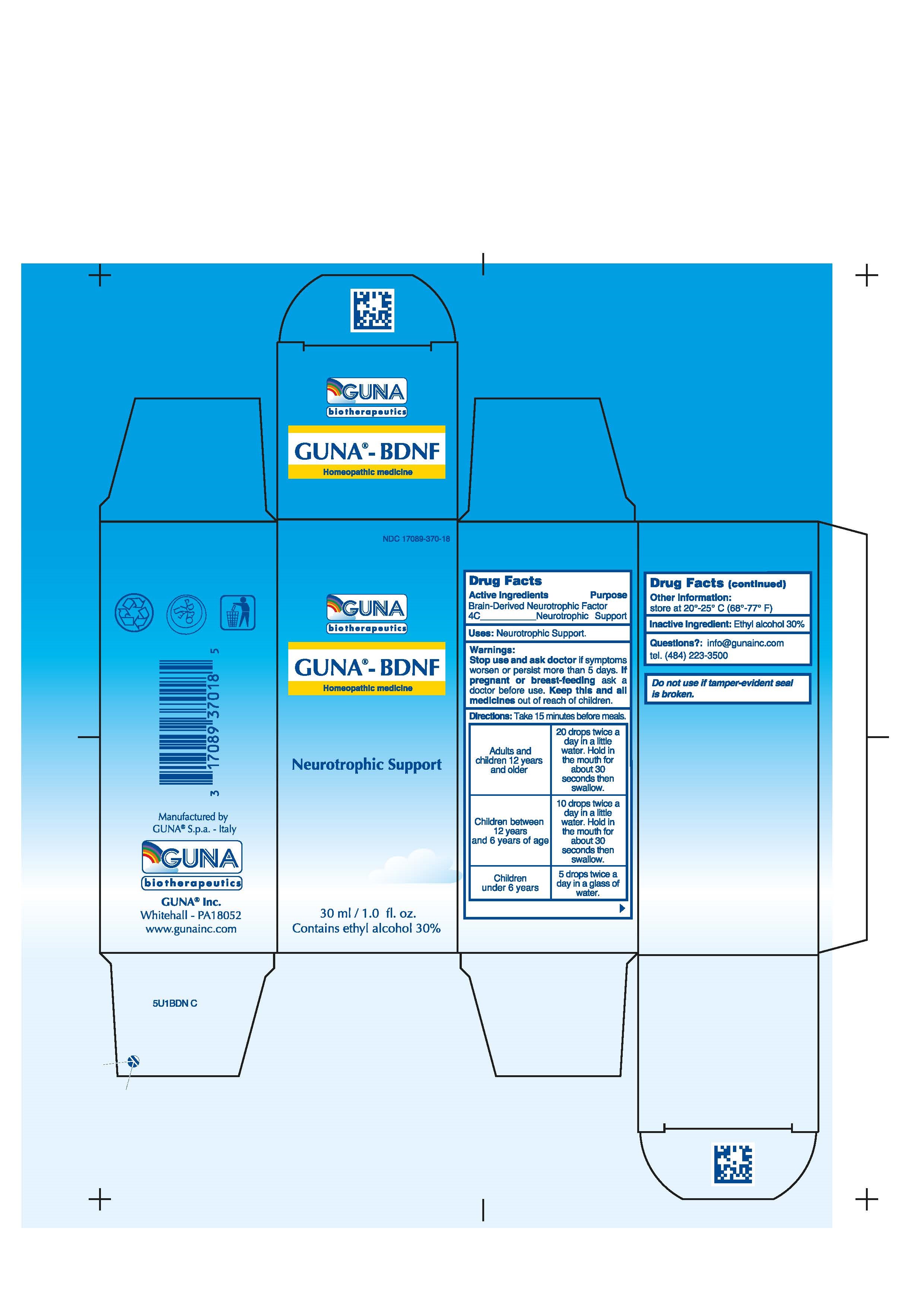
GUNA-BDNF- brain-derived neurotrophic factor solution/ drops
GUNA-BDNF by
Drug Labeling and Warnings
GUNA-BDNF by is a Homeopathic medication manufactured, distributed, or labeled by Guna spa. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
-
DIRECTIONS
Take 15 minutes before meals.
Adults and children 12 years and older 20 drops twice a day in a little water. Hold in the mouth for about 30 seconds then swallow.
Children between 12 years and 6 years of age 10 drops twice a day in a little water. Hold in the mouth for about 30 seconds then swallow.
Children under 6 years 5 drops twice a day in a glass of water. - INACTIVE INGREDIENT
- INDICATIONS & USAGE
-
DOSAGE & ADMINISTRATION
Adults and children twelve years and older: 20 drops twice a day in a little water- Hold in the mouth for about 30 seconds then swallow.
Children between 12 years and 6 years of age: 10 drops twice a day in a little water- Hold in the mouth for about 30 seconds then swallow.
Children under 6 years: 5 drops twice a day in a glass of water.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GUNA-BDNF
brain-derived neurotrophic factor solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 17089-370 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BRAIN-DERIVED NEUROTROPHIC FACTOR HUMAN (UNII: 7171WSG8A2) (BRAIN-DERIVED NEUROTROPHIC FACTOR HUMAN - UNII:7171WSG8A2) BRAIN-DERIVED NEUROTROPHIC FACTOR HUMAN 4 [hp_C] in 30 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) 9 mL in 30 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 17089-370-18 1 in 1 BOX 03/31/2011 1 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/31/2011 Labeler - Guna spa (430538264) Establishment Name Address ID/FEI Business Operations Guna spa 338587646 manufacture(17089-370)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.