SODIUM CHLORIDE injection
Sodium Chloride by
Drug Labeling and Warnings
Sodium Chloride by is a Prescription medication manufactured, distributed, or labeled by US MedSource, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- BOXED WARNING (What is this?)
-
DESCRIPTION
This preparation is designed for parenteral use only after addition of drugs that require dilution or must be dissolved in an aqueous vehicle prior to injection.
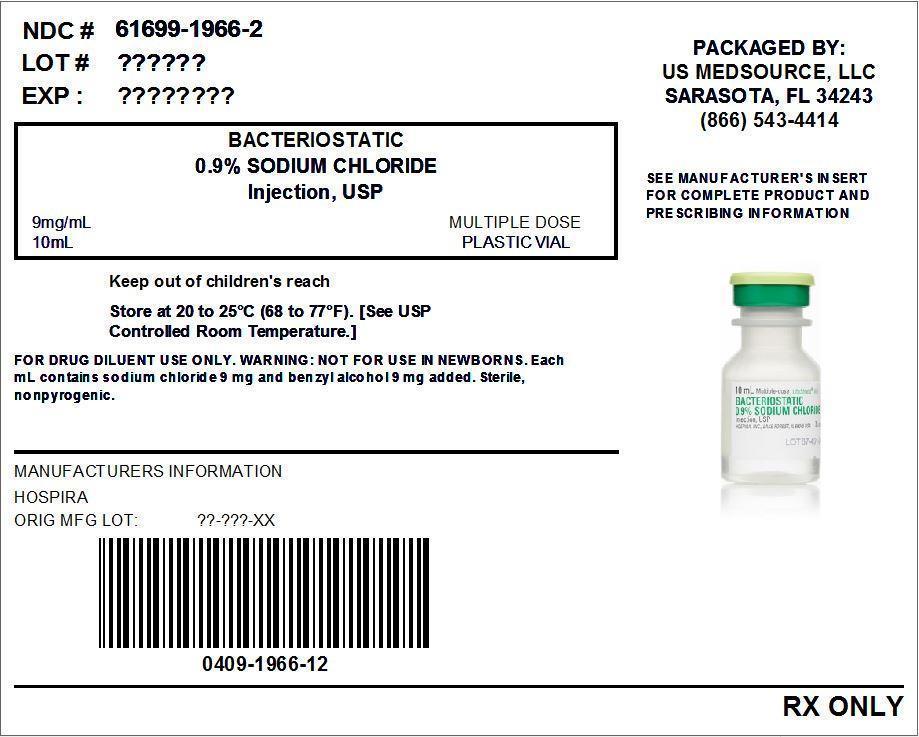
Bacteriostatic 0.9% Sodium Chloride Injection, USP is a sterile, nonpyrogenic, isotonic solution of sodium chloride in water for injection. Each milliliter (mL) contains sodium chloride 9 mg and 0.9% (9 mg/mL) benzyl alcohol added as a bacteriostatic preservative. May contain hydrochloric acid for pH adjustment. It is supplied in a multiple-dose container from which repeated withdrawals may be made to dilute or dissolve drugs for medication. The pH is 5.0 (4.5 to 7.0).
Sodium Chloride, USP is chemically designated NaCl, a white crystalline powder freely soluble in water.
The semi-rigid vial is fabricated from a specially formulated polyolefin. It is a copolymer of ethylene and propylene. The safety of the plastic has been confirmed by tests in animals according to USP biological standards for plastic containers. The container requires no vapor barrier to maintain the proper drug concentration.
-
CLINICAL PHARMACOLOGY
Sodium chloride in water dissociates to provide sodium (Na+) and chloride (Cl−) ions. These ions are normal constituents of the body fluids (principally extracellular) and are essential for maintaining electrolyte balance.
The distribution and excretion of sodium (Na+) and chloride (Cl−) are largely under the control of the kidney which maintains a balance between intake and output.
The small volume of fluid and amount of sodium chloride provided by Bacteriostatic 0.9% Sodium Chloride Injection, USP, when used only as a vehicle for parenteral injection of drugs, is unlikely to exert a significant effect on fluid and electrolyte balance except possibly in neonates and very small infants.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight. Average normal adult daily requirement ranges from two to three liters (1.0 to 1.5 liters each for insensible water loss by perspiration and urine production).
Water balance is maintained by various regulatory mechanisms. Water distribution depends primarily on the concentration of electrolytes in the body compartments and sodium (Na+) plays a major role in maintaining physiologic equilibrium.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Due to the potential toxicity of benzyl alcohol in neonates, solutions containing benzyl alcohol must not be used in this patient population.
Parenteral preparations with benzyl alcohol should not be used for fluid or sodium chloride replacement.
Parenteral preparations containing benzyl alcohol should not be used in epidural or spinal anesthetic procedures.
-
WARNINGS
Benzyl alcohol, a preservative in Bacteriostatic Sodium Chloride Injection, USP has been associated with toxicity in neonates. Data are unavailable on the toxicity of other preservatives in this age group. Preservative-free Sodium Chloride Injection should be used for flushing intravascular catheters. Where a sodium chloride solution is required for preparing or diluting medications for use in neonates, only preservative-free Sodium Chloride Injection should be used.
-
PRECAUTIONS
Consult the manufacturer’s instructions for choice of vehicle, appropriate dilution or volume for dissolving the drugs to be injected, including the route and rate of injection.
Inspect reconstituted (diluted or dissolved) drugs for clarity (if soluble) and freedom from unexpected precipitation or discoloration prior to administration.
Pregnancy Category C. Animal reproduction studies have not been conducted with Bacteriostatic 0.9% Sodium Chloride Injection, USP. It is also not known whether Bacteriostatic 0.9% Sodium Chloride Injection containing additives can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Bacteriostatic 0.9% Sodium Chloride Injection containing additives should be given to a pregnant woman only if clearly needed.
Pediatric Use
The safety and effectiveness in the pediatric population are based on the similarity of the clinical conditions of the pediatric and adult populations. However, due to potential toxicity of benzyl alcohol in neonates, solutions containing benzyl alcohol are contraindicated in this patient population.
Drug Interactions
Some drugs for injection may be incompatible in a given vehicle, or when combined in the same vehicle or in a vehicle containing benzyl alcohol. Consult with pharmacist, if available.
Use aseptic technique for single or multiple entry and withdrawal from all containers.
When diluting or dissolving drugs, mix thoroughly and use promptly.
Do not store reconstituted solutions of drugs for injection unless otherwise directed by the manufacturer of the solute.
Do not use unless the solution is clear and seal intact.
-
ADVERSE REACTIONS
Reactions which may occur because of this solution, added drugs or the technique of reconstitution or administration include febrile response, local tenderness, abscess, tissue necrosis or infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection and extravasation.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate countermeasures, and if possible, retrieve and save the remainder of the unused vehicle for examination.
Although adverse reactions to intravenous, intramuscular or subcutaneous injection of 0.9% benzyl alcohol are not known to occur in man, experimental studies of small volume parenteral preparations containing 0.9% benzyl alcohol in several species of animals have indicated that an estimated intravenous dose up to 30 mL may be safely given to an adult without toxic effects. Administration of an estimated 9 mL to a 6 kg neonate or infant is potentially capable of producing blood pressure changes.
-
OVERDOSAGE
Use only as a diluent or solvent. This parenteral preparation is unlikely to pose a threat of sodium chloride or fluid overload except possibly in neonates and very small infants. In the event these should occur, re-evaluate the patient and institute appropriate corrective measures. See PRECAUTIONS and ADVERSE REACTIONS.
-
DOSAGE AND ADMINISTRATION
The volume of the preparation to be used for diluting or dissolving any drug for injection, is dependent on the vehicle concentration, dose and route of administration as recommended by the manufacturer.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. See PRECAUTIONS.
-
HOW SUPPLIED
Bacteriostatic 0.9% Sodium Chloride Injection, USP is supplied in multiple-dose 10, 20 and 30 mL plastic fliptop vials (List No. 1966) or in multiple-dose 10 and 30 mL plastic LifeShield® fliptop vials* (List No. 1966).
*Intended for use with the LifeShield® Blunt Cannula.
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.]
Revised: April, 2005
©Hospira 2005 EN-1169 Printed in USA
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
- Package Label
-
INGREDIENTS AND APPEARANCE
SODIUM CHLORIDE
sodium chloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 61699-1966(NDC:0409-1966) Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 9 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) 9 mg in 1 mL WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 61699-1966-2 1 in 1 BAG 1 10 mL in 1 VIAL, MULTI-DOSE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018800 07/25/2014 Labeler - US MedSource, LLC (032793851) Establishment Name Address ID/FEI Business Operations US MedSource, LLC 032793851 repack(61699-1966)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.