TRALEMENT- trace elements injection 4 injection, solution
Tralement by
Drug Labeling and Warnings
Tralement by is a Prescription medication manufactured, distributed, or labeled by American Regent, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use TRALEMENT® safely and effectively. See full prescribing information for TRALEMENT® .
TRALEMENT® (trace elements injection 4*) for intravenous use
Initial U.S. Approval: 2020
INDICATIONS AND USAGE
Tralement is a combination of trace elements (zinc sulfate, cupric sulfate, manganese sulfate and selenious acid) indicated in adult and pediatric patients weighing at least 10 kg as a source of zinc, copper, manganese, and selenium for parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. (1)
DOSAGE AND ADMINISTRATION
- Single-dose vial amd multiple-dose vial. Not for direct intravenous infusion. (2.1)
- See full prescribing information for information on preparation, administration and general dosing considerations. (2.1, 2.2, 2.3, 2.4)
Recommended Dosage - Each mL of Tralement provides zinc 3 mg, copper 0.3 mg, manganese 55 mcg, and selenium 60 mcg. (2.5)
-
Adults and Pediatric Patients Weighing at Least 50 kg: The recommended dosage of Tralement is 1 mL per day added to parenteral nutrition.
Tralement is not recommended for patients who may require a lower dosage of one or more of the individual trace elements. (2.5) -
Pediatric Patients Weighing 10 kg to 49 kg: The recommended dosage of Tralement based on body weight is 0.2 mL to 0.8 mL per day added to
parenteral nutrition. Tralement does not provide the recommended daily dosage of zinc (in heavier patients in some weight bands), copper or selenium. Additional supplementation using single trace element products may be needed for these patients. For complete dosing information see table in the full prescribing information. (2.4, 2.5) - Monitor trace element concentrations in blood during long-term administration of parenteral nutrition. (2.5)
DOSAGE FORMS AND STRENGTHS
Injection: (3)
- 1 mL in a single-dose vial and 10 mL in a multiple-dose vial. Each mL contains zinc 3 mg, copper 0.3 mg, manganese 55 mcg, and selenium 60 mcg.
WARNINGS AND PRECAUTIONS
- Pulmonary Embolism due to Pulmonary Vascular Precipitates: If signs of pulmonary distress occur, stop the infusion and initiate a medical evaluation. (5.1)
- Vein Damage and Thrombosis: Solutions with osmolarity of 900 mOsmol/L or more must be infused through a central catheter. (2.1, 5.2)
- Neurologic Toxicity with Manganese: Monitor for clinical signs and symptoms of neurotoxicity, whole blood manganese concentrations and liver function tests in patients receiving long-term Tralement. Discontinue Tralement and consider brain magnetic resonance imaging (MRI) if toxicity suspected. (5.3)
- Hepatic Accumulation of Copper and Manganese: Assess for development of hepatic or biliary dysfunction. Monitor concentrations of copper and manganese in patients with cholestasis, biliary dysfunction or cirrhosis receiving Tralement long-term. (5.4)
- Aluminum Toxicity: Increased risk in patients with renal impairment, including preterm infants. (5.5, 8.4)
- Monitoring and Laboratory Tests: Monitor blood zinc, copper, manganese, and selenium concentrations, fluid and electrolyte status, serum osmolarity, blood glucose, liver and kidney function, blood count and coagulation parameters. (5.6, 2.4)
- Hypersensitivity Reactions with Zinc and Copper: If reactions occur, discontinue Tralement and initiate appropriate medical treatment. (5.7)
To report SUSPECTED ADVERSE REACTIONS, contact American Regent, Inc. at 1-800-734-9236 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
ADVERSE REACTIONS
No adverse reactions related to zinc, copper, selenium, or manganese have been reported in patients receiving intravenously administered parenteral solutions containing these trace elements within the recommended dosage range. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Actavis at 1-800-432-8534 or FDA at 1-800-FDA-1088, or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Pediatric Patients Weighing Less than 10 kg: Tralement is not approved for use in this subpopulation because the product does not provide an adequate dosage of zinc, copper, or selenium and exceeds the recommended dosage of manganese. (8.4)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 7/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Information
2.2 Preparation and Administration Instructions
2.3 Preparation Instructions for Admixing Using a Parenteral Nutrition Container
2.4 Overview of Dosing
2.5 Recommended Dosage and Monitoring in Adult and Pediatric Patients
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Pulmonary Embolism due to Pulmonary Vascular Precipitates
5.2 Vein Damage and Thrombosis
5.3 Neurologic Toxicity with Manganese
5.4 Hepatic Accumulation of Copper and Manganese
5.5 Aluminum Toxicity
5.6 Monitoring and Laboratory Tests
5.7 Hypersensitivity Reactions with Zinc and Copper
6 ADVERSE REACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
16 HOW SUPPLIED
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Information
Tralement is supplied as a single-dose vial and multiple-dose vial for admixture use only. It is not for direct intravenous infusion. Prior to administration, Tralement must be transferred to a separate parenteral nutrition container and used as an admixture in parenteral nutrition solution.
The final parenteral nutrition solution is for intravenous infusion into a central or peripheral vein. The choice of a central or peripheral venous route should depend on the osmolarity of the final infusate. Solutions with osmolarity of 900 mOsmol/L or greater must be infused through a central catheter [see Warnings and Precautions (5.2)].
2.2 Preparation and Administration Instructions
- Tralement is not for direct intravenous infusion. Prior to administration, Tralement must be prepared and used as an admixture in parenteral nutrition solution.
- Add Tralement to the parenteral nutrition solution in a suitable work area such as a laminar flow hood (or an equivalent clean air compounding area). The key factor in the preparation is careful aseptic technique to avoid inadvertent touch contamination during mixing of solutions and addition of other nutrients.
- Inspect the parenteral nutrition solution containing Tralement for particulate matter before admixing, after admixing, and prior to administration.
2.3 Preparation Instructions for Admixing Using a Parenteral Nutrition Container
- Inspect Tralement single-dose vial and multiple-dose vial for particulate matter.
- Transfer Tralement to the parenteral nutrition container after the admixture of amino acids, dextrose, lipid emulsion (if added), and electrolytes solutions is prepared.
- Because additives may be incompatible, evaluate all additions to the parenteral nutrition container for compatibility and stability of the resulting preparation. Consult with pharmacist, if available. For introducing additives to the parenteral nutrition container, use aseptic technique.
- An interaction may occur between cupric ion and ascorbic acid; therefore, multivitamin additives should be added to the admixed parenteral nutrition solution shortly before infusion.
- Inspect the final parenteral nutrition solution containing Tralement to ensure that:
o Precipitates have not formed during mixing or addition on additives.
o The emulsion has not separated, if lipid emulsion has been added. Separation of the emulsion can be visibly identified by a yellowish streaking or the accumulation of yellowish droplets in the admixed emulsion.
o Discard if any precipitates are observed.
Stability and Storage
- Single-dose vial. Discard unused portion.
- Multiple-dose vial: May puncture up to 20 times. Discard any unused portion after 28 days.
- Use parenteral nutrition solutions containing Tralement promptly after mixing. Any storage of the admixture should be under refrigeration from 2ºC to 8ºC (36ºF to 46ºF) and limited to a period of no longer than 9 days. After removal from refrigeration, use promptly and complete the infusion within 24 hours. Discard any remaining admixture.
- Protect the parenteral nutrition solution from light.
2.4 Overview of Dosing
- Prior to administration of parenteral nutrition solution containing Tralement, correct severe fluid, electrolyte, and acid-base disorders.
- The dosage of the final parenteral nutrition solution containing Tralement must be based on the concentrations of all components in the solution, the patient’s clinical condition, nutritional requirements, and the contribution of oral or enteral intake.
- For pediatric patients weighing 10 to 49 kg, Tralement does not provide the recommended daily dosage of zinc (in heavier patients in some weight bands), copper or selenium. Additional supplementation using single trace element products may be needed for these patients [see Dosage and Administration (2.5)].
- Monitor fluid and electrolyte status during treatment use of Tralement and adjust the parenteral nutrition solution as needed
2.5 Recommended Dosage and Monitoring in Adult and Pediatric Patients
Tralement is a fixed-combination product. Each mL of Tralement provides zinc 3 mg, copper 0.3 mg, manganese 55 mcg, and selenium 60 mcg.
Tralement is recommended only for patients who require supplementation with all four of the individual trace elements (i.e., zinc, copper, manganese and selenium).
Adults and Pediatric Patients Weighing at least 50 kg:
The recommended dosage of Tralement is 1 mL per day added to parenteral nutrition (zinc 3 mg/copper 0.3 mg/manganese 55 mcg/selenium 60 mcg). Tralement is not recommended for those patients who may require a lower dosage of one or more of the individual trace elements.
Pediatric Patients Weighing 10 kg to 49 kg:
The recommended dosage of Tralement by volume to be added to parenteral nutrition is based on body weight and ranges from 0.2 mL to 0.8 mL per day as shown in Table 1.Body Weight Recommended
Weight-Based
Dosage of
Tralement
in VolumeAmount of Trace Element Provided by the Corresponding Tralement Volume Zinc Copper Manganese Selenium 10 kg to 19 kg 0.2 mL 600 mcg 60 mcg 11 mcg 12 mcg 20 kg to 29 kg 0.4 mL 1,200 mcg 120 mcg 22 mcg 24 mcg 30 kg to 39 kg 0.6 mL 1,800 mcg 180 mcg 33 mcg 36 mcg 40 kg to 49 kg 0.8 mL 2,400 mcg 240 mcg 44 mcg 48 mcg Use Additional Supplementation with Tralement
For pediatric patients weighing 10 kg to 49 kg, additional zinc (in heavier patients in some weight bands), copper and selenium may be needed to meet the recommended daily dosage of these trace elements, shown below. To determine the additional amount of supplementation that is needed, compare the calculated daily recommended dosage based on the body weight of the patient to the amount of each trace element provided by Tralement (Table 1) and other oral or enteral nutrition sources.- Zinc: 50 mcg/kg/day (up to 3,000 mcg/day)
- Copper: 20 mcg/kg/day (up to 300 mcg/day)
- Selenium: 2 mcg/kg/day (up to 60 mcg/day)
Do not supplement Tralement with additional manganese. Accumulation of manganese in the brain can occur with long-term administration with higher than the recommended dosage of 1 mcg/kg/day (up to 55 mcg/day) [see Warnings and Precautions (].
Monitoring- Monitor serum zinc, copper, and selenium concentrations and manganese whole blood concentrations during long-term administration of parenteral nutrition.
- Trace elements concentrations may vary depending on the assay used and the laboratory reference range. The collection, processing, and storage of the blood samples should be performed according to the laboratory’s sample requirements for analysis.
o Zinc: In serum, the reported concentration range in healthy adults is 60 to 140 mcg/dL. Zinc concentrations in hemolyzed samples may be falsely elevated due to release of zinc from erythrocytes.
o Copper: In serum, the reported concentration range in healthy adults is 70 to 175 mcg/dL; consider obtaining concentrations of ceruloplasmin along with serum copper.
o Manganese: In whole blood, the reported concentration range in healthy adults is 4 to 16 mcg/L.
o Selenium: In serum, the reported concentration range in healthy adults is 7 to 19 mcg/dL.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Tralement is contraindicated in patients with hypersensitivity to zinc or copper [see Warnings and Precautions (5.7)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Pulmonary Embolism due to Pulmonary Vascular Precipitates
Pulmonary vascular precipitates causing pulmonary vascular emboli and pulmonary distress have been reported in patients receiving parenteral nutrition. The cause of precipitate formation has not been determined in all cases; however, in some fatal cases, pulmonary emboli occurred as a result of calcium phosphate precipitates. Precipitation has occurred following passage through an in-line filter; in vivo precipitate formation may also have occurred. If signs of pulmonary distress occur, stop the parenteral nutrition infusion and initiate a medical evaluation. In addition to inspection of the solution [see Dosage and Administration (2.2, 2.3)], the infusion set, and catheter should also periodically be checked for precipitates.
5.2 Vein Damage and Thrombosis
Tralement must be prepared and used as an admixture in parenteral nutrition solution. It is not for direct intravenous infusion. In addition, consider the osmolarity of the final parenteral nutrition solution in determining peripheral versus central administration. Solution with an osmolarity of 900 mOsmol/L or
greater must be infused through a central catheter [see Dosage and Administration (2.1)]. The infusion of hypertonic nutrient solution into a peripheral vein may result in vein irritation, vein damage, and/or thrombosis. The primary complication of peripheral access is venous thrombophlebitis, which manifests
as pain, erythema, tenderness or a palpable cord. Remove the catheter as soon as possible, if thrombophlebitis develops.5.3 Neurologic Toxicity with Manganese
Manganese accumulation in the basal ganglia has been reported in adult and pediatric patients on long-term parenteral nutrition receiving manganese at higher than recommended dosages and in association with cholestatic liver disease. Some adult patients with brain MRI findings reportedly experienced neuropsychiatric symptoms, including changes in mood or memory, seizures and/or parkinsonian-like tremors, dysarthria, mask-face, and halting gait. Some pediatric patients experienced dystonic movements or seizures. Brain MRI findings and clinical symptoms have also been observed in patients who received manganese at or below the recommended dosage and with normal blood manganese concentrations. Regression of symptoms and MRI findings have occurred over weeks to months following discontinuation of manganese in most patients but have not always completely resolved.
Monitor patients receiving long-term parenteral nutrition solutions containing Tralement for neurologic signs and symptoms and routinely monitor whole blood manganese concentrations and liver function tests. In case of suspected manganese toxicity or new neuro-psychiatric manifestations, temporarily discontinue Tralement, check manganese whole blood concentrations, and consider brain MRI evaluation.
Monitor patients receiving Tralement for cholestasis or other biliary liver disease. Consider individual trace element products as an alternative to Tralement in patients with hepatic and/or biliary dysfunction [see Warnings and Precautions (5.4)].
5.4 Hepatic Accumulation of Copper and Manganese
Copper is primarily eliminated in the bile and excretion is decreased in patients with cholestasis and/or cirrhosis. Hepatic accumulation of copper and manganese have been reported in autopsies of patients receiving long-term parenteral nutrition containing copper and manganese at dosages higher
than recommended.Patients receiving parenteral nutrition with cholestasis and/or cirrhosis are at increased risk of manganese brain deposition and neurotoxicity [see Warnings and Precautions (5.3)].
Administration of copper to patients with cholestasis, and/or cirrhosis may cause hepatic accumulation of copper. Administration of copper to patients with Wilson disease, an inborn error of copper metabolism with defect in hepatocellular copper transport, may cause both increased hepatic accumulation of copper and aggravation of the underlying hepatocellular degeneration.
For patients with cholestasis, biliary dysfunction, or cirrhosis, monitor hepatic and biliary function during long-term administration of Tralement. If a patient develops signs or symptoms of hepatic or biliary dysfunction during the use of Tralement, obtain serum concentrations of copper and ceruloplasmin as well as manganese whole blood concentrations. Consider using individual trace element products in patients with hepatic and/or biliary dysfunction [see Use in Specific Populations (8.6)].
5.5 Aluminum Toxicity
Tralement contains aluminum that may be toxic. Aluminum may reach toxic levels with prolonged parenteral administration if kidney function is impaired. Preterm infants, including preterm neonates, are particularly at risk because their kidneys are immature and they require large amounts of calcium and phosphate solutions, which contain aluminum.
Research indicates that patients with impaired kidney function, including preterm infants and premature neonates, who receive parenteral levels of aluminum at greater than 4 to 5 mcg/kg/day accumulate aluminum at levels associated with central nervous system and bone toxicity. Tissue loading may occur at even lower rates of administration or lower daily amounts.
Exposure to aluminum from Tralement is no more than 0.1 mcg/kg/day. When prescribing Tralement for use in parenteral nutrition containing other small volume parenteral products, the total daily patient exposure to aluminum from the admixture should be considered and maintained at no more than 5 mcg/kg/day [see Use in Specific Populations (8.4)].
5.6 Monitoring and Laboratory Tests
Monitor blood zinc, copper, manganese, and selenium concentrations, fluid and electrolyte status, serum osmolarity, blood glucose, liver and kidney function, blood count, and coagulation parameters during use of parenteral nutrition containing Tralement [see Dosage and Administration (2.4)].
5.7 Hypersensitivity Reactions with Zinc and Copper
Hypersensitivity reactions to subcutaneously administered zinc-containing insulin products and copper-containing intrauterine devices (IUD) were identified in postmarketing case reports. If hypersensitivity reactions occur in patients receiving Tralement in parenteral nutrition, discontinue Tralement, and initiate appropriate medical treatment [see Contraindications (4)].
Zinc
For patients prescribed zinc-containing insulin products, reported reactions included injection site induration, erythema, pruritus, papular rash, generalized urticaria, facial swelling, and dyspnea. Patients did not manifest symptoms after changing to zinc-free insulin or another insulin product with a reduced amount of zinc. In some cases, allergy testing confirmed the allergy to the zinc component of the insulin product.Copper
For women with copper IUD implantation, reported reactions included diffuse eczematous dermatitis, maculopapular skin eruption, urticaria, and angioedema of the eyelids, face, and labia weeks or months after IUD insertion. In most cases, the patch test for copper was positive, and the adverse reactions resolved after IUD removal. -
6 ADVERSE REACTIONS
The following adverse reactions were identified in clinical studies or post-marketing reports. Given that some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions with other components of parenteral nutrition solutions:
Pulmonary embolism due to pulmonary vascular precipitates [see Warnings and Precautions (5.1)]
Vein damage and thrombosis [see Warnings and Precautions (5.2)]
Aluminum toxicity [see Warnings and Precautions (5.5)]
Adverse reactions with the use of trace elements administered parenterally or by other routes of administration:
Neurologic toxicity with manganese [see Warnings and Precautions (5.3)]
Hepatic accumulation of copper and manganese [see Warnings and Precautions (5.4)]
Hypersensitivity reactions with zinc and copper [see Warnings and Precautions (5.7)] -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Administration of the recommended dose of Tralement in parenteral nutrition is not expected to cause major birth defects, miscarriage, or adverse maternal or fetal outcomes. Deficiency of trace elements may result in adverse pregnancy and fetal outcomes (see Clinical Considerations). Animal reproduction studies have not been conducted with Tralement or with the individual trace elements.The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-associated Maternal and/or Embryo-Fetal Risk
Deficiencies of trace elements, including zinc, copper, manganese, and selenium are associated with adverse pregnancy and fetal outcomes. Pregnant women have an increased metabolic demand for trace elements. Parenteral nutrition with Tralement should be considered if a pregnant woman’s nutritional requirements cannot be fulfilled by oral or enteral intake.8.2 Lactation
Risk Summary
Zinc, copper, manganese, and selenium are present in human milk. Administration of the approved recommended dose of Tralement in parenteral nutrition is not expected to cause harm to a breastfed infant. There is no information on the effects of zinc sulfate, cupric sulfate, manganese sulfate, or selenious acid on milk production. The developmental and health benefits of breastfeeding should be considered, along with the mother’s clinical need for Tralement and any potential adverse effects on the breastfed infant from Tralement or from the underlying maternal condition.
8.4 Pediatric Use
Tralement is approved for use in pediatric patients weighing at least 10 kg as a source of zinc, copper, manganese, and selenium for parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. Safety and dosing recommendations in pediatric patients are based on published literature describing controlled studies of products containing zinc, copper, manganese, and selenium [see Dosage and Administration (2.2)].
Tralement is not approved for use in pediatric patients weighing less than 10 kg because the product does not provide an adequate dosage of zinc, copper, or selenium to meet the needs of this subpopulation and exceeds the recommended dosage of manganese.
8.5 Geriatric Use
Reported clinical experience has not identified a difference in requirements for zinc, copper, manganese, or selenium between elderly and younger patients.
8.6 Hepatic Impairment
Copper is primarily excreted in the bile. Excretion is decreased in patients with cholestasis and/or cirrhosis. Manganese is found, and presumed to be excreted, in bile [see Clinical Pharmacology (12.3)]. Hepatic accumulation of copper and manganese have been reported with long-term administration in parenteral nutrition at dosages higher than recommended [see Warnings and Precautions (5.4)].
For patients with cholestasis, biliary dysfunction, or cirrhosis, monitor hepatic and biliary function during long-term administration of Tralement. If a patient develops signs or symptoms of hepatic or biliary dysfunction during use of Tralement, obtain serum concentrations of copper and ceruloplasmin as well as manganese whole blood concentrations. Consider using individual trace element products in patients with hepatic and/or biliary dysfunction.
-
10 OVERDOSAGE
There is no information on overdos Management of overdosage is supportive care based on presenting signs and symptoms. Obtain blood
samples for laboratory testing of the individual trace elements and ceruloplasmin for copper.Zinc
Acute zinc toxicity was reported in an infant who received an inadvertent 1,000-fold overdose of zinc in parenteral nutrition that led to cardiac failure and death. Zinc toxicity in adult patients receiving 17 to 400-fold the recommended dosage in parenteral nutrition for 2.5 to 60 days reported signs and
symptoms including vomiting, diarrhea, hyperamylasemia, thrombocytopenia, and anemia. The zinc serum concentration was 2 to 30-fold the upper end of the reported range in healthy subjects in these cases.Copper
Acute copper toxicity was reported in patients with oral, intravenous, or subcutaneous administration. Clinical manifestations included metallic taste, nausea, vomiting, abdominal pain, and multi-organ failure involving kidney, liver, blood, and cardiovascular systems. Chelating agents can be used for treatment of acute toxicity. Long-term administration of parenteral copper above recommended dosage may result in significant accumulation of copper in the liver, brain, and other tissues with possible organ damage [see Warnings and Precautions (5.4)].
Manganese
Acute manganese toxicity was reported in adult patients following infusion of manganese more than 10,000-fold the recommended dosage and after use of dialysis fluid contaminated with manganese. Signs and symptoms included skin flushing, acute pancreatitis, elevated whole blood manganese concentrations, and MRI evidence of brain accumulation of manganese. Chronic infusion and oral intake of manganese above recommended dosage have resulted in neuropsychiatric symptoms and MRI evidence of brain accumulation of manganese [see Warnings and Precautions (5.3)].Selenium
Acute selenium toxicity was reported with oral overdosage of greater than 1 g/day. Symptoms included nausea, vomiting, diarrhea, abdominal pain, garlic breath odor, and altered mental status. Death from circulatory collapse was reported after oral ingestion of 5 to 10 g of selenium with blood concentrations ranging 10 to 50-fold the upper end of the reported range in healthy subjects.
-
11 DESCRIPTION
Tralement® (trace elements injection 4*, USP) is a sterile, non-pyrogenic, clear, and colorless to slightly blue solution, intended for use as a combination of four trace elements and an additive to intravenous solutions for parenteral nutrition. It contains no preservative.
Each single-dose vial contains 1 mL and each multiple-dose vial contains 10 mL. *Each mL contains zinc 3 mg (equivalent to zinc sulfate 7.41 mg), copper 0.3 mg (equivalent to cupric sulfate 0.75 mg), manganese 55 mcg (equivalent to manganese sulfate 151 mcg), selenium 60 mcg (equivalent to selenious acid 98 mcg), and water for injection. Sulfuric acid may be added to adjust pH between 1.5 and 3.5.
Zinc sulfate exists as a heptahydrate. The structural formula is:
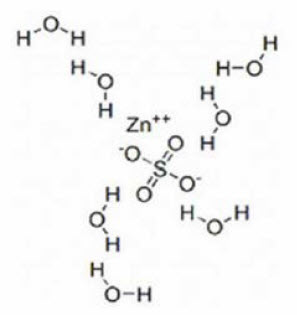
Molecular formula: ZnSO4 7H2O.
Molecular weight: 287.54 g/mol.Cupric sulfate exists as a pentahydrate. The structural formula is:
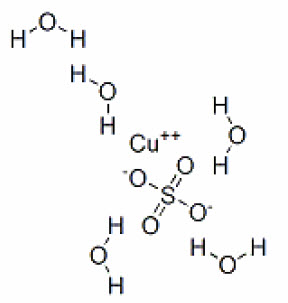
Molecular formula: CuSO4 5H2O.
Molecular weight: 249.69 g/mol.Manganese sulfate exists as a monohydrate. The structural formula is:
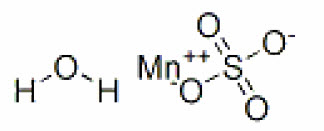
Molecular formula: MnSO4 H2O.
Molecular weight: 169.02 g/mol.
The structural formula of selenious acid is:
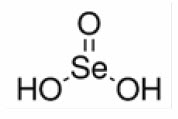
Molecular formula: H2SeO3.
Molecular weight: 128.97 g/mol.Tralement contains no more than 6,000 mcg/L of aluminum.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Zinc
Zinc functions as a cofactor of various enzymes including DNA polymerases, RNA polymerases, alcohol dehydrogenase, and alkaline phosphatases. Zinc is a coordinator of protein structural folding that interacts with a variety of proteins, lipids, and nucleic acids. In addition, zinc is a catalyst of essential biochemical reactions, including activation of substrates of carbonic anhydrase in erythrocyte.
Copper
Copper is a cofactor for many metalloenzymes acting as an oxidase to achieve reduction of molecular oxygen. Examples of copper metalloenzymes include but are not limited to lysyl oxidase, monoamine oxidase, ferroxidase, cytochrome C oxidase, dopamine beta monooxygenase, tyrosinase, and superoxide dismutase.\Manganese
Manganese is essential for the normal catalytic activity of several metalloenzymes including manganese superoxide dismutase, arginase, glutamine synthetase, phosphoenolpyruvate decarboxylase, and pyruvate carboxylase. Manganese contributes to the normal function of several other enzyme families
including the oxidoreductases, transferases, hydrolases, lyases, isomerases, and ligases.Selenium
Selenious acid is converted in vivo to hydrogen selenide via glutathione-involved electron reductions. Hydrogen selenide acts as a selenium pool to form selenoproteins which include, but are not limited to, glutathione peroxidase, iodothyronine deiodinase, peroxidase, and thioredoxins.12.2 Pharmacodynamics
The exposure-response relationship and the time course of pharmacodynamic response are unknown for zinc, copper, manganese, and selenium.
12.3 Pharmacokinetics
Zinc
Over 85% of total body zinc is found in skeletal muscle and bone. In blood, zinc is mainly localized within erythrocytes. Approximately 80% of serum zinc is bound to albumin and the remainder to α-2-macroglobulin and amino acids. In adults, zinc is primarily excreted via the gastrointestinal tract and eliminated in the feces. A smaller amount of zinc is excreted via the kidneys in the urine. Urinary zinc excretion rates in very low birth weight preterm infants are relatively high in the neonatal period, and they decline to a level on a body weight basis that is similar to that of normal adults by two months of age.
Copper
In plasma, about 7% of copper is bound to albumin and amino acids. In the liver, about 93% of copper is bound to ceruloplasmin and released to the serum. Copper is excreted in bile and into the gastrointestinal tract where it is not reabsorbed. Copper is also eliminated through the kidneys.
Manganese
Manganese is widely distributed in body tissues including liver and specific brain regions such as the basal ganglia. The concentrations of manganese are higher in erythrocytes compared to the plasma or serum concentrations. In human plasma, manganese is bound to albumin and β1-globulin. Manganese is
found in human bile suggesting biliary excretion.Selenium
In humans, 85% of intravenous administered 75Se was protein-bound within 4 to 6 hours and 95% by 24 hours. -
16 HOW SUPPLIED
Tralement (trace elements injection 4*, USP) is a clear, colorless to slightly blue solution supplied as follows:
NDC: 0517-9305-01 1 mL single-dose vial
NDC: 0517-9305-25 Packaged in trays containing 25 vials per tray
*Each mL of Tralement contains zinc 3 mg, copper 0.3 mg, manganese 55 mcg, and selenium 60 mcg.
Vial closure is not made with natural rubber latex.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature].
Store admixed solution at 2ºC to 8ºC (36ºF to 46ºF) [see Dosage and Administration (2.3)].
-
17 PATIENT COUNSELING INFORMATION
Inform patients, caregivers, and home healthcare providers of the following risks of Tralement:
Pulmonary embolism due to pulmonary vascular precipitates [see Warnings and Precautions (5.1)]
Vein damage and thrombosis [see Warnings and Precautions (5.2)]
Neurologic toxicity with manganese [see Warnings and Precautions (5.3)]
Hepatic accumulation of copper and manganese [see Warnings and Precautions (5.4)]
Aluminum toxicity [see Warnings and Precautions (5.5)]
Hypersensitivity reactions with zinc and copper [see Warnings and Precautions (5.7) - SPL UNCLASSIFIED SECTION
-
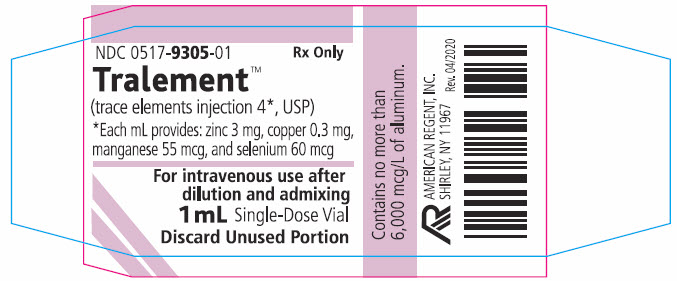
PRINCIPAL DISPLAY PANEL - Container Label (1 mL)
0517-9305-01
Rx Only
Tralement® (trace elements injection 4*, USP)
*Each mL provides: zinc 3 mg, copper 0.3 mg, manganese 55 mcg, and selenium 60 mcgContains no more than 6,000 mcg/L of aluminum.
For intravenous infusion after admixingFor use in adult and pediatric patients weighing at least 10 kg
1 mL Single-Dose Vial
Discard Unused Portion

-
PRINCIPAL DISPLAY PANEL - Carton Labeling (1 mL)
0517-9305-25
Rx Only
Tralement® (trace elements injection 4*, USP)
*Each mL provides: zinc 3 mg, copper 0.3 mg, manganese 55 mcg, and selenium 60 mcgFor intravenous infusion after admixing
For use in adult and pediatric patients weighing at least 10 kg
25 x 1 mL Single-Dose Vials
Discard Unused Portion

-
INGREDIENTS AND APPEARANCE
TRALEMENT
trace elements injection 4 injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0517-9305 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC SULFATE (UNII: 89DS0H96TB) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 3 mg in 1 mL CUPRIC SULFATE (UNII: LRX7AJ16DT) (CUPRIC CATION - UNII:8CBV67279L) CUPRIC CATION 0.3 mg in 1 mL MANGANESE SULFATE (UNII: W00LYS4T26) (MANGANOUS CATION - UNII:H6EP7W5457) MANGANOUS CATION 55 ug in 1 mL SELENIOUS ACID (UNII: F6A27P4Q4R) (SELENIOUS ACID - UNII:F6A27P4Q4R) SELENIOUS ACID 60 ug in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0517-9305-25 25 in 1 TRAY 09/29/2020 1 NDC: 0517-9305-01 1 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209376 09/29/2020 Labeler - American Regent, Inc. (002033710) Establishment Name Address ID/FEI Business Operations American Regent, Inc. 002033710 analysis(0517-9305) , manufacture(0517-9305)
Trademark Results [Tralement]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 TRALEMENT 88181165 not registered Live/Pending |
Luitpold Pharmaceuticals, Inc. 2018-11-05 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.