AMBRISENTAN tablet, film coated
ambrisentan by
Drug Labeling and Warnings
ambrisentan by is a Prescription medication manufactured, distributed, or labeled by Sun Pharmaceutical Industries, Inc., Sun Pharmaceutical Industries Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use AMBRISENTAN TABLETS safely and effectively. See full prescribing information for AMBRISENTAN TABLETS.
AMBRISENTAN tablets, for oral use
Initial U.S. Approval: 2007WARNING: EMBRYO-FETAL TOXICITY
See full prescribing information for complete boxed warning.
- Do not administer ambrisentan to a pregnant female because it may cause fetal harm (4.1, 5.1, 8.1).
- Females of reproductive potential: Exclude pregnancy before the start of treatment, monthly during treatment, and 1 month after stopping treatment. Prevent pregnancy during treatment and for one month after stopping treatment by using acceptable methods of contraception (2.2, 8.3).
- Because of the risk of embryo-fetal toxicity, for all female patients, ambrisentan is only available through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called Ambrisentan REMS (5.2).
INDICATIONS AND USAGE
Ambrisentan is an endothelin receptor antagonist indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1): (1)
- To improve exercise ability and delay clinical worsening.
Studies establishing effectiveness included trials predominantly in patients with WHO Functional Class II–III symptoms and etiologies of idiopathic or heritable PAH (60%) or PAH associated with connective tissue diseases (34%). (1)
DOSAGE AND ADMINISTRATION
Initiate treatment at 5 mg once daily (2.1).
Titrate at 4-week intervals as needed and tolerated (2.1).
Do not split, crush, or chew tablets (2.1). (2)
DOSAGE FORMS AND STRENGTHS
Tablet: 5 mg and 10 mg (3)
CONTRAINDICATIONS
Pregnancy (4.1)
Idiopathic Pulmonary Fibrosis (4.2) (4)
WARNINGS AND PRECAUTIONS
- Fluid retention may require intervention (5.3).
- If patients develop acute pulmonary edema during initiation of therapy with ambrisentan, consider underlying pulmonary veno-occlusive disease and discontinue treatment if necessary (5.4).
- Decreases in sperm count have been observed in patients taking endothelin receptor antagonists (5.5).
- Decreases in hemoglobin have been observed within the first few weeks; measure hemoglobin at initiation, at 1 month, and periodically thereafter (5.6).
ADVERSE REACTIONS
Most common adverse reactions (>3% compared to placebo) are peripheral edema, nasal congestion, sinusitis, and flushing (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Sun Pharmaceutical Industries, Inc. at 1-800-818-4555 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. (6)
DRUG INTERACTIONS
Cyclosporine increases ambrisentan exposure; limit ambrisentan dose to 5 mg once daily . (7)
USE IN SPECIFIC POPULATIONS
Breastfeeding: Choose ambrisentan or breastfeeding (8.2).
Not recommended in patients with moderate or severe hepatic impairment (8.7). (8)See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 10/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: EMBRYO-FETAL TOXICITY
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Adult Dosage
2.2 Pregnancy Testing in Females of Reproductive Potential
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 Pregnancy
4.2 Idiopathic Pulmonary Fibrosis
5 WARNINGS AND PRECAUTIONS
5.1 Embryo-fetal Toxicity
5.2 Ambrisentan -Risk Evaluation and Mitigation Strategy (REMS)
5.3 Fluid Retention
5.4 Pulmonary Edema with Pulmonary Veno-occlusive Disease (PVOD)
5.5 Decreased Sperm Counts
5.6 Hematological Changes
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Pulmonary Arterial Hypertension (PAH)
14.3 Long-term Treatment of PAH
14.4 Adverse Effects in Idiopathic Pulmonary Fibrosis (IPF)
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: EMBRYO-FETAL TOXICITY
Do not administer ambrisentan to a pregnant female because it may cause fetal harm. Ambrisentan is very likely to produce serious birth defects if used by pregnant females, as this effect has been seen consistently when it is administered to animals [see Contraindications (4.1), Warnings and Precautions (5.1), and Use in Specific Populations (8.1)].
Exclude pregnancy before the initiation of treatment with ambrisentan. Females of reproductive potential must use acceptable methods of contraception during treatment with ambrisentan and for one month after treatment. Obtain monthly pregnancy tests during treatment and 1 month after discontinuation of treatment [see Dosage and Administration (2.2) and Use in Specific Populations (8.3)].
Because of the risk of embryo-fetal toxicity, for all female patients, ambrisentan is only available through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the Ambrisentan REMS [see Warnings and Precautions (5.2)].
-
1 INDICATIONS AND USAGE
Ambrisentan is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1):
To improve exercise ability and delay clinical worsening.
Studies establishing effectiveness included predominantly patients with WHO Functional Class II–III symptoms and etiologies of idiopathic or heritable PAH (60%) or PAH associated with connective tissue diseases (34%). - 2 DOSAGE AND ADMINISTRATION
-
3 DOSAGE FORMS AND STRENGTHS
5 mg and 10 mg film-coated tablets for oral administration
- Each 5 mg tablet is pale pink colored, round shape, biconvex beveled edge film-coated tablet debossed with “236” on one side and plain on the other side.
- Each 10 mg tablet is deep pink colored, oval shape, biconvex film-coated tablet debossed with “237” on one side and plain on the other side.
-
4 CONTRAINDICATIONS
4.1 Pregnancy
Ambrisentan may cause fetal harm when administered to a pregnant female. Ambrisentan is contraindicated in females who are pregnant. Ambrisentan was consistently shown to have teratogenic effects when administered to animals. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to a fetus [see Warnings and Precautions (5.1, 5.2) and Use in Specific Populations (8.1)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Embryo-fetal Toxicity
Ambrisentan may cause fetal harm when administered during pregnancy and is contraindicated for use in females who are pregnant. In females of reproductive potential, exclude pregnancy prior to initiation of therapy, ensure use of acceptable contraceptive methods, and obtain monthly pregnancy tests [see Dosage and Administration (2.2), and Use in Specific Populations (8.1, 8.3)].
Ambrisentan is only available for females through a restricted program under a REMS [see Warnings and Precautions (5.2)].
5.2 Ambrisentan -Risk Evaluation and Mitigation Strategy (REMS)
For all females, ambrisentan is available only through a restricted program under a REMS called the Ambrisentan REMS, because of the risk of embryo-fetal toxicity [see Contraindications (4.1), Warnings and Precautions (5.1), and Use in Specific Populations (8.1, 8.3)].
Important requirements of the Ambrisentan REMS include the following:
- Prescribers must be certified with the Ambrisentan REMS by enrolling and completing training.
- All females, regardless of reproductive potential, must enroll in the Ambrisentan REMS prior to initiating ambrisentan. Male patients are not enrolled in the REMS.
- Females of reproductive potential must comply with the pregnancy testing and contraception requirements [see Use in Specific Populations (8.3)].
- Pharmacies that dispense ambrisentan must be certified with the Ambrisentan REMS and must dispense to female patients who are authorized to receive ambrisentan.
Further information is available at www.ambrisentanrems.us.com or 1-888-417-3172.
5.3 Fluid Retention
Peripheral edema is a known class effect of endothelin receptor antagonists, and is also a clinical consequence of PAH and worsening PAH. In the placebo-controlled studies, there was an increased incidence of peripheral edema in patients treated with doses of 5 mg or 10 mg ambrisentan compared to placebo [see Adverse Reactions (6.1)]. Most edema was mild to moderate in severity.
In addition, there have been postmarketing reports of fluid retention in patients with pulmonary hypertension, occurring within weeks after starting ambrisentan. Patients required intervention with a diuretic, fluid management, or, in some cases, hospitalization for decompensating heart failure.
If clinically significant fluid retention develops, with or without associated weight gain, further evaluation should be undertaken to determine the cause, such as ambrisentan or underlying heart failure, and the possible need for specific treatment or discontinuation of ambrisentan therapy.
5.4 Pulmonary Edema with Pulmonary Veno-occlusive Disease (PVOD)
If patients develop acute pulmonary edema during initiation of therapy with vasodilating agents such as ambrisentan, the possibility of PVOD should be considered, and if confirmed ambrisentan should be discontinued.
5.5 Decreased Sperm Counts
Decreased sperm counts have been observed in human and animal studies with another endothelin receptor antagonist and in animal fertility studies with ambrisentan. Ambrisentan may have an adverse effect on spermatogenesis. Counsel patients about potential effects on fertility [see Use in Specific Populations (8.6) and Nonclinical Toxicology (13.1)].
5.6 Hematological Changes
Decreases in hemoglobin concentration and hematocrit have followed administration of other endothelin receptor antagonists and were observed in clinical studies with ambrisentan. These decreases were observed within the first few weeks of treatment with ambrisentan, and stabilized thereafter. The mean decrease in hemoglobin from baseline to end of treatment for those patients receiving ambrisentan in the 12-week placebo-controlled studies was 0.8 g/dL.
Marked decreases in hemoglobin (>15% decrease from baseline resulting in a value below the lower limit of normal) were observed in 7% of all patients receiving ambrisentan (and 10% of patients receiving 10 mg) compared to 4% of patients receiving placebo. The cause of the decrease in hemoglobin is unknown, but it does not appear to result from hemorrhage or hemolysis.
In the long-term open-label extension of the two pivotal clinical studies, mean decreases from baseline (ranging from 0.9 to 1.2 g/dL) in hemoglobin concentrations persisted for up to 4 years of treatment.
There have been postmarketing reports of decreases in hemoglobin concentration and hematocrit that have resulted in anemia requiring transfusion.
Measure hemoglobin prior to initiation of ambrisentan, at one month, and periodically thereafter. Initiation of ambrisentan therapy is not recommended for patients with clinically significant anemia. If a clinically significant decrease in hemoglobin is observed and other causes have been excluded, consider discontinuing ambrisentan.
-
6 ADVERSE REACTIONS
Clinically significant adverse reactions that appear in other sections of the labeling include:
- Embryo-fetal Toxicity [see Warnings and Precautions (5.1),Use in Specific Populations (8.1)]
- Fluid Retention [see Warnings and Precautions (5.3)]
- Pulmonary Edema with PVOD [see Warnings and Precautions (5.4)]
- Decreased Sperm Count [see Warnings and Precautions (5.5)]
- Hematologic Changes [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Safety data for ambrisentan are presented from two 12-week, placebo-controlled studies (ARIES-1 and ARIES-2) in patients with pulmonary arterial hypertension (PAH). The exposure to ambrisentan in these studies ranged from 6 days to 100 days.
In ARIES-1 and ARIES-2, a total of 261 patients received ambrisentan at doses of 2.5mg, 5mg, or 10mg once daily and 132 patients received placebo. The adverse reactions that occurred in >3% more patients receiving ambrisentan than receiving placebo are shown in Table 1.
Table 1 Adverse Reactions with Placebo-Adjusted Rates >3% in ARIES-1 and ARIES-2Placebo
(N=132)
Ambrisentan
(N=261)
Adverse Reaction
n (%)
n (%)
Placebo-adjusted (%)
Peripheral edema
14 (11)
45 (17)
6
Nasal congestion
2 (2)
15 (6)
4
Sinusitis
0 (0)
8 (3)
3
Flushing
1 (1)
10 (4)
3
Most adverse drug reactions were mild to moderate and only nasal congestion was dose-dependent.
Few notable differences in the incidence of adverse reactions were observed for patients by age or sex. Peripheral edema was similar in younger patients (<65 years) receiving ambrisentan (14%; 29/205) or placebo (13%; 13/104), and was greater in elderly patients (≥65 years) receiving ambrisentan (29%; 16/56) compared to placebo (4%; 1/28). The results of such subgroup analyses must be interpreted cautiously.
The incidence of treatment discontinuations due to adverse events other than those related to PAH during the clinical trials in patients with PAH was similar for ambrisentan (2%; 5/261 patients) and placebo (2%; 3/132 patients). The incidence of patients with serious adverse events other than those related to PAH during the clinical trials in patients with PAH was similar for placebo (7%; 9/132 patients) and for ambrisentan (5%; 13/261 patients).
During 12-week controlled clinical trials, the incidence of aminotransferase elevations >3 x upper limit of normal (ULN) were 0% on ambrisentan and 2.3% on placebo. In practice, cases of hepatic injury should be carefully evaluated for cause.
Use in Patients with Prior Endothelin Receptor Antagonist (ERA) Related Serum Liver Enzyme AbnormalitiesIn an uncontrolled, open-label study, 36 patients who had previously discontinued endothelin receptor antagonists (ERAs: bosentan, an investigational drug, or both) due to aminotransferase elevations >3 x ULN were treated with ambrisentan. Prior elevations were predominantly moderate, with 64% of the ALT elevations <5 x ULN, but 9 patients had elevations >8 x ULN. Eight patients had been rechallenged with bosentan and/or the investigational ERA and all eight had a recurrence of aminotransferase abnormalities that required discontinuation of ERA therapy. All patients had to have normal aminotransferase levels on entry to this study. Twenty-five of the 36 patients were also receiving prostanoid and/or phosphodiesterase type 5 (PDE5) inhibitor therapy. Two patients discontinued early (including one of the patients with a prior 8 x ULN elevation). Of the remaining 34 patients, one patient experienced a mild aminotransferase elevation at 12 weeks on ambrisentan 5 mg that resolved with decreasing the dosage to 2.5 mg, and that did not recur with later escalations to 10 mg. With a median follow-up of 13 months and with 50% of patients increasing the dose of ambrisentan to 10 mg, no patients were discontinued for aminotransferase elevations. While the uncontrolled study design does not provide information about what would have occurred with readministration of previously used ERAs or show that ambrisentan led to fewer aminotransferase elevations than would have been seen with those drugs, the study indicates that ambrisentan may be tried in patients who have experienced asymptomatic aminotransferase elevations on other ERAs after aminotransferase levels have returned to normal.
6.2 Postmarketing Experience
The following adverse reactions were identified during post approval use of ambrisentan. Because these reactions were reported voluntarily from a population of uncertain size, it is not possible to estimate reliably the frequency or to establish a causal relationship to drug exposure: anemia requiring transfusion [see Warnings and Precautions (5.6)] heart failure (associated with fluid retention), symptomatic hypotension, and hypersensitivity (e.g., angioedema, rash).
Elevations of liver aminotransferases (ALT, AST) have been reported with ambrisentan use; in most cases alternative causes of the liver injury could be identified (heart failure, hepatic congestion, hepatitis, alcohol use, hepatotoxic medications). Other endothelin receptor antagonists have been associated with elevations of aminotransferases, hepatotoxicity, and cases of liver failure [see Adverse Reactions (6.1)].
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on data from animal reproduction studies, ambrisentan may cause fetal harm when administered to a pregnant woman and is contraindicated during pregnancy. There are limited data on ambrisentan use in pregnant women. In animal reproduction studies, ambrisentan was teratogenic in rats and rabbits at doses which resulted in exposures of 3.5 and 1.7 times, respectively, the human dose of 10 mg per day [see Animal Data]. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, advise the patient of the potential hazard to a fetus [see Contraindications (4.1), Warnings and Precautions (5.1)].
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
Ambrisentan was teratogenic at oral dosages of ≥15 mg/kg/day (AUC 51.7 hmcg/mL) in rats and ≥7 mg/kg/day (24.7 hmcg/mL) in rabbits; it was not studied at lower dosages. These dosages are of 3.5 and 1.7 times, respectively, the human dose of 10 mg per day (14.8 hmcg/mL) based on AUC. In both species, there were abnormalities of the lower jaw and hard and soft palate, malformation of the heart and great vessels, and failure of formation of the thymus and thyroid.
A preclinical study in rats has shown decreased survival of newborn pups (mid and high dosages) and effects on testicle size and fertility of pups (high dosage) following maternal treatment with ambrisentan from late gestation through weaning. The mid and high dosages were 51 x, and 170 x (on a mg/m2 body surface area basis) the maximum oral human dose of 10 mg and an average adult body weight of 70 kg. These effects were absent at a maternal dosage 17 x the human dose based on mg/m2.
8.2 Lactation
Risk Summary
It is not known whether ambrisentan is present in human milk. Because many drugs are present in human milk and because of the potential for serious adverse reactions in breastfed infants from ambrisentan, a decision should be made whether to discontinue breastfeeding or discontinue ambrisentan, taking into account the importance of the drug to the mother.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Female patients of reproductive potential must have a negative pregnancy test prior to initiation of treatment, monthly pregnancy test during treatment, and pregnancy test 1 month after stopping treatment with ambrisentan. Advise patients to contact their healthcare provider if they become pregnant or suspect they may be pregnant. Perform a pregnancy test if pregnancy is suspected for any reason. For positive pregnancy tests, counsel patient on the potential risk to the fetus and patient options [see Boxed Warning and Dosage and Administration (2.2)].
Contraception
Female patients of reproductive potential must use acceptable methods of contraception during treatment with ambrisentan and for 1 month after stopping treatment with ambrisentan. Patients may choose one highly effective form of contraception (intrauterine device (IUD), contraceptive implant, or tubal sterilization) or a combination of methods (hormone method with a barrier method or two barrier methods). If a partner’s vasectomy is the chosen method of contraception, a hormone or barrier method must be used along with this method. Counsel patients on pregnancy planning and prevention, including emergency contraception, or designate counseling by another healthcare provider trained in contraceptive counseling [see Boxed Warning].
Infertility
Males
In a 6-month study of another endothelin receptor antagonist, bosentan, 25 male patients with WHO functional class III and IV PAH and normal baseline sperm count were evaluated for effects on testicular function. There was a decline in sperm count of at least 50% in 25% of the patients after 3 or 6 months of treatment with bosentan. One patient developed marked oligospermia at 3 months, and the sperm count remained low with 2 follow-up measurements over the subsequent 6 weeks. Bosentan was discontinued and after 2 months the sperm count had returned to baseline levels. In 22 patients who completed 6 months of treatment, sperm count remained within the normal range and no changes in sperm morphology, sperm motility, or hormone levels were observed. Based on these findings and preclinical data [see Nonclinical Toxicology (13.1)] from endothelin receptor antagonists, it cannot be excluded that endothelin receptor antagonists such as ambrisentan have an adverse effect on spermatogenesis. Counsel patients about the potential effects on fertility [see Warnings and Precautions (5.5)].
8.4 Pediatric Use
Safety and effectiveness of ambrisentan in pediatric patients have not been established.
Juvenile Animal Data
In juvenile rats administered ambrisentan orally once daily during postnatal day 7 to 26, 36, or 62, a decrease in brain weight (−3% to −8%) with no morphologic or neurobehavioral changes occurred after breathing sounds, apnea, and hypoxia were observed, at exposures approximately 1.8 to 7.0 times human pediatric exposures at 10 mg, based on AUC.
8.5 Geriatric Use
In the two placebo-controlled clinical studies of ambrisentan, 21% of patients were ≥65 years old and 5% were ≥75 years old. The elderly (age ≥65 years) showed less improvement in walk distances with ambrisentan than younger patients did, but the results of such subgroup analyses must be interpreted cautiously. Peripheral edema was more common in the elderly than in younger patients.
8.6 Renal Impairment
The impact of renal impairment on the pharmacokinetics of ambrisentan has been examined using a population pharmacokinetic approach in PAH patients with creatinine clearances ranging between 20 and 150 mL/min. There was no significant impact of mild or moderate renal impairment on exposure to ambrisentan [see Clinical Pharmacology (12.3)]. Dose adjustment of ambrisentan in patients with mild or moderate renal impairment is therefore not required. There is no information on the exposure to ambrisentan in patients with severe renal impairment.
The impact of hemodialysis on the disposition of ambrisentan has not been investigated.
8.7 Hepatic Impairment
Preexisting Hepatic Impairment
The influence of preexisting hepatic impairment on the pharmacokinetics of ambrisentan has not been evaluated. Because there is in vitro and in vivo evidence of significant metabolic and biliary contribution to the elimination of ambrisentan, hepatic impairment might be expected to have significant effects on the pharmacokinetics of ambrisentan [see Clinical Pharmacology (12.3)]. Ambrisentan is not recommended in patients with moderate or severe hepatic impairment. There is no information on the use of ambrisentan in patients with mild preexisting impaired liver function; however, exposure to ambrisentan may be increased in these patients.
Elevation of Liver Transaminases
Other endothelin receptor antagonists (ERAs) have been associated with aminotransferase (AST, ALT) elevations, hepatotoxicity, and cases of liver failure [see Adverse Reactions (6.1, 6.2)]. In patients who develop hepatic impairment after ambrisentan initiation, the cause of liver injury should be fully investigated. Discontinue ambrisentan if elevations of liver aminotransferases are >5 x ULN or if elevations are accompanied by bilirubin >2 x ULN, or by signs or symptoms of liver dysfunction and other causes are excluded.
-
10 OVERDOSAGE
There is no experience with overdosage of ambrisentan. The highest single dose of ambrisentan administered to healthy volunteers was 100 mg, and the highest daily dose administered to patients with PAH was 10 mg once daily. In healthy volunteers, single doses of 50 mg and 100 mg (5 to 10 times the maximum recommended dose) were associated with headache, flushing, dizziness, nausea, and nasal congestion. Massive overdosage could potentially result in hypotension that may require intervention.
-
11 DESCRIPTION
Ambrisentan is an endothelin receptor antagonist that is selective for the endothelin type-A (ETA) receptor. The chemical name of ambrisentan is (+)-(2S)-2-[(4,6-dimethylpyrimidin-2-yl)oxy]-3-methoxy-3,3-diphenylpropanoic acid. It has a molecular formula of C22H22N2O4 and a molecular weight of 378.42. It contains a single chiral center determined to be the (S) configuration and has the following structural formula:
Figure 1 Ambrisentan Structural Formula
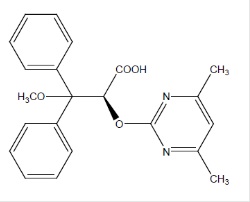
Ambrisentan is a white to off-white, crystalline solid. It is a carboxylic acid with a pKa of 4.0. Ambrisentan is practically insoluble in water and in aqueous solutions at low pH. Solubility increases in aqueous solutions at higher pH. In the solid state ambrisentan is very stable, is not hygroscopic, and is not light sensitive.
Ambrisentan is available as 5 mg and 10 mg film-coated tablets for once daily oral administration. The tablets include the following inactive ingredients: anhydrous lactose, colloidal silicon dioxide, croscarmellose sodium, magnesium stearate and microcrystalline cellulose. The tablets are film-coated with a coating material containing FD&C Red #40 aluminum lake, lecithin, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. Each round, pale pink ambrisentan tablet contains 5 mg of ambrisentan. Each oval, deep pink ambrisentan tablet contains 10 mg of ambrisentan. Ambrisentan tablets are unscored.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Endothelin-1 (ET-1) is a potent autocrine and paracrine peptide. Two receptor subtypes, ETA and ETB, mediate the effects of ET-1 in the vascular smooth muscle and endothelium. The primary actions of ETA are vasoconstriction and cell proliferation, while the predominant actions of ETB are vasodilation, antiproliferation, and ET-1 clearance.
In patients with PAH, plasma ET-1 concentrations are increased as much as 10-fold and correlate with increased mean right atrial pressure and disease severity. ET-1 and ET-1 mRNA concentrations are increased as much as 9-fold in the lung tissue of patients with PAH, primarily in the endothelium of pulmonary arteries. These findings suggest that ET-1 may play a critical role in the pathogenesis and progression of PAH.
Ambrisentan is a high-affinity (Ki=0.011 nM) ETA receptor antagonist with a high selectivity for the ETA versus ETB receptor (>4000-fold). The clinical impact of high selectivity for ETA is not known.
12.2 Pharmacodynamics
Cardiac Electrophysiology
In a randomized, positive- and placebo-controlled, parallel-group study, healthy subjects received either ambrisentan 10 mg daily followed by a single dose of 40 mg, placebo followed by a single dose of moxifloxacin 400 mg, or placebo alone. Ambrisentan 10 mg daily had no significant effect on the QTc interval. The 40 mg dose of ambrisentan increased mean QTc at tmax by 5 ms with an upper 95% confidence limit of 9 ms. For patients receiving ambrisentan 5 mg to 10 mg daily and not taking metabolic inhibitors, no significant QT prolongation is expected.
12.3 Pharmacokinetics
The pharmacokinetics of ambrisentan (S-ambrisentan) in healthy subjects is dose proportional. The absolute bioavailability of ambrisentan is not known. Ambrisentan is absorbed with peak concentrations occurring approximately 2 hours after oral administration in healthy subjects and PAH patients. Food does not affect its bioavailability. In vitro studies indicate that ambrisentan is a substrate of P-gp. Ambrisentan is highly bound to plasma proteins (99%). The elimination of ambrisentan is predominantly by non-renal pathways, but the relative contributions of metabolism and biliary elimination have not been well characterized. In plasma, the AUC of 4-hydroxymethyl ambrisentan accounts for approximately 4% relative to parent ambrisentan AUC. The in vivo inversion of S-ambrisentan to R-ambrisentan is negligible. The mean oral clearance of ambrisentan is 38 mL/min and 19 mL/min in healthy subjects and in PAH patients, respectively. Although ambrisentan has a 15-hour terminal half-life, the mean trough concentration of ambrisentan at steady-state is about 15% of the mean peak concentration and the accumulation factor is about 1.2 after long-term daily dosing, indicating that the effective half-life of ambrisentan is about 9 hours.
Drug Interactions
In Vitro Studies
Studies with human liver tissue indicate that ambrisentan is metabolized by CYP3A, CYP2C19, and uridine 5’-diphosphate glucuronosyltransferases (UGTs) 1A9S, 2B7S, and 1A3S. In vitro studies suggest that ambrisentan is a substrate of the Organic Anion Transporting Polypeptides OATP1B1 and OATP1B3, and P-glycoprotein (P-gp). Drug interactions might be expected because of these factors; however, a clinically relevant interaction has been demonstrated only with cyclosporine [see Drug Interactions (7)]. In vitro studies found ambrisentan to have little to no inhibition of human hepatic transporters. Ambrisentan demonstrated weak dose-dependent inhibition of OATP1B1, OATP1B3, and NTCP (IC50 of 47 μM, 45 μM, and approximately 100 μM, respectively) and no transporter-specific inhibition of BSEP, BRCP, P-gp, or MRP2. Ambrisentan does not inhibit or induce drug metabolizing enzymes at clinically relevant concentrations.
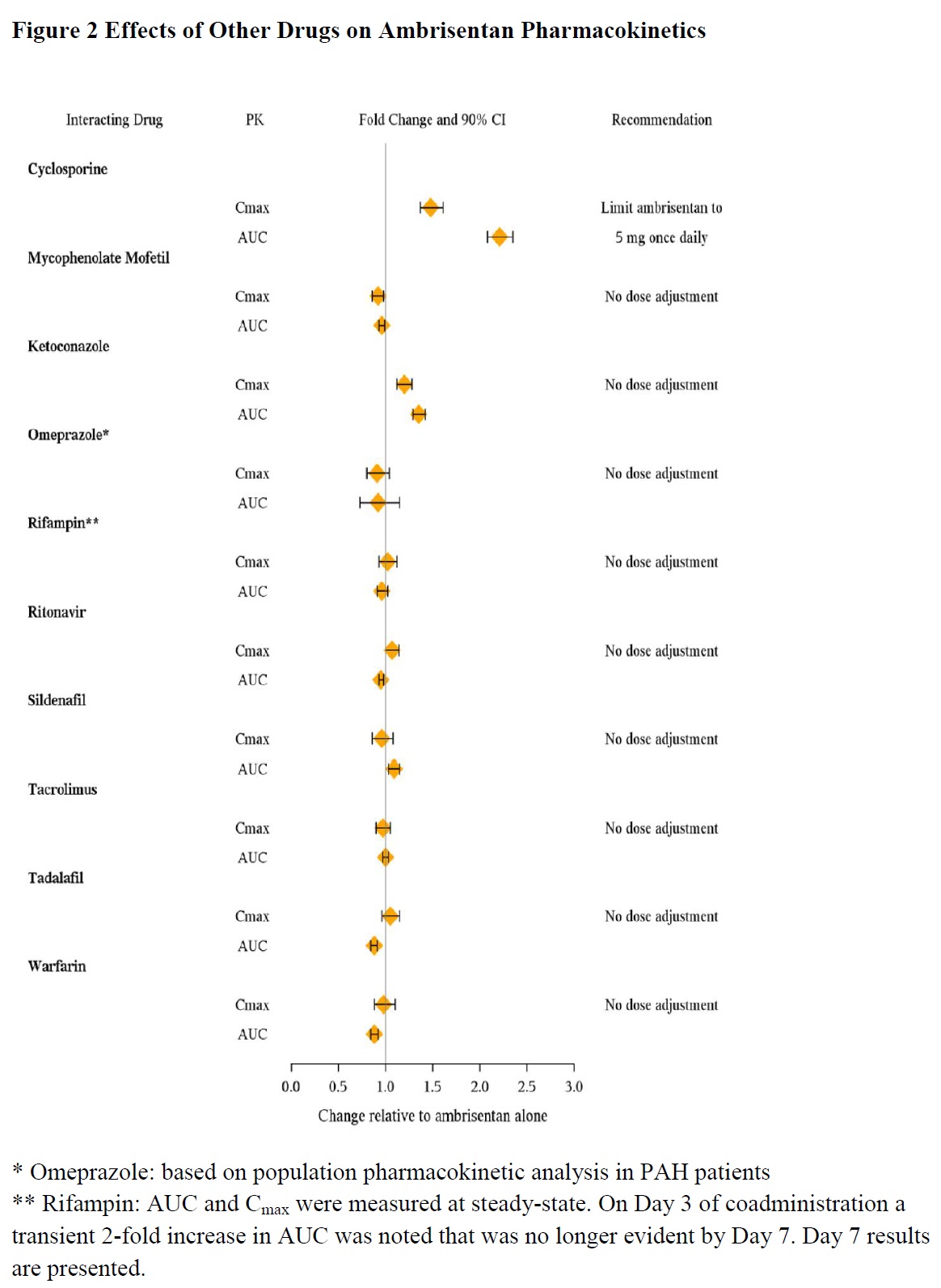
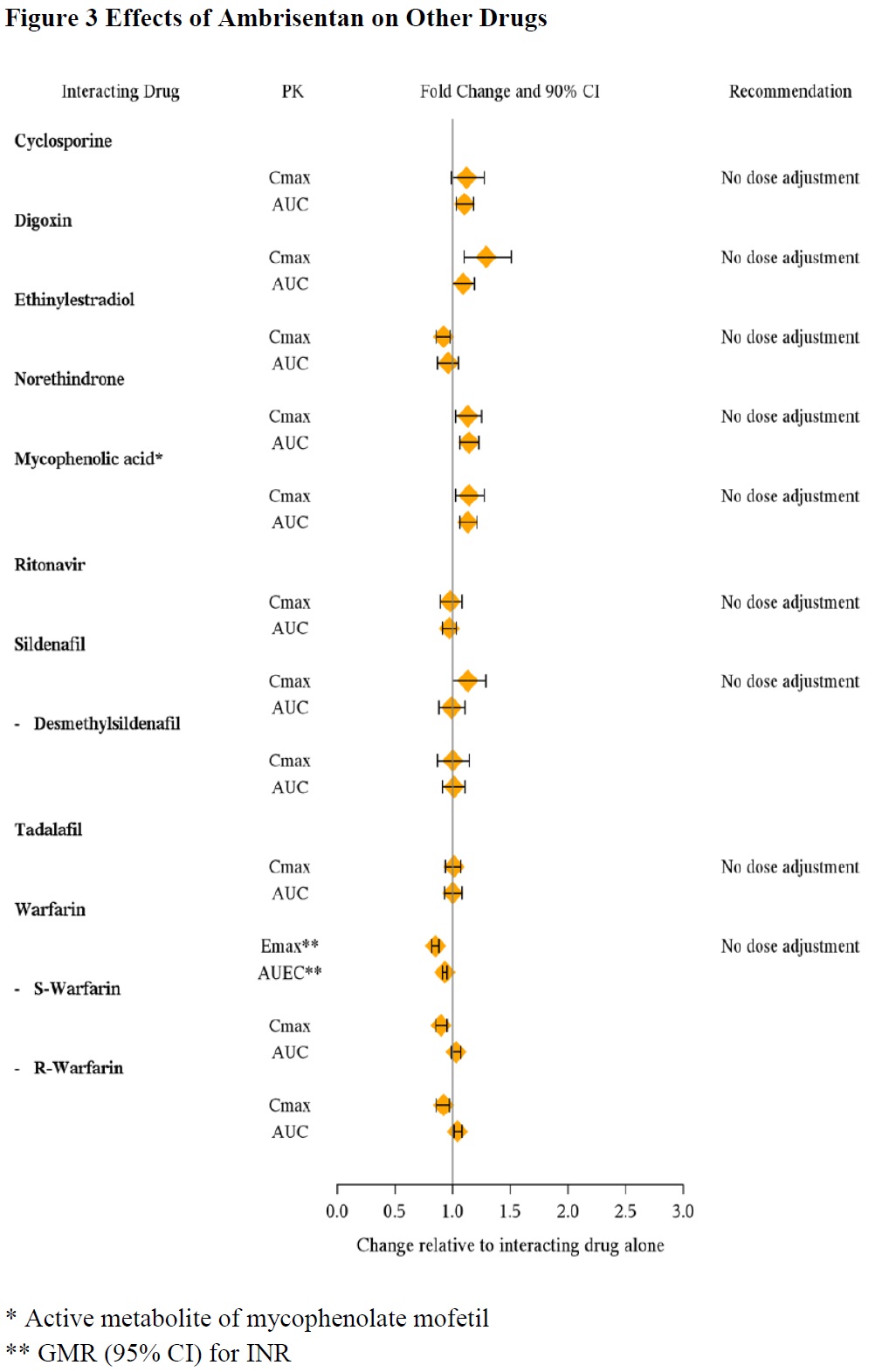
In Vivo Studies
The effects of other drugs on ambrisentan pharmacokinetics and the effects of ambrisentan on the exposure to other drugs are shown in Figure 2 and Figure 3, respectively.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Oral carcinogenicity studies of up to two years duration were conducted at starting doses of 10, 30, and 60 mg/kg/day in rats (8 to 48 times the maximum recommended human dose [MRHD] on a mg/m2 basis) and at 50, 150, and 250 mg/kg/day in mice (28 to 140 times the MRHD). In the rat study, the high- and mid-dose male and female groups had their doses lowered to 40 and 20 mg/kg/day, respectively, in week 51 because of effects on survival. The high-dose males and females were taken off drug completely in weeks 69 and 93, respectively. The only evidence of ambrisentan-related carcinogenicity was a positive trend in male rats, for the combined incidence of benign basal cell tumor and basal cell carcinoma of skin/subcutis in the mid-dose group (high-dose group excluded from analysis), and the occurrence of mammary fibroadenomas in males in the high-dose group. In the mouse study, high-dose male and female groups had their doses lowered to 150 mg/kg/day in week 39 and were taken off drug completely in week 96 (males) or week 76 (females). In mice, ambrisentan was not associated with excess tumors in any dosed group.
Positive findings of clastogenicity were detected, at drug concentrations producing moderate to high toxicity, in the chromosome aberration assay in cultured human lymphocytes. There was no evidence for genetic toxicity of ambrisentan when tested in vitro in bacteria (Ames test) or in vivo in rats (micronucleus assay, unscheduled DNA synthesis assay).
The development of testicular tubular atrophy and impaired fertility has been linked to the chronic administration of endothelin receptor antagonists in rodents. Testicular tubular degeneration was observed in rats treated with ambrisentan for two years at doses ≥10 mg/kg/day (8-fold MRHD). Increased incidences of testicular findings were also observed in mice treated for two years at doses ≥50 mg/kg/day (28-fold MRHD). Effects on sperm count, sperm morphology, mating performance, and fertility were observed in fertility studies in which male rats were treated with ambrisentan at oral doses of 300 mg/kg/day (236-fold MRHD). At doses of ≥10 mg/kg/day, observations of testicular histopathology in the absence of fertility and sperm effects were also present.
-
14 CLINICAL STUDIES
14.1 Pulmonary Arterial Hypertension (PAH)
Two 12-week, randomized, double-blind, placebo-controlled, multicenter studies were conducted in 393 patients with PAH (WHO Group 1). The two studies were identical in design except for the doses of ambrisentan and the geographic region of the investigational sites. ARIES-1 compared once-daily doses of 5 mg and 10 mg ambrisentan to placebo, while ARIES-2 compared once-daily doses of 2.5 mg and 5 mg ambrisentan to placebo. In both studies, ambrisentan or placebo was added to current therapy, which could have included a combination of anticoagulants, diuretics, calcium channel blockers, or digoxin, but not epoprostenol, treprostinil, iloprost, bosentan, or sildenafil. The primary study endpoint was 6-minute walk distance. In addition, clinical worsening, WHO functional class, dyspnea, and SF-36® Health Survey were assessed.
Patients had idiopathic or heritable PAH (64%) or PAH associated with connective tissue diseases (32%), HIV infection (3%), or anorexigen use (1%). There were no patients with PAH associated with congenital heart disease.
Patients had WHO functional class I (2%), II (38%), III (55%), or IV (5%) symptoms at baseline. The mean age of patients was 50 years, 79% of patients were female, and 77% were Caucasian.
Submaximal Exercise Ability
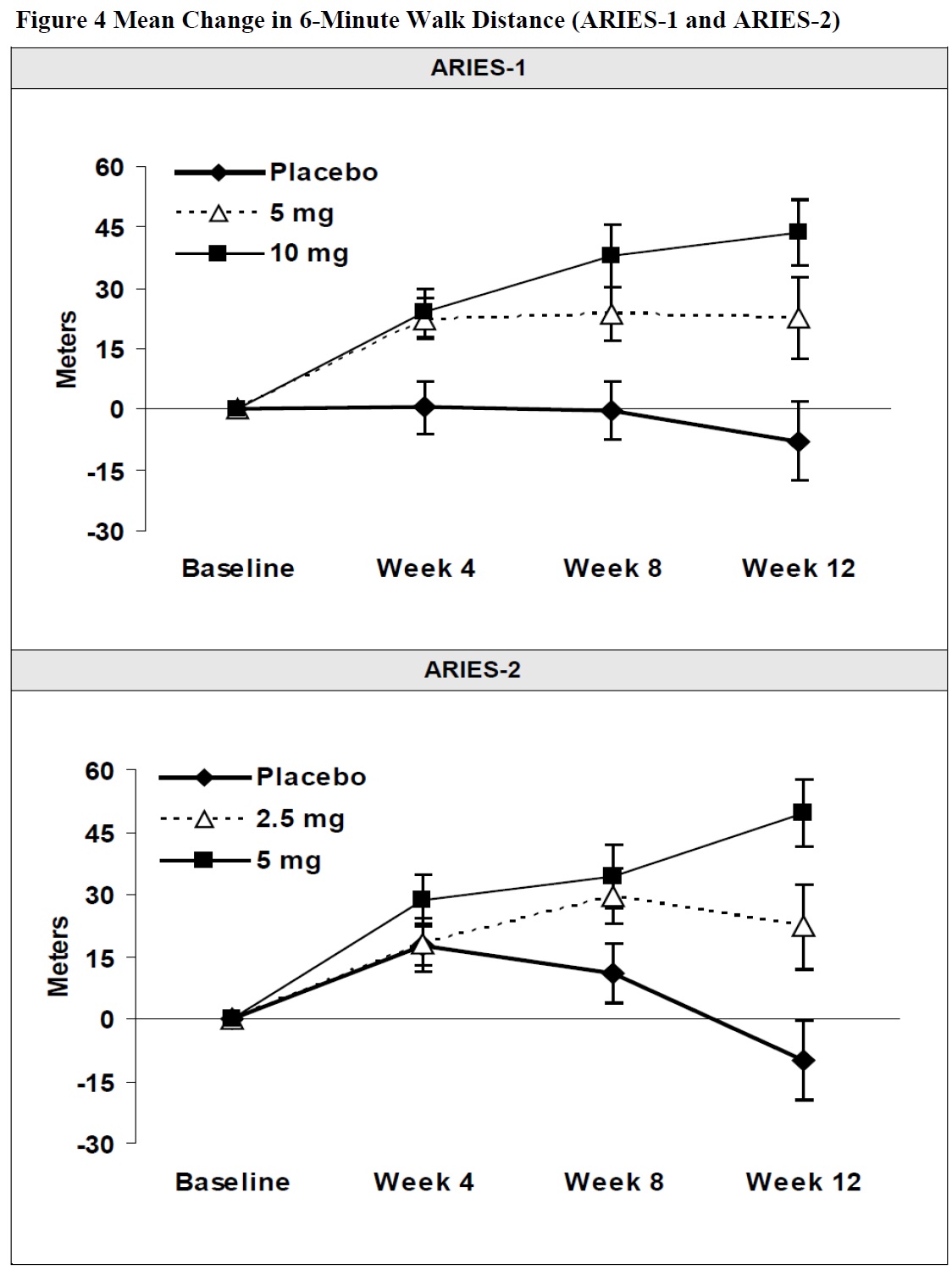
Results of the 6-minute walk distance at 12 weeks for the ARIES-1 and ARIES-2 studies are shown in Table 3 and Figure 4.
Table 3 Changes from Baseline in 6-Minute Walk Distance (meters) (ARIES-1 and ARIES-2)
ARIES-1
ARIES-2
Placebo (N=67)
5mg
(N=67)
10mg
(N=67)
Placebo (N=65)
2.5mg (N=64)
5mg
(N=63)
Baseline
342 ± 73
340 ± 77
342 ± 78
343 ± 86
347 ± 84
355 ± 84
Mean change from baseline
-8 ± 79
23 ± 83
44 ± 63
-10 ± 94
22 ± 83
49 ± 75
Placebo-adjusted mean change from baseline
-
31
51
-
32
59
Placebo-adjusted median change from baseline
-
27
39
-
30
45
p-valuea
-
0.008
<0.001
-
0.022
<0.001
Mean ± standard deviation
a p-values are Wilcoxon rank sum test comparisons of ambrisentan to placebo at Week 12 stratified by idiopathic or heritable PAH and non-idiopathic, non-heritable PAH patients
Mean change from baseline in 6-minute walk distance in the placebo and ambrisentan groups. Values are expressed as mean ± standard error of the mean.
In both studies, treatment with ambrisentan resulted in a significant improvement in 6-minute walk distance for each dose of ambrisentan and the improvements increased with dose. An increase in 6-minute walk distance was observed after 4 weeks of treatment with ambrisentan, with a dose-response observed after 12 weeks of treatment. Improvements in walk distance with ambrisentan were smaller for elderly patients (age ≥65) than younger patients and for patients with secondary PAH than for patients with idiopathic or heritable PAH. The results of such subgroup analyses must be interpreted cautiously.
Clinical Worsening
Time to clinical worsening of PAH was defined as the first occurrence of death, lung transplantation, hospitalization for PAH, atrial septostomy, study withdrawal due to the addition of other PAH therapeutic agents, or study withdrawal due to early escape. Early escape was defined as meeting two or more of the following criteria: a 20% decrease in the 6-minute walk distance; an increase in WHO functional class; worsening right ventricular failure; rapidly progressing cardiogenic, hepatic, or renal failure; or refractory systolic hypotension. The clinical worsening events during the 12-week treatment period of the ambrisentan clinical trials are shown in Table 4 and Figure 5.
Table 4 Time to Clinical Worsening (ARIES-1 and ARIES-2)
ARIES-1
ARIES-2
Placebo
(N=67)
Ambrisentan
(N=134)
Placebo
(N=65)
Ambrisentan
(N=127)
Clinical worsening, no. (%)
7 (10%)
4 (3%)
13 (22%)
8 (6%)
Hazard ratio
-
0.28
-
0.30
p-value, Log-rank test
-
0.030
-
0.005
Intention-to-treat population.
Note: Patients may have had more than one reason for clinical worsening.
Nominal p-values
There was a significant delay in the time to clinical worsening for patients receiving ambrisentan compared to placebo. Results in subgroups such as the elderly were also favorable.
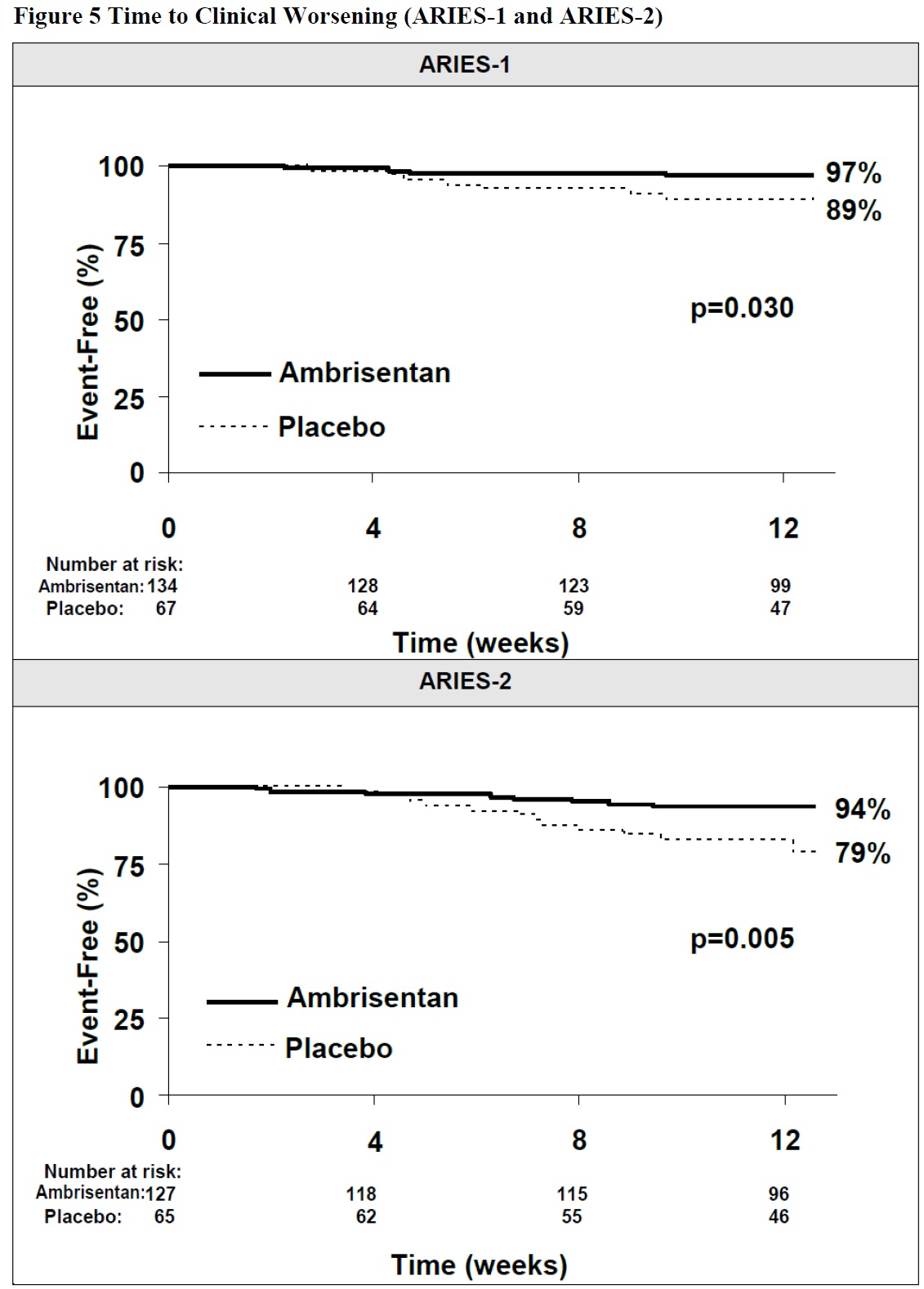
Time from randomization to clinical worsening with Kaplan-Meier estimates of the proportions of patients without events in ARIES-1 and ARIES-2.
p-values shown are the log-rank comparisons of ambrisentan to placebo stratified by idiopathic or heritable PAH and non-idiopathic, non-heritable PAH patients.
14.3 Long-term Treatment of PAH
In long-term follow-up of patients who were treated with ambrisentan (2.5 mg, 5 mg, or 10 mg once daily) in the two pivotal studies and their open-label extension (N=383), Kaplan-Meier estimates of survival at 1, 2, and 3 years were 93%, 85%, and 79%, respectively. Of the patients who remained on ambrisentan for up to 3 years, the majority received no other treatment for PAH. These uncontrolled observations do not allow comparison with a group not given ambrisentan and cannot be used to determine the long-term effect of ambrisentan on mortality.
14.4 Adverse Effects in Idiopathic Pulmonary Fibrosis (IPF)
A randomized controlled study in patients with IPF, with or without pulmonary hypertension (WHO Group 3), compared ambrisentan (N=329) to placebo (N=163). The study was terminated after 34 weeks for lack of efficacy, and was found to demonstrate a greater risk of disease progression or death on ambrisentan. More patients taking ambrisentan died (8% vs. 4%), had a respiratory hospitalization (13% vs. 6%), and had a decrease in FVC/DLCO (17% vs. 12%) [see Contraindications (4.2)].
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Ambrisentan film-coated tablets are supplied as follows:
Tablet Strength
Package Configuration
NDC No.
Description of Tablet; Debossed on Tablet
5 mg
30 count carton
(3 x 10 unit-dose)
47335-236-64
round biconvex; pale pink; debossed with “236” on one side and plain on the other side.
30 count bottle
47335-236-83
10 count carton
(1 x 10 unit-dose)
47335-236-66
10 mg
30 count carton
(3 x 10 unit-dose)
47335-237-64
Oval biconvex; deep pink; debossed with “237” on one side and plain on the other side.
30 count bottle
47335-237-83
10 count carton
(1 x 10 unit-dose)
47335-237-66
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature]. Store ambrisentan in its original packaging.
-
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-approved patient labeling (Medication Guide).
Embryo-fetal Toxicity
Instruct patients on the risk of fetal harm when ambrisentan is used in pregnancy [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)]. Female patients must enroll in the Ambrisentan REMS. Instruct females of reproductive potential to immediately contact their physician if they suspect they may be pregnant.
AmbrisentanRisk Evaluation and Mitigation Strategy (REMS)
For female patients, ambrisentan is only available through a restricted program called the Ambrisentan REMS [see Contraindications (4.1), Warnings and Precautions (5.2)]. Male patients are not enrolled in the Ambrisentan REMS.
Inform female patients (and their guardians, if applicable) of the following notable requirements:
- All female patients must sign an enrollment form.
- Female patients of reproductive potential must comply with the pregnancy testing and contraception requirements [see Use in Specific Populations (8.3)].
- Females of reproductive potential must be counseled on the use of emergency contraception in the event of unprotected sex or known or suspected contraceptive failure.
- Pre-pubertal females must report any changes in their reproductive status immediately to their prescriber.
Ambrisentan is available only from certified pharmacies participating in the program. Therefore, provide patients with the telephone number and website for information on how to obtain the product.
Hepatic Effects
Advise patients of the symptoms of potential liver injury and instruct them to report any of these symptoms to their physician.
Hematological Change
Advise patients of the importance of hemoglobin testing.
Other Risks Associated with ambrisentan
Instruct patients that the risks associated with ambrisentan also include the following:
- Decreases in sperm count
- Fluid overload
Administration
Advise patients not to split, crush, or chew tablets.
-
Medication Guide
Ambrisentan (am" bri senʹ tan) Tablets
Read this Medication Guide before you start taking ambrisentan tablets and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your doctor about your medical condition or your treatment.
What is the most important information I should know about ambrisentan tablets?
- Serious birth defects.
Ambrisentan tablets can cause serious birth defects if taken during pregnancy.
-
- Females must not be pregnant when they start taking ambrisentan tablets or become pregnant during treatment with ambrisentan tablets.
- Females who are able to get pregnant must have a negative pregnancy test before beginning treatment with ambrisentan tablets, each month during treatment with ambrisentan tablets, and one month after stopping ambrisentan tablets. Talk to your doctor about your menstrual cycle. Your doctor will decide when to do a pregnancy test, and will order a pregnancy test for you depending on your menstrual cycle.
- Females who are able to get pregnant are females who:
- have entered puberty, even if they have not started their menstrual period, and
- have a uterus, and
- have not gone through menopause. Menopause means that you have not had a menstrual period for at least 12 months for natural reasons, or that you have had your ovaries removed.
- Females who are not able to get pregnant are females who:
- have not yet entered puberty, or
- do not have a uterus, or
- have gone through menopause. Menopause means that you have not had a menstrual period for at least 12 months for natural reasons, or that you have had your ovaries removed, or
- who are infertile for any other medical reason and this infertility is permanent and cannot be reversed
- Females who are able to get pregnant are females who:
Females who are able to get pregnant must use two acceptable forms of birth control during treatment with ambrisentan tablets, and for one month after stopping ambrisentan tablets because the medicine may still be in the body.
- If you have had a tubal sterilization or have an IUD (intrauterine device) or progesterone implant, these methods can be used alone and no other form of birth control is needed.
- Talk with your doctor or gynecologist (a doctor who specializes in female reproduction) to find out about options for acceptable forms of birth control that you may use to prevent pregnancy during treatment with ambrisentan tablets.
If you decide that you want to change the form of birth control that you use, talk with your doctor or gynecologist to be sure that you choose another acceptable form of birth control.
See the chart below for Acceptable Birth Control Options during treatment with ambrisentan tablets.
Acceptable Birth Control Options
Option 1
Option 2
Option 3
Option 4
One method from this list:
OROne method from this list:
OROne method from this list:
OROne method from this list:
Standard intrauterine device (Copper T 380A IUD)
Intrauterine system (LNg 20 IUS – progesterone IUD)
Tubal sterilization
Progesterone implant
Estrogen and progesterone oral contraceptives (“the pill”)
Estrogen and progesterone transdermal patch
Vaginal ring
Progesterone injection
Diaphragm with spermicide
Cervical cap with spermicide
Partner’s vasectomy
PLUS
One method from this list:
Male condom
Diaphragm with spermicide
Cervical cap with spermicide
Estrogen and progesterone oral contraceptive (“the pill”)
Estrogen and progesterone transdermal patch
Vaginal ring
Prolgesterone injection
PLUS
One method from this list:
Male condom
PLUS
One method from this list:
Male condom
Diaphragm with spermicide
Cervical cap with spermicide
- Do not have unprotected sex. Talk to your doctor or pharmacist right away if you have unprotected sex or if you think your birth control has failed. Your doctor may tell you to use emergency birth control.
- Tell your doctor right away if you miss a menstrual period or think you may be pregnant for any reason.
If you are the parent or caregiver of a female child who started taking ambrisentan tablets before reaching puberty, you should check your child regularly to see if she is developing signs of puberty. Tell your doctor right away if you notice that she is developing breast buds or pubic hair. Your doctor should decide if your child has reached puberty. Your child may reach puberty before having her first menstrual period.
Females can only receive ambrisentan tablets through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the Ambrisentan REMS. If you are a female who can get pregnant, you must talk to your doctor, understand the benefits and risks of ambrisentan tablets, and agree to all of the instructions in the Ambrisentan REMS.
Males can receive ambrisentan tablets without taking part in the Ambrisentan REMS.
What are ambrisentan tablets?
- Ambrisentan tablets are a prescription medicine used to treat pulmonary arterial hypertension (PAH), which is high blood pressure in the arteries of your lungs.
- Ambrisentan tablets can improve your ability to exercise and it can help slow down the worsening of your physical condition and symptoms.
- It is not known if ambrisentan tablets are safe and effective in children.
Who should not take ambrisentan tablets?
Do not take ambrisentan tablets if:
- you are pregnant, plan to become pregnant, or become pregnant during treatment with ambrisentan tablets. Ambrisentan tablets can cause serious birth defects. (See “What is the most important information I should know about ambrisentan tablets?”) Serious birth defects from ambrisentan tablets happen early in pregnancy.
- you have a condition called Idiopathic Pulmonary Fibrosis (IPF)
What should I tell my doctor before taking ambrisentan tablets?
Before you take ambrisentan tablets, tell your doctor if you:
- have been told that you have a low red blood cell level (anemia)
- have liver problems
- have any other medical conditions
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Ambrisentan tablets and other medicines may affect each other, causing side effects. Do not start any new medicines until you check with your doctor.
Especially tell your doctor if you take the medicine cyclosporine (Gengraf*, Neoral*, Sandimmune*). Your doctor may need to change your dose of ambrisentan tablets.
How should I take ambrisentan tablets?
- Ambrisentan tablets will be mailed to you by a certified pharmacy. Your doctor will give you complete details.
- Take ambrisentan tablets exactly as your doctor tells you to take it. Do not stop taking ambrisentan tablets unless your doctor tells you to stop.
- You can take ambrisentan tablets with or without food.
- Do not split, crush or chew ambrisentan tablets.
- It will be easier to remember to take ambrisentan tablets if you take it at the same time each day.
- If you take more than your regular dose of ambrisentan tablets, call your doctor right away.
- If you miss a dose, take it as soon as you remember that day. Take your next dose at the regular time. Do not take two doses at the same time to make up for a missed dose.
What should I avoid while taking ambrisentan tablets?
- Do not get pregnant while taking ambrisentan tablets. (See the serious birth defects section of the Medication Guide above called “What is the most important information I should know about ambrisentan tablets?”) If you miss a menstrual period, or think you might be pregnant, call your doctor right away.
- It is not known if ambrisentan passes into your breast milk. You should not breastfeed if you are taking ambrisentan tablets. Talk to your doctor about the best way to feed your baby if you take ambrisentan tablets.
What are the possible side effects of ambrisentan tablets?
Ambrisentan tablets can cause serious side effects including:
- See “What is the most important information I should know about ambrisentan tablets?”
- Swelling all over the body (fluid retention) can happen within weeks after starting ambrisentan tablets. Tell your doctor right away if you have any unusual weight gain, tiredness, or trouble breathing while taking ambrisentan tablets. These may be symptoms of a serious health problem. You may need to be treated with medicine or need to go to the hospital.
- Decreased sperm count. Decreased sperm counts have happened in some men taking a medicine that is like ambrisentan tablets. A decreased sperm count may affect the ability to father a child. Tell your doctor if being able to have children is important to you.
- Low red blood cell levels (anemia) can happen during the first weeks after starting ambrisentan tablets. If this happens, you may need a blood transfusion. Your doctor will do blood tests to check your red blood cells before starting ambrisentan tablets. Your doctor may also do these tests during treatment with ambrisentan tablets.
The most common side effects of ambrisentan tablets include:
- swelling of hands, legs, ankles and feet (peripheral edema)
- stuffy nose (nasal congestion)
- inflamed nasal passages (sinusitis)
- hot flashes or getting red in the face (flushing)
Some medicines that are like ambrisentan tablets can cause liver problems. Tell your doctor if you get any of these symptoms of a liver problem while taking ambrisentan tablets:
- loss of appetite
- nausea or vomiting
- fever
- achiness
- generally do not feel well
- pain in the upper right stomach (abdominal) area
- yellowing of your skin or the whites of your eyes
- dark urine
- itching
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of ambrisentan tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. For more information call 1-800-818-4555.
How should I store ambrisentan tablets?Store ambrisentan tablets at room temperature between 68ºF to 77ºF (20ºC to 25ºC), in the package they come in.
Keep ambrisentan tablets and all medicines out of the reach of children.
General information about the safe and effective use of ambrisentan tablets
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ambrisentan tablets for a condition for which it was not prescribed. Do not give ambrisentan tablets to other people, even if they have the same symptoms that you have. It may harm them.
This Medication Guide summarizes the most important information about ambrisentan tablets. If you would like more information, ask your doctor. You can ask your doctor or pharmacist for information about ambrisentan tablets that is written for health professionals.
What are the ingredients in ambrisentan tablets?
Active ingredient: ambrisentan
Inactive Ingredients: anhydrous lactose, colloidal silicon dioxide, croscarmellose sodium, magnesium stearate and microcrystalline cellulose. The tablets are film-coated with a coating material containing FD&C Red #40 aluminum lake, lecithin, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
* All trademarks are the property of their respective owners.
Distributed by:
Sun Pharmaceutical Industries, Inc.
Cranbury, NJ 08512
Manufactured by:
Sun Pharmaceutical Industries Ltd.
Survey No. 259/15,
Dadra-396 191, (U.T. of D & NH), India.
PGPI0364B
ISS. 10/2019
This Medication Guide has been approved by the U.S. Food and Drug Administration. -
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 47335-236-66
Ambrisentan Tablets
5 mg
PHARMACIST: Please dispense with Medication Guide provided separately to each patient.
Rx only
10 Tablets(1 X 10 Unit-dose)
SUN PHARMA

-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 47335-237-66
Ambrisentan Tablets
10 mg
PHARMACIST: Please dispense with Medication Guide provided separately to each patient.
Rx only
10 Tablets (1 X 10 Unit-dose)
SUN PHARMA

-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 47335-236-83
Ambrisentan Tablets
5 mg
PHARMACIST: Please dispense with Medication Guide provided separately to each patient.
Rx only
30 Tablets
SUN PHARMA

-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 47335-237-83
Ambrisentan Tablets
10 mg
PHARMACIST: Please dispense with Medication Guide provided separately to each patient.
Rx only
30 Tablets
SUN PHARMA

-
INGREDIENTS AND APPEARANCE
AMBRISENTAN
ambrisentan tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 47335-236 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AMBRISENTAN (UNII: HW6NV07QEC) (AMBRISENTAN - UNII:HW6NV07QEC) AMBRISENTAN 5 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) FD&C RED NO. 40 (UNII: WZB9127XOA) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ALUMINUM OXIDE (UNII: LMI26O6933) Product Characteristics Color PINK (PALE PINK) Score no score Shape ROUND Size 7mm Flavor Imprint Code 236 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 47335-236-83 30 in 1 BOTTLE; Type 0: Not a Combination Product 04/29/2019 2 NDC: 47335-236-66 1 in 1 CARTON 04/29/2019 2 NDC: 47335-236-60 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC: 47335-236-64 3 in 1 CARTON 04/29/2019 3 NDC: 47335-236-60 10 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210784 04/29/2019 AMBRISENTAN
ambrisentan tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 47335-237 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AMBRISENTAN (UNII: HW6NV07QEC) (AMBRISENTAN - UNII:HW6NV07QEC) AMBRISENTAN 10 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) FD&C RED NO. 40 (UNII: WZB9127XOA) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ALUMINUM OXIDE (UNII: LMI26O6933) Product Characteristics Color PINK (DEEP PINK) Score no score Shape OVAL Size 10mm Flavor Imprint Code 237 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 47335-237-83 30 in 1 BOTTLE; Type 0: Not a Combination Product 04/29/2019 2 NDC: 47335-237-66 1 in 1 CARTON 04/29/2019 2 NDC: 47335-237-60 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC: 47335-237-64 3 in 1 CARTON 04/29/2019 3 NDC: 47335-237-60 10 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210784 04/29/2019 Labeler - Sun Pharmaceutical Industries, Inc. (146974886) Establishment Name Address ID/FEI Business Operations Sun Pharmaceutical Industries Limited 725959238 ANALYSIS(47335-236, 47335-237) , MANUFACTURE(47335-236, 47335-237)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.