 30142-479-67 48 in 1 CARTON 08/05/2011 2 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC: 30142-479-78 1 in 1 CARTON 08/30/2001 3 100 in 1 BOTTLE; Type 0: Not a Combination Product 4 NDC: 30142-479-79 1 in 1 CARTON 04/13/2018 4 400 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 09/15/1987 Labeler - Kroger Company (006999528)
Trademark Results [Allergy]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ALLERGY 97069656 not registered Live/Pending |
Rodriguez, Kent J 2021-10-12 |
 ALLERGY 90819383 not registered Live/Pending |
Rodriguez, Kent J 2021-07-09 |
 ALLERGY 87534443 5406627 Live/Registered |
RSM Medical Inc. 2017-07-19 |
 ALLERGY 87518600 5400551 Live/Registered |
RSM Medical Inc. 2017-07-06 |
 ALLERGY 74392251 not registered Dead/Abandoned |
Danta, Inc. 1993-05-18 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.